Abstract
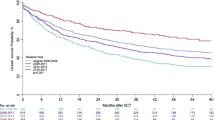
Autologous stem cell transplantation (autoSCT) can achieve long-term remission in primary refractory or relapsed Hodgkin lymphoma (r/r HL); however, still up to 50% of patients relapse after autoSCT. In this retrospective analysis, we investigated the impact of autologous stem cell transplantation in a consecutive, unselected cohort of primary refractory and relapsed Hodgkin lymphoma patients (n = 66) with the majority of patients treated in the pre-brentuximab vedotin and immune checkpoint inhibitor era. In our cohort, a 5-year overall survival (OS) from autoSCT of 59.5% and a 5-year progression-free survival (PFS) after autoSCT of 46.1% was achieved. Multivariate analysis revealed primary refractory disease and early relapse (< 12 months) after initial therapy as well as the presence of B symptoms at relapse as independent risk factors associated with a higher risk for relapse and an inferior PFS and OS. Several other clinical factors, including the presence of extranodal disease at relapse and failure to achieve a complete response to salvage chemotherapy, were associated with a trend towards an inferior survival. Patients relapsing after autoSCT had a particularly poor outcome, regardless of eligibility to undergo allogeneic stem cell transplantation (alloSCT). We further evaluated recently published prognostic models for r/r HL patients undergoing autoSCT and could validate several risk scores in our independent “real world” cohort.




Similar content being viewed by others
References
Engert A, Diehl V, Franklin J, Lohri A, Dörken B, Ludwig W-D, Koch P, Hänel M, Pfreundschuh M, Wilhelm M (2009) Escalated-dose BEACOPP in the treatment of patients with advanced-stage Hodgkin’s lymphoma: 10 years of follow-up of the GHSG HD9 study. J Clin Oncol 27(27):4548–4554
Meyer RM, Gospodarowicz MK, Connors JM, Pearcey RG, Bezjak A, Wells WA, Burns BF, Winter JN, Horning SJ, Dar AR (2005) Randomized comparison of ABVD chemotherapy with a strategy that includes radiation therapy in patients with limited-stage Hodgkin’s lymphoma: National Cancer Institute of Canada clinical trials Group and the Eastern Cooperative Oncology Group. J Clin Oncol 23(21):4634–4642
Eichenauer D, Aleman B, André M, Federico M, Hutchings M, Illidge T, Engert A, Ladetto M (2018) Hodgkin lymphoma: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 29(Suppl 4):iv19–iv29. https://doi.org/10.1093/annonc/mdy080
Kuruvilla J, Keating A, Crump M (2011) How I treat relapsed and refractory Hodgkin lymphoma. Blood 117(16):4208–4217
Younes A, Gopal AK, Smith SE, Ansell SM, Rosenblatt JD, Savage KJ, Ramchandren R, Bartlett NL, Cheson BD, De Vos S (2012) Results of a pivotal phase II study of brentuximab vedotin for patients with relapsed or refractory Hodgkin’s lymphoma. J Clin Oncol 30(18):2183–2189
Herrera A, Palmer J, Martin P, Armenian S, Tsai N-C, Kennedy N, Sahebi F, Cao T, Budde L, Mei M (2017) Autologous stem-cell transplantation after second-line brentuximab vedotin in relapsed or refractory Hodgkin lymphoma. Ann Oncol 29(3):724–730
Bierman P, Lynch J, Bociek R, Whalen V, Kessinger A, Vose J, Armitage J (2002) The international prognostic factors project score for advanced Hodgkin’s disease is useful for predicting outcome of autologous hematopoietic stem cell transplantation. Ann Oncol 13(9):1370–1377
Hahn T, McCarthy PL, Carreras J, Zhang M-J, Lazarus HM, Laport GG, Montoto S, Hari PN (2013) Simplified validated prognostic model for progression-free survival after autologous transplantation for hodgkin lymphoma. Biol Blood Marrow Transplant 19(12):1740–1744
Sureda A, Constans M, Iriondo A, Arranz R, Caballero M, Vidal M, Petit J, Lopez A, Lahuerta J, Carreras E (2005) Prognostic factors affecting long-term outcome after stem cell transplantation in Hodgkin's lymphoma autografted after a first relapse. Ann Oncol 16(4):625–633
Moskowitz CH, Nademanee A, Masszi T, Agura E, Holowiecki J, Abidi MH, Chen AI, Stiff P, Gianni AM, Carella A (2015) Brentuximab vedotin as consolidation therapy after autologous stem-cell transplantation in patients with Hodgkin’s lymphoma at risk of relapse or progression (AETHERA): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 385(9980):1853–1862
Armand P, Engert A, Younes A, Fanale M, Santoro A, Zinzani PL, Timmerman JM, Collins GP, Ramchandren R, Cohen JB (2018) Nivolumab for relapsed/refractory classic Hodgkin lymphoma after failure of autologous hematopoietic cell transplantation: extended follow-up of the multicohort single-arm phase II CheckMate 205 trial. J Clin Oncol 36(14):1428
Ansell SM, Lesokhin AM, Borrello I, Halwani A, Scott EC, Gutierrez M, Schuster SJ, Millenson MM, Cattry D, Freeman GJ (2015) PD-1 blockade with nivolumab in relapsed or refractory Hodgkin’s lymphoma. N Engl J Med 372(4):311–319
Bröckelmann P, Müller H, Casasnovas O, Hutchings M, von Tresckow B, Jürgens M, McCall S, Morschhauser F, Fuchs M, Borchmann P (2017) Risk factors and a prognostic score for survival after autologous stem-cell transplantation for relapsed or refractory Hodgkin lymphoma. Ann Oncol 28(6):1352–1358
Satwani P, Ahn KW, Carreras J, Abdel-Azim H, Cairo MS, Cashen A, Chen AI, Cohen JB, Costa LJ, Dandoy C (2015) A prognostic model predicting autologous transplantation outcomes in children, adolescents and young adults with Hodgkin lymphoma. Bone Marrow Transplant 50(11):1416–1423
Hahn T, Benekli M, Wong C, Moysich K, Hyland A, Michalek A, Alam A, Baer M, Bambach B, Czuczman M (2005) A prognostic model for prolonged event-free survival after autologous or allogeneic blood or marrow transplantation for relapsed and refractory Hodgkin’s disease. Bone Marrow Transplant 35(6):557–566
Josting A, Franklin J, May M, Koch P, Beykirch MK, Heinz J, Rudolph C, Diehl V, Engert A (2002) New prognostic score based on treatment outcome of patients with relapsed Hodgkin’s lymphoma registered in the database of the German Hodgkin’s lymphoma study group. J Clin Oncol 20(1):221–230
Colwill R, Crump M, Couture F, Danish R, Stewart AK, Sutton D, Scott JG, Sutcliffe SB, Brandwein JM, Keating A (1995) Mini-BEAM as salvage therapy for relapsed or refractory Hodgkin’s disease before intensive therapy and autologous bone marrow transplantation. J Clin Oncol 13(2):396–402
Crump M, Smith AM, Brandwein J, Couture F, Sherret H, Sutton D, Scott JG, McCrae J, Murray C, Pantalony D (1993) High-dose etoposide and melphalan, and autologous bone marrow transplantation for patients with advanced Hodgkin’s disease: importance of disease status at transplant. J Clin Oncol 11(4):704–711
Kuruvilla J, Nagy T, Pintilie M, Tsang R, Keating A, Crump M (2006) Similar response rates and superior early progression-free survival with gemcitabine, dexamethasone, and cisplatin salvage therapy compared with carmustine, etoposide, cytarabine, and melphalan salvage therapy prior to autologous stem cell transplantation for recurrent or refractory Hodgkin lymphoma. Cancer 106(2):353–360
Cheson BD, Fisher RI, Barrington SF, Cavalli F, Schwartz LH, Zucca E, Lister TA (2014) Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol 32(27):3059
Cheson BD, Ansell S, Schwartz L, Gordon LI, Advani R, Jacene HA, Hoos A, Barrington SF, Armand P (2016) Refinement of the Lugano classification lymphoma response criteria in the era of immunomodulatory therapy. Blood 128(21):2489–2496
Brugger W, Bross K, Frisch J, Dern P, Weber B, Mertelsmann R, Kanz L (1992) Mobilization of peripheral blood progenitor cells by sequential administration of interleukin-3 and granulocyte-macrophage colony-stimulating factor following polychemotherapy with etoposide, ifosfamide, and cisplatin. Blood 79(5):1193–1200
Reece DE, Connors JM, Spinelli JJ, Barnett MJ, Fairey RN, Klingemann HG, Nantel SH, O'Reilly S, Shepherd JD, Sutherland HJ et al (1994) Intensive therapy with cyclophosphamide, carmustine, etoposide +/− cisplatin, and autologous bone marrow transplantation for Hodgkin's disease in first relapse after combination chemotherapy. Blood 83(5):1193–1199
Linch D, Goldstone A, McMillan A, Chopra R, Hudson GV, Winfield D, Hancock B, Moir D, Milligan D (1993) Dose intensification with autologous bone-marrow transplantation in relapsed and resistant Hodgkin’s disease: results of a BNLI randomised trial. Lancet 341(8852):1051–1054
Schmitz N, Pfistner B, Sextro M, Sieber M, Carella AM, Haenel M, Boissevain F, Zschaber R, Müller P, Kirchner H (2002) Aggressive conventional chemotherapy compared with high-dose chemotherapy with autologous haemopoietic stem-cell transplantation for relapsed chemosensitive Hodgkin's disease: a randomised trial. Lancet 359(9323):2065–2071
Elstrom RL, Martin P, Rua SH, Shore TB, Furman RR, Ruan J, Pearse RN, Coleman M, Mark T, Leonard JP (2012) Autologous stem cell transplant is feasible in very elderly patients with lymphoma and limited comorbidity. Am J Hematol 87(4):433–435
Puig N, Pintilie M, Seshadri T, Al-Farsi K, Franke N, Keating A, Kuruvilla J, Crump M (2011) High-dose chemotherapy and auto-SCT in elderly patients with Hodgkin’s lymphoma. Bone Marrow Transplant 46(10):1339–1344
Stamatoullas A, Brice P, Gueye M, Mareschal S, Chevallier P, Bouabdallah R, Nguyenquoc S, Francois S, Turlure P, Ceballos P (2016) Autologous stem cell transplantation for patients aged 60 years or older with refractory or relapsed classical Hodgkin’s lymphoma: a retrospective analysis from the French Society of Bone Marrow Transplantation and Cell Therapies (SFGM-TC). Bone Marrow Transplant 51(7):928–932
Horning SJ, Chao NJ, Negrin RS, Hoppe RT, Long GD, Hu WW, Wong RM, Brown BW, Blume KG (1997) High-dose therapy and autologous hematopoietic progenitor cell transplantation for recurrent or refractory Hodgkin's disease: analysis of the Stanford University results and prognostic indices. Blood 89(3):801–813
Tarella C, Cuttica A, Vitolo U, Liberati M, Di Nicola M, Cortelazzo S, Rosato R, Rosanelli C, Di Renzo N, Musso M (2003) High-dose sequential chemotherapy and peripheral blood progenitor cell autografting in patients with refractory and/or recurrent Hodgkin lymphoma: a multicenter study of the intergruppo Italiano Linfomi showing prolonged disease free survival in patients treated at first recurrence. Cancer 97(11):2748–2759
Chan FC, Mottok A, Gerrie AS, Power M, Nijland M, Diepstra A, van den Berg A, Kamper P, d'Amore F, d'Amore AL (2017) Prognostic model to predict post-autologous stem-cell transplantation outcomes in classical Hodgkin lymphoma. J Clin Oncol 35(32):3722–3733
Garcia-Sanz R, Sureda A, de la Cruz F, Canales M, Gonzalez A, Pinana J, Rodriguez A, Gutierrez A, Domingo-Domenech E, Sanchez-Gonzalez B (2019) Brentuximab vedotin and ESHAP is highly effective as second-line therapy for Hodgkin lymphoma patients (long-term results of a trial by the Spanish GELTAMO group). Ann Oncol 30(4):612–620
Younes A, Bartlett NL, Leonard JP, Kennedy DA, Lynch CM, Sievers EL, Forero-Torres A (2010) Brentuximab vedotin (SGN-35) for relapsed CD30-positive lymphomas. N Engl J Med 363(19):1812–1821
Ansell SM (2017) Nivolumab in the treatment of Hodgkin lymphoma. Clin Cancer Res 23(7):1623–1626
Vardhana S, Younes A (2016) The immune microenvironment in Hodgkin lymphoma: T cells, B cells, and immune checkpoints. Haematologica 101(7):794–802
Moskowitz CH, Nimer SD, Zelenetz AD, Trippett T, Hedrick EE, Filippa DA, Louie D, Gonzales M, Walits J, Coady-Lyons N (2001) A 2-step comprehensive high-dose chemoradiotherapy second-line program for relapsed and refractory Hodgkin disease: analysis by intent to treat and development of a prognostic model. Blood 97(3):616–623
Lohri A, Barnett M, Fairey R, O'Reilly S, Phillips G, Reece D, Voss N, Connors J (1991) Outcome of treatment of first relapse of Hodgkin’s disease after primary chemotherapy: identification of risk factors from the British Columbia experience 1970 to 1988. Blood 77(10):2292–2298
Popat U, Hosing C, Saliba RM, Anderlini P, Van Besien K, Przepiorka D, Khouri IF, Gajewski J, Claxton D, Giralt S (2004) Prognostic factors for disease progression after high-dose chemotherapy and autologous hematopoietic stem cell transplantation for recurrent or refractory Hodgkin’s lymphoma. Bone Marrow Transplant 33(10):1015–1023
Pfreundschuh MG, Rueffer U, Larhan B, Schmitz N, Brosteanu O, Hasenclever D, Haas R, Kirchner H, Koch P, Kuse R (1994) Dexa-BEAM inpatients with Hodgkin’s disease refractory to multidrug chemotherapy regimens: a trial of the German Hodgkin’s Disease Study Group. J Clin Oncol 12(3):580–586
Ribrag V, Nasr F, Bouhris J, Bosq J, Brault P, Girinsky T, Cosset J, Munck J, Corti C, Decaudin D (1998) VIP (etoposide, ifosfamide and cisplatinum) as a salvage intensification program in relapsed or refractory Hodgkin’s disease. Bone Marrow Transplant 21(10):969–974
LaCasce AS, Bociek RG, Sawas A, Caimi P, Agura E, Matous J, Ansell SM, Crosswell HE, Islas-Ohlmayer M, Behler C (2018) Brentuximab vedotin plus bendamustine: a highly active first salvage regimen for relapsed or refractory Hodgkin lymphoma. Blood 132(1):40–48
Santoro A, Mazza R, Pulsoni A, Re A, Bonfichi M, Zilioli VR, Salvi F, Merli F, Anastasia A, Luminari S, Annechini G, Gotti M, Peli A, Liberati AM, Di Renzo N, Castagna L, Giordano L, Carlo-Stella C (2016) Bendamustine in combination with gemcitabine and vinorelbine is an effective regimen as induction chemotherapy before autologous stem-cell transplantation for relapsed or refractory Hodgkin lymphoma: final results of a multicenter phase II study. J Clin Oncol 34(27):3293–3299. https://doi.org/10.1200/jco.2016.66.4466
Herrera AF, Moskowitz AJ, Bartlett NL, Vose JM, Ramchandren R, Feldman TA, LaCasce AS, Ansell SM, Moskowitz CH, Fenton K (2018) Interim results of brentuximab vedotin in combination with nivolumab in patients with relapsed or refractory Hodgkin lymphoma. Blood 131(11):1183–1194
Josting A, Rudolph C, Reiser M, Mapara M, Sieber M, Kirchner H, Dörken B, Hossfeld D, Diehl V, Engert A (2002) Time-intensified dexamethasone/cisplatin/cytarabine: an effective salvage therapy with low toxicity in patients with relapsed and refractory Hodgkin’s disease. Ann Oncol 13(10):1628–1635
Olivieri A, Brunori M, Capelli D, Montanari M, Massidda D, Gini G, Lucesole M, Poloni A, Offidani M, Candela M (2004) Salvage therapy with an outpatient DHAP schedule followed by PBSC transplantation in 79 lymphoma patients: an intention to mobilize and transplant analysis. Eur J Haematol 72(1):10–17
Brandwein J, Callum J, Sutcliffe S, Scott J, Keating A (1990) Evaluation of cytoreductive therapy prior to high dose treatment with autologous bone marrow transplantation in relapsed and refractory Hodgkin’s disease. Bone Marrow Transplant 5(2):99–103
Stewart AK, Brandwein JM, Sutcliffe SB, Scott JG, Keating A (1991) Mini-beam as salvage chemotherapy for refractory Hodgkin’s disease and non-Hodgkin’s lymphoma. Leuk Lymphoma 5(2–3):111–115
Grn A, Ambinder RF, Piantadosi S, Abrams RA, Brodsky RA, Vogelsang GB, Zahurak ML, Fuller D, Miller CB, Noga SJ (2001) Long-term results of blood and marrow transplantation for Hodgkin’s lymphoma. J Clin Oncol 19(23):4314–4321
Gajewski JL, Phillips GL, Sobocinski KA, Armitage JO, Gale RP, Champlin RE, Herzig RH, Hurd DD, Jagannath S, Klein JP (1996) Bone marrow transplants from HLA-identical siblings in advanced Hodgkin’s disease. J Clin Oncol 14(2):572–578
Peniket A, De Elvira MR, Taghipour G, Cordonnier C, Gluckman E, De Witte T, Santini G, Blaise D, Greinix H, Ferrant A (2003) An EBMT registry matched study of allogeneic stem cell transplants for lymphoma: allogeneic transplantation is associated with a lower relapse rate but a higher procedure-related mortality rate than autologous transplantation. Bone Marrow Transplant 31(8):667–678
Rivas MM, Berro M, Prates MV, Yantorno S, Fiad L, Arbelbide JA, Basquiera AL, Ferini GA, García JJ, García PA (2019) Allogeneic stem cell transplantation improves survival in relapsed Hodgkin lymphoma patients achieving complete remission after salvage treatment. Bone Marrow Transplant. https://doi.org/10.1038/s41409-019-0640-z
Covut F, Pinto R, Cooper BW, Tomlinson B, Metheny L, Malek E, Lazarus HM, de Lima M, Caimi PF (2017) Nivolumab before and after allogeneic hematopoietic cell transplantation. Bone Marrow Transplant 52(7):1054–1056. https://doi.org/10.1038/bmt.2017.44
El Cheikh J, Massoud R, Abudalle I, Haffar B, Mahfouz R, Kharfan-Dabaja MA, Jisr T, Mougharbel A, Ibrahim A, Bazarbachi A (2017) Nivolumab salvage therapy before or after allogeneic stem cell transplantation in Hodgkin lymphoma. Bone Marrow Transplant 52(7):1074–1077. https://doi.org/10.1038/bmt.2017.69
Merryman RW, Kim HT, Zinzani PL, Carlo-Stella C, Ansell SM, Perales MA, Avigdor A, Halwani AS, Houot R, Marchand T, Dhedin N, Lescaut W, Thiebaut-Bertrand A, Francois S, Stamatoullas-Bastard A, Rohrlich PS, Labussiere Wallet H, Castagna L, Santoro A, Bachanova V, Bresler SC, Srivastava A, Kim H, Pesek E, Chammas M, Reynolds C, Ho VT, Antin JH, Ritz J, Soiffer RJ, Armand P (2017) Safety and efficacy of allogeneic hematopoietic stem cell transplant after PD-1 blockade in relapsed/refractory lymphoma. Blood 129(10):1380–1388. https://doi.org/10.1182/blood-2016-09-738385
Ijaz A, Khan AY, Malik SU, Faridi W, Fraz MA, Usman M, Tariq MJ, Durer S, Durer C, Russ A (2019) Significant risk of graft-versus-host disease with exposure to checkpoint inhibitors before and after allogeneic transplantation. Biol Blood Marrow Transplant 25(1):94–99
Merryman RW, Armand P (2017) Immune checkpoint blockade and hematopoietic stem cell transplant. Curr Hematol Malignancy Rep 12(1):44–50
Bauml J, Seiwert TY, Pfister DG, Worden F, Liu SV, Gilbert J, Saba NF, Weiss J, Wirth L, Sukari A (2017) Pembrolizumab for platinum-and cetuximab-refractory head and neck cancer: results from a single-arm, phase II study. J Clin Oncol 35(14):1542
Langer C, Gadgeel S, Borghaei H, Papadimitrakopoulou V, Patnaik A, Powell S, Gentzler R, Martins R, Stevenson J, Jalal S (2016) KEYNOTE-021 investigators. Carboplatin and pemetrexed with or without pembrolizumab for advanced, non-squamous non-small-cell lung cancer: a randomised, phase 2 cohort of the open-label KEYNOTE-021 study. Lancet Oncol 17(11):1497–1508
Schvartsman G, Peng SA, Bis G, Lee JJ, Benveniste MF, Zhang J, Roarty EB, Lacerda L, Swisher S, Heymach JV (2017) Response rates to single-agent chemotherapy after exposure to immune checkpoint inhibitors in advanced non-small cell lung cancer. Lung Cancer 112:90–95
Yan Y, Kumar AB, Finnes H, Markovic SN, Park S, Dronca RS, Dong H (2018) Combining immune checkpoint inhibitors with conventional cancer therapy. Front Immunol 9:1739. https://doi.org/10.3389/fimmu.2018.01739
Yan Y, Cao S, Liu X, Harrington SM, Bindeman WE, Adjei AA, Jang JS, Jen J, Li Y, Chanana P (2018) CX3CR1 identifies PD-1 therapy–responsive CD8+ T cells that withstand chemotherapy during cancer chemoimmunotherapy. JCI Insight 3(8). https://doi.org/10.1172/jci.insight.97828
Derer A, Frey B, Fietkau R, Gaipl US (2016) Immune-modulating properties of ionizing radiation: rationale for the treatment of cancer by combination radiotherapy and immune checkpoint inhibitors. Cancer Immunol Immunother 65(7):779–786
Twyman-Saint Victor C, Rech AJ, Maity A, Rengan R, Pauken KE, Stelekati E, Benci JL, Xu B, Dada H, Odorizzi PM (2015) Radiation and dual checkpoint blockade activate non-redundant immune mechanisms in cancer. Nature 520(7547):373–377
Alvarez I, Sureda A, Caballero MD, Urbano-Ispizua A, Ribera JM, Canales M, García-Conde J, Sanz G, Arranz R, Bernal MT (2006) Nonmyeloablative stem cell transplantation is an effective therapy for refractory or relapsed hodgkin lymphoma: results of a Spanish prospective cooperative protocol. Biol Blood Marrow Transplant 12(2):172–183
Anderlini P, Saliba R, Acholonu S, Giralt SA, Andersson B, Ueno NT, Hosing C, Khouri IF, Couriel D, De Lima M (2008) Fludarabine-melphalan as a preparative regimen for reduced-intensity conditioning allogeneic stem cell transplantation in relapsed and refractory Hodgkin’s lymphoma: the updated MD Anderson Cancer Center experience. Haematologica 93(2):257–264
Chen R, Palmer JM, Popplewell L, Shen J, Smith E, Delioukina M, Kogut N, Rosenthal J, Forman S, Nademanee A (2011) Reduced intensity allogeneic hematopoietic cell transplantation can induce durable remission in heavily pretreated relapsed Hodgkin lymphoma. Ann Hematol 90(7):803–808
Gayoso J, Balsalobre P, Pascual M, Castilla-Llorente C, López-Corral L, Kwon M, Serrano D, Piñana J, Herrera P, Ferrá C (2016) Busulfan-based reduced intensity conditioning regimens for haploidentical transplantation in relapsed/refractory Hodgkin lymphoma: Spanish multicenter experience. Bone Marrow Transplant 51(10):1307–1312
Raiola A, Dominietto A, Varaldo R, Ghiso A, Galaverna F, Bramanti S, Todisco E, Sarina B, Giordano L, Ibatici A (2014) Unmanipulated haploidentical BMT following non-myeloablative conditioning and post-transplantation CY for advanced Hodgkin’s lymphoma. Bone Marrow Transplant 49(2):190–194
Younes A, Santoro A, Shipp M, Zinzani PL, Timmerman JM, Ansell S, Armand P, Fanale M, Ratanatharathorn V, Kuruvilla J (2016) Nivolumab for classical Hodgkin's lymphoma after failure of both autologous stem-cell transplantation and brentuximab vedotin: a multicentre, multicohort, single-arm phase 2 trial. Lancet Oncol 17(9):1283–1294
Di Stasi A, De Angelis B, Rooney CM, Zhang L, Mahendravada A, Foster AE, Heslop HE, Brenner MK, Dotti G, Savoldo B (2009) T lymphocytes coexpressing CCR4 and a chimeric antigen receptor targeting CD30 have improved homing and antitumor activity in a Hodgkin tumor model. Blood 113(25):6392–6402
Wang C-M, Wu Z-Q, Wang Y, Guo Y-L, Dai H-R, Wang X-H, Li X, Zhang Y-J, Zhang W-Y, Chen M-X (2017) Autologous T cells expressing CD30 chimeric antigen receptors for relapsed or refractory Hodgkin lymphoma: an open-label phase I trial. Clin Cancer Res 23(5):1156–1166
Wang C-M, Wu Z-Q, Wang Y, Guo Y-L, Wang X-H, Li X, Dai H-R, Zhang Y-J, Zhang W-Y, Chen M-X (2015) Autologous T cells expressing CD30 chimeric antigen receptors for relapsed or refractory Hodgkin's lymphoma: an open-label phase 1 trial. Lancet 386:S12
Ramos CA, Ballard B, Zhang H, Dakhova O, Gee AP, Mei Z, Bilgi M, Wu MF, Liu H, Grilley B, Bollard CM, Chang BH, Rooney CM, Brenner MK, Heslop HE, Dotti G, Savoldo B (2017) Clinical and immunological responses after CD30-specific chimeric antigen receptor-redirected lymphocytes. J Clin Invest 127(9):3462–3471. https://doi.org/10.1172/jci94306
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Fig. 5
Overall and progression-free survival in subgroups 1. Kaplan-Meier curves regarding overall (OS) and progression-free (PFS) survival in subgroups divided by histology (nodular sclerosis or other, a-b), age (above or under 40 years, c-d) and the presence of extranodal disease at relapse (e-f). Abbreviations: OS overall survival, PFS progression-free survival, NS nodular sclerosis (PNG 990 kb).
Fig. 6
Overall and progression-free survival in subgroups 2. Kaplan-Meier curves regarding overall (OS) and progression-free (PFS) survival in subgroups divided by salvage chemotherapy regimen (a-b), response to salvage chemotherapy (c-d) and LDH level (e-f). Abbreviations: OS overall survival, PFS progression-free survival, LDH lactate dehydrogenase (PNG 868 kb).
Fig. 7
Prognostic score evaluation 2. Kaplan-Meier curves regarding progression-free (PFS) survival of all evaluable patients divided into subgroups according to previously published risk scores (Reece score a-b [23], Josting score c-d [16]). For each score, PFS is shown both for the observation period for which the score was evaluated (a,c) for and the complete follow-up period (b,d) (PNG 610 kb).
Rights and permissions
About this article
Cite this article
Roerden, M., Sökler, M., Kanz, L. et al. Outcome after autologous stem cell transplantation in primary refractory or relapsed Hodgkin lymphoma—a long-term follow-up single center experience. Ann Hematol 99, 265–276 (2020). https://doi.org/10.1007/s00277-019-03900-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-019-03900-x