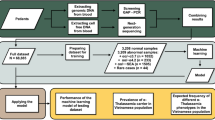
Abstract
The discovery of fetal DNA (f-DNA) opens the possibility of early non-invasive procedure for detection of paternally inherited mutation of beta-thalassemia. Since 2002, some studies have examined the sensitivity and specificity of this method for detection of paternally inherited mutation of thalassemia in pregnant women at risk of having affected babies. We conducted a systematic review of published articles that evaluated using this method for early detection of paternally inherited mutation in maternal plasma. A sensitive search of multiple databases was done in which nine studies met our inclusion criteria. The sensitivity and specificity was 99 and 99 %, respectively. The current study found that detection of paternally inherited mutation of thalassemia using analysis of cell-free fetal DNA is highly accurate. This method could replace conventional and invasive methods.




Similar content being viewed by others
References
Sodani P, Gaziev D, Polchi P et al (2004) New approach for bone marrow transplantation in patients with class 3 thalassemia aged younger than 17 years. Blood 104:1201–1203
Modell B, Khan M, Darlison M, Westwood M, Ingram D, Pennell D (2008) Improved survival of thalassaemia major in the UK and relation to T2* cardiovascular magnetic resonance. J Cardiovasc Magn Reson 10:42
Sadelain M (2006) Recent advances in globin gene transfer for the treatment of beta-thalassemia and sickle cell anemia. Curr Opin Hematol 13(3):142–148
Kosaryan M, Vahidshahi K, Siami R, Nazari M, Karami H, Ehteshami S (2009) Knowledge, attitude and practice of reproductive behavior in Iranian minor thalassemia couples. Saudi Med J 30(6):835–883
Psihogios V, Rodda C, Reid E, Clark M, Clarke C, Bowden D (2002) Reproductive health in individual with homozygous beta-thalassemia: knowledge, attitude, and behaviour. Fertil Steril 77(1):119–127
Modell B, Samavat A (2004) Iranian national thalassemia screening program. BMJ 329:1134–1137
Swanson A, Sehnert AJ, Bhatt S (2013) Non-invasive prenatal testing: technologies, clinical assays and implementation strategies for women’s healthcare practitioners. Curr Genet Med Rep 1(2):113–121
Papageorgiou EA, Patsalis PC (2012) Non-invasive prenatal diagnosis of aneuploidies: new technologies and clinical applications. Genome Med 4(5):46
Tan EM, Schur PH, Carr RI, Kunkel HG (1966) Deoxyribonucleic acid (DNA) and antibodies to DNA in the serum of patients with systemic lupus erythematosus. J Clin Invest 45:1732–1740
Butt AN, Shalchi Z, Hamaoui K et al (2006) Circulating nucleic acids and diabetic complications. Ann N Y Acad Sci 1075:1–9
Rainer TH, Wong LKS, Lam W et al (2003) Prognostic use of circulating plasma nucleic acid concentrations in patients with acute stroke. Clin Chem 49:562–569
Dennis LYM, Rainer Timothy H, Chan Lisa YS, Hjelm N, Cocks R (2000) Plasma DNA as a prognostic marker in trauma patients. Clin Chem 6:3319–3323
Margraf S, Logters T, Reipen J (2008) Neutrophil-derived circulating free DNA (cf-DNA/NETS): a potential prognostic marker for post traumatic development of inflammatory second hit and sepsis. Shock 30(4):352–358
Alp A, Us D, Hascelik G (2004) Comparison of manual and automated (MagNA Pure) nucleic acid isolation methods in molecular diagnosis of HIV infections. Mikrobiyol Bul 38:77–83
Hibi K, Robinson CR, Booker S (1998) Molecular detection of genetic alterations in the serum of colorectal cancer patients. Cancer Res 58:1405–1407
Swarup V, Rajeswari M (2007) Circulating (cell-free) nucleic acids—a promising, non-invasive tool for early detection of several human diseases. FEBS Lett 581(5):795–799
Lo Y, Corbetta N, Chamberlain PF, Rai V, Sargent L, Redman C (1997) Presence of fetal DNA in maternal plasma and serum. Lancet 350(9076):485–487
Hahn S, Rusterholz C, Hösli I, Lapaire O (2011) Cell-free nucleic acids as potential markers for preeclampsia. Placenta 32(1):s17–s20
Anthony R, Gregg SJ, Gross RG (2013) ACMG statement on noninvasive prenatal screening for fetal aneuploidy. Gene Med 15(5):395–398
Tynan JA, Angkachatchai V, Ehrich M (2011) Multiplexed analysis of circulating cell-free fetal nucleic acids for noninvasive prenatal diagnostic RHD testing. AJOG 204(3):251.e1–251.e6
Lo D, Zhang J, Leung T, Lau T, Chang A, Hjelm N (1999) Rapid clearance of fetal DNA from maternal plasma. Am J Hum Genet 64:218–224
Chiu RW, Cantor CR, Lo Y (2009) Non-invasive prenatal diagnosis by single molecule counting technologies. Trends Genet 25(7):324–331
Hill M, Barrett AN, White H, Chitty LS (2012) Uses of cell free fetal DNA in maternal circulation. Best Pract Res Cl Ob 26(5):639–654
Hahn S, Lapaire O, Tercanli S, Kolla V, Hösli I (2011) Determination of fetal chromosome aberrations from fetal DNA in maternal blood: has the challenge finally been met? Expert Rev Mol Med 13(1):e16
Wright CF, Burton H (2009) The use of cell-free fetal nucleic acids in maternal blood for non-invasive prenatal diagnosis. Hum Reprod Update 15(1):139–151
Papasavva T, van IJcken WFJ, Kockx CEM (2013) Next generation sequencing of SNPs for non-invasive prenatal diagnosis: challenges and feasibility as illustrated by an application to b-thalassemia. Eur J Hum Genet 21:1403–1410
Ehrich M, Deciu C, Zwiefelhofer T et al (2011) Noninvasive detection of fetal trisomy 21 by sequencing of DNA in maternal blood: a study in a clinical setting. Am J Obstet Gyneacology 204(3):205e1–205e11
Lam K-WG, Jiang P, Liao GJ et al (2012) Noninvasive prenatal diagnosis of monogenic diseases by targeted massively parallel sequencing of maternal plasma: application to β-thalassemia. Clin Chem 58(10):1467–1475
Khorram Khorshid HR, Zargari M, Sadeghi MR, Edalatkhah H, Shahhosseny M, Kamali K (2013) Early fetal gender determination using real-time PCR analysis of cell-free fetal DNA during 6th-10th weeks of gestation. Acta Medica Iranica 51(4):209–214
Cochrane Methods Working Group (1996) Working Group on Systematic Review of Screening and Diagnostic Tests. Recommended Methods :16–2
Wright CF, Wei Y, Higgins JP, Sagoo GS (2012) Non-invasive prenatal diagnostic test accuracy for fetal sex using cell-free DNA a review and meta-analysis. BMC Res Notes 5(1):476
Tong Y-K, Lo Y (2006) Diagnostic developments involving cell-free (circulating) nucleic acids. Clin Chim Acta 363(1):187–196
Colah R, Gorakshakar A, Nadkarni A (2011) Invasive & non-invasive approaches for prenatal diagnosis of haemoglobinopathies: experiences from India. Indian J Med Res 134(4):552
Zimmermann BG, Grill S, Holzgreve W, Zhong XY, Jackson LG, Hahn S (2008) Digital PCR: a powerful new tool for noninvasive prenatal diagnosis? Prenat Diagn 28(12):1087–1093
Babkina N, Graham JM Jr (2014) New genetic testing in prenatal diagnosis. Semin Fetal Neonatal Med 19(3):214–219
Alberry M, Soothill P, editors (2008) Non-invasive prenatal diagnosis: implications for antenatal diagnosis and management of high-risk pregnancies. Semin Fetal Neonatal Med 13(2): 84–90
Lucy Raymond F, Whittaker J, Jenkins L, Lench N, Chitty LS (2010) Molecular prenatal diagnosis: the impact of modern technologies. Prenat Diagn 30(7):674–681
Hahn S, Zhong XY, Holzgreve W (2008) Recent progress in non-invasive prenatal diagnosis. Semin Fetal Neonatal Med 13(2):57–62
Cousens NE, Gaff CL, Metcalfe SA, Delatycki MB (2010) Carrier screening for beta-thalassaemia: a review of international practice. Eur J Hum Genet 18(10):1077–1083
Chiu RW, Lau TK, Leung TN, Chow KC, Chui DH, Lo Y (2002) Prenatal exclusion of β thalassaemia major by examination of maternal plasma. Lancet 360(9338):998–1000
Ding C, Chiu RW, Lau TK et al (2004) MS analysis of single-nucleotide differences in circulating nucleic acids: application to noninvasive prenatal diagnosis. Proc Natl Acad Sci U S A 101(29):10762–10767
Li Y, Di Naro E, Vitucci A, Zimmermann B, Holzgreve W, Hahn S (2005) Detection of paternally inherited fetal point mutations for β-thalassemia using size-fractionated cell-free DNA in maternal plasma. JAMA 293(7):843–849
Papasavva T, Kalakoutis G, Kalikas I et al (2006) Noninvasive prenatal diagnostic assay for the detection of β‐thalassemia. Ann NY Acad Sci 1075(1):148–153
Tungwiwat W, Fucharoen G, Fucharoen S, Ratanasiri T, Sanchaisuriya K, Sae-Ung N (2007) Application of maternal plasma DNA analysis for noninvasive prenatal diagnosis of Hb E-β-thalassemia. Transl Res 150(5):319–325
Lazaros L, Hatzi E, Bouba I et al (2008) Non-invasive first-trimester detection of paternal beta-globin gene mutations and polymorphisms as predictors of thalassemia risk at chorionic villous sampling. Eur J Obstet Gynecol Reprod Biol 140(1):17–20
Papasavva T, Kalikas I, Kyrri A, Kleanthous M (2008) Arrayed primer extension for the noninvasive prenatal diagnosis of β‐thalassemia based on detection of single nucleotide polymorphisms. Ann NY Acad Sci 1137(1):302–308
Galbiati S, Foglieni B, Travi M et al (2008) Peptide-nucleic acid-mediated enriched polymerase chain reaction as a key point for non-invasive prenatal diagnosis of β-thalassemia. Haematologica 93(4):610–614
Chan K, Yam I, Leung K, Tang M, Chan T, Chan V (2010) Detection of paternal alleles in maternal plasma for non-invasive prenatal diagnosis of β-thalassemia: a feasibility study in southern Chinese. Eur J Obstet Gynecol Reprod Biol 150(1):28–33
Lo Y, Chan K, Sun H et al (2010) Maternal plasma DNA sequencing reveals the genome-wide genetic and mutational profile of the fetus. Sci Transl Med 2(61):61–91
Galbiati S, Brisci A, Damin F et al (2012) Fetal DNA in maternal plasma: a noninvasive tool for prenatal diagnosis of beta-thalassemia. Expert Opin Biol Th 12(S1):S181–S187
Guang-hua LI, Ka-bin R, Yan-fei L et al (2011) Prenatal diagnosis of β-thalassaemia using cell-free fetal DNA in maternal plasma. J South Med Univ 31(8):1437–1439
Phylipsen M, Yamsri S, Treffers EE et al (2012) Non‐invasive prenatal diagnosis of beta‐thalassemia and sickle‐cell disease using pyrophosphorolysis‐activated polymerization and melting curve analysis. Prenat Diagn 32(6):578–587
Prajantasen T, Fucharoen S, Fucharoen G, Siriratmanawong N, Pinmuang-ngam C (2013) Prenatal and post-natal screening of β-thalassemia and hemoglobin E genes in Thailand using denaturing high performance liquid chromatography. Mol Biol Rep 40(4):3173–3179
Papasavva TE, Lederer CW, Traeger‐Synodinos J et al (2013) A minimal set of SNPs for the noninvasive prenatal diagnosis of β‐thalassaemia. Ann Hum Genet 77(2):115–124
Dwamena B (2007) MIDAS: Stata module for meta-analytical integration of diagnostic test accuracy studies. Statistical Software Components. Boston College Department of Economics
Irwig L, Macaskill P, Glasziou P, Fahey M (1995) Meta-analytic methods for diagnostic test accuracy. J Clin Epidemiol 48(1):119–130
Tsui N, Kadir R, Chan A (2011) Noninvasive prenatal diagnosis of hemophilia by microfluidics digital PCR analysis of maternal plasma DNA. Blood 117(13):3684–3691
Gudran P, Mingshih L (2004) Principle and application of digital PCR. Expert Rev Mol Diagn 4(1):41–47
Acknowledgments
Many thanks for all persons who help us for the preparation this review paper. The funding of this research is supported by Mazandaran University of Medical Science.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by Mazandaran University of Medical Science.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zafari, M., Kosaryan, M., Gill, P. et al. Non-invasive prenatal diagnosis of β-thalassemia by detection of the cell-free fetal DNA in maternal circulation: a systematic review and meta-analysis. Ann Hematol 95, 1341–1350 (2016). https://doi.org/10.1007/s00277-016-2620-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-016-2620-3