Abstract
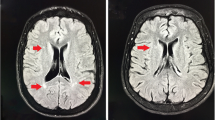
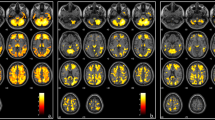
We aimed to determine the frequency of asymptomatic brain lesions in a group of patients with β-thalassemia intermedia (β-TI) and to evaluate correlation of asymptomatic brain lesions with splenectomy, thrombocytosis, blood transfusions, and clinical parameters. Ninety five neurologically intact patients with β-TI were randomly enrolled in this cross-sectional study. Diffusion-weighted imaging brain MRI was performed in every patient to detect cerebral white matter lesions (WML). We found an overall frequency of 15 (15.8 %) for WMLs, 14 (23.7 %) in splenectomized, and 1 (2.8 %) in nonsplenectomized patients. The presence of WML was significantly associated with splenectomy (P = 0.008) and thrombocytosis (P = 0.009). However, after adjustment for splenectomy, thrombocytosis was not significantly associated with the presence of WML (P > 0.05). The number of patients with regular blood transfusions and normal MRI was not significantly higher compared to those with abnormal findings (52.5 % vs. 26.7 %; P = 0.092). In untransfused patients, hydroxyurea (HU) administration was associated with a lower incidence of WML (P < 0.001). Although in univariate analysis either splenectomy or thrombocytosis showed significant correlation with the presence of single or multiple WMLs, thrombocytosis by itself did not significantly contribute in developing asymptomatic brain lesions. The lack of significant correlation between lesions and regular blood transfusions could be related to the treatment with HU in untransfused patients, which increased fetal hemoglobin levels and improved the morphology and the pathological indices of the red blood cells. Larger prospective studies are suggested for the accurate evaluation of the correlation of these factors with developing asymptomatic brain lesions.

Similar content being viewed by others
References
Eldor A, Rachmilewitz EA (2002) The hypercoagulable state in thalassemia. Blood 99:36–43
Karimi M, Khanlari M, Rachmilewitz EA (2008) Cerebrovascular accident in beta-thalassemia major (beta-TM) and beta-thalassemia intermedia (beta-TI). Am J Hematol 83:77–79
Karimi M, Haghpanah S, Farhadi A et al (2012) Genotype-phenotype relationship of patients with beta-thalassemia taking hydroxyurea: a 13-year experience in Iran. Int J Hematol 95:51–56
Cappellini MD, Robbiolo L, Bottasso BM et al (2000) Venous thromboembolism and hypercoagulability in splenectomized patients with thalassaemia intermedia. Br J Haematol 111:467–473
Eldor A, Maclouf J, Lellouche F et al (1993) A chronic hypercoagulable state and life-long platelet activation in beta thalassemia major. Southeast Asian J Trop Med Public Health 24(Suppl 1):92–95
Goldschmidt N, Spectre G, Brill A et al (2008) Increased platelet adhesion under flow conditions is induced by both thalassemic platelets and red blood cells. Thromb Haemost 100:864–870
Moratelli S, De Sanctis V, Gemmati D et al (1998) Thrombotic risk in thalassemic patients. J Pediatr Endocrinol Metab 11(Suppl 3):915–921
Naithani R, Chandra J, Narayan S et al (2006) Thalassemia major—on the verge of bleeding or thrombosis? Hematology 11:57–61
Shirahata A, Funahara Y, Opartkiattikul N et al (1992) Protein C and protein S deficiency in thalassemic patients. Southeast Asian J Trop Med Public Health 23(Suppl 2):65–73
Visudhiphan S, Ketsa-Ard K, Tumliang S et al (1994) Significance of blood coagulation and platelet profiles in relation to pulmonary thrombosis in beta-thalassemia/Hb E. Southeast Asian J Trop Med Public Health 25:449–456
Karami H, Vahidshahi K, Kosarian M et al. Assessment of coagulation state and its related factors in thalassemia intermedia patients referred to thalassemia research center at Booali Sina Hospital Sari/IR Iran in 2007. Pak J Biol Sci 13
Musumeci S, Leonardi S, Di Dio R et al (1987) Protein C and antithrombin III in polytransfused thalassemic patients. Acta Haematol 77:30–33
Borenstain-Ben Yashar V, Barenholz Y, Hy-Am E et al (1993) Phosphatidylserine in the outer leaflet of red blood cells from beta-thalassemia patients may explain the chronic hypercoagulable state and thrombotic episodes. Am J Hematol 44:63–65
Shenkman B (2008) Thrombotic complications in thalassemic patients: contribution of red blood cells and platelets. Thromb Haemost 100:735
Taher A, Isma’eel H, Mehio G et al (2006) Prevalence of thromboembolic events among 8,860 patients with thalassaemia major and intermedia in the Mediterranean area and Iran. Thromb Haemost 96:488–491
Karimi M, Bagheri H, Rastgu F et al (2010) Magnetic resonance imaging to determine the incidence of brain ischaemia in patients with beta-thalassaemia intermedia. Thromb Haemost 103:989–993
Manfre L, Giarratano E, Maggio A et al (1999) MR imaging of the brain: findings in asymptomatic patients with thalassemia intermedia and sickle cell-thalassemia disease. AJR Am J Roentgenol 173:1477–1480
Metarugcheep P, Chanyawattiwongs S, Srisubat K et al (2008) Clinical silent cerebral infarct (SCI) in patients with thalassemia diseases assessed by magnetic resonance imaging (MRI). J Med Assoc Thail 91:889–894
Taher AT, Musallam KM, Nasreddine W et al (2010) Asymptomatic brain magnetic resonance imaging abnormalities in splenectomized adults with thalassemia intermedia. J Thromb Haemost 8:54–59
Mikulis DJ, Roberts TP (2007) Neuro MR: protocols. J Magn Reson Imaging 26:838–847
Cappellini MD, Grespi E, Cassinerio E et al (2005) Coagulation and splenectomy: an overview. Ann N Y Acad Sci 1054:317–324
Adams RJ (2000) Lessons from the stroke prevention trial in sickle cell anemia (STOP) study. J Child Neurol 15:344
Adams RJ, McKie VC, Hsu L et al (1998) Prevention of a first stroke by transfusions in children with sickle cell anemia and abnormal results on transcranial Doppler ultrasonography. N Engl J Med 339:5–11
Ware RE, Zimmerman SA, Sylvestre PB et al (2004) Prevention of secondary stroke and resolution of transfusional iron overload in children with sickle cell anemia using hydroxyurea and phlebotomy. J Pediatr 145:346–352
Gulbis B, Haberman D, Dufour D et al (2005) Hydroxyurea for sickle cell disease in children and for prevention of cerebrovascular events: the Belgian experience. Blood 105:2685–2690
Karimi M, Darzi H, Yavarian M (2005) Hematologic and clinical responses of thalassemia intermedia patients to hydroxyurea during 6 years of therapy in Iran. J Pediatr Hematol Oncol 27:380–385
Johnson C, Telen MJ (2008) Adhesion molecules and hydroxyurea in the pathophysiology of sickle cell disease. Haematologica 93:481–485
AA D, Ghebremeskel K, MI E et al (2011) Hydroxyurea therapy mobilises arachidonic acid from inner cell membrane aminophospholipids in patients with homozygous sickle cell disease. J Lipids 2011
Brun M, Bourdoulous S, Couraud P et al (2003) Hydroxyurea downregulates endothelin-1 gene expression and upregulates ICAM-1 gene expression in cultured human endothelial cells. Pharmacogenomics J 3:215–226
Enzinger C, Smith S, Fazekas F et al (2006) Lesion probability maps of white matter hyperintensities in elderly individuals: results of the Austrian stroke prevention study. J Neurol 253:1064–1070
Longstreth WT Jr, Manolio TA, Arnold A et al (1996) Clinical correlates of white matter findings on cranial magnetic resonance imaging of 3301 elderly people. The Cardiovascular Health Study. Stroke 27:1274–1282
Acknowledgments
We would like to thank Shiraz University of Medical sciences for financial and approval support and MRI technicians of Faghihi Hospital Shiraz, Iran.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karimi, M., Haghpanah, S., Bagheri, M.H. et al. Frequency and distribution of asymptomatic brain lesions in patients with β-thalassemia intermedia. Ann Hematol 91, 1833–1838 (2012). https://doi.org/10.1007/s00277-012-1527-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-012-1527-x