Abstract
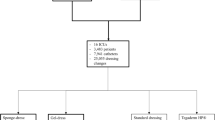
The objective of the study was to evaluate the effectiveness of chlorhexidine-impregnated sponges for reducing catheter-related infections of central venous catheters inserted for cancer chemotherapy. The method used was a randomized, prospective, open, controlled clinical study (three-step group sequential analysis protocol). The patients were from two high dependency units at a university hospital undergoing chemotherapy for haematological or oncological malignancies requiring central venous catheters (CVCs) expected to remain in place for at least 5 days. Six hundred and one patients with 9,731 catheterization days were studied between January 2004 and January 2006. Patients admitted for chemotherapy received chlorhexidine and silver sulfadiazine-impregnated triple-lumen CVCs under standardized conditions and were randomized to the groups receiving a chlorhexidine gluconate-impregnated wound dressing or a standard sterile dressing. Daily routine included clinical assessment of the insertion site (swelling, pain, redness), temperature, white blood count and C-reactive protein. Catheters remained in place until they were no longer needed or when a CVC-related infection was suspected. Infection was confirmed with blood cultures via the catheter lumina and peripheral blood cultures according to the time-to-positivity method. Six hundred and one patients were included. The groups were comparable with respect to demographic and clinical data. The incidence of CVC-related infections were 11.3% (34 of 301) and 6.3% (19 of 300) in the control and chlorhexidine-impregnated wound dressing groups, respectively (p = 0.016, relative risk 0.54; confidence interval 0.31–0.94). Especially, catheter-related infections at internal jugular vein insertions could be reduced (p = 0.018). No adverse effects related to the intervention were observed. The use of chlorhexidine-impregnated wound dressings significantly reduced the incidence of CVC-related infections in patients receiving chemotherapy.
Similar content being viewed by others
References
Dettenkofer M, Wenzler-Rottele S, Babikir R et al (2005) Surveillance of nosocomial sepsis and pneumonia in patients with a bone marrow or peripheral blood stem cell transplant: a multicenter project. Clin Infect Dis 40(7):926–931
Hanna H, Afif M, Alakech B et al (2004) Central venous catheter-related bacteremia due to gram-negative bacilli: significance of catheter removal in preventing relapse. Infect Control Hosp Epidemiol 25(8):646–469
Jaeger K, Zenz S, Jüttner B et al (2005) Reduction of catheter-related infections in neutropenic patients: a prospective controlled randomized trial using a chlorhexidine and silver sulfadiazine-impregnated central venous catheter. Ann Hematol 84(4):258–262
Raad I, Hanna HA, Alakech B et al (2004) Differential time to positivity: a useful method for diagnosing catheter-related bloodstream infections. Ann Intern Med 140(1):18–25
Maki DG, Ringer M, Alvarado CJ (1991) Prospective randomised trial of povidone–iodine, alcohol, and chlorhexidine for prevention of infection associated with central venous and arterial catheters. Lancet 338(8763):339–343
Garland JS, Alex CP, Mueller CD et al (2001) A randomized trial comparing povidone-iodine to a chlorhexidine gluconate-impregnated dressing for prevention of central venous catheter infection in neonates. Pediatrics 107(6):1431–1416
Levy I, Katz J, Solter E et al (2005) Chlorhexidine-impregnated dressing for prevention of colonization of central-venous catheters in infants and children: a randomized controlled study. Pediatr Inf Dis 24(8):676–679
Roberts B, Cheung D (1998) Biopatch—a new concept in antimicrobial dressings for invasive devices. Aust Crit Care 11(1):16–19
O’Grady NP, Alexander M, Dellinger EP , Gerberding JL, Heard SO, Maki DG, Masur H, McCormick RD, Mermel LA, Pearson ML, Raad I, Randolph A (2002) Guidelines fort he prevention of intravascular catheter-related infections. Morb Mortal Wkly Rep Recomm Rep. 52(RR-10):1–29
Maki DG, Weise CE, Sarafin HW (1977) A semiquantitative method for identifying intravenous-catheter-related infections. N Engl J Med 296(23):1305–1309
Blot F, Schmidt E, Nitenberg G et al (1998) Earlier positivity of central-venous- versus peripheral-blood cultures is highly predictive of catheter-related sepsis. J Clin Microbiol 36(1):105–109
Pronovost P, Needham D, Berenholtz S et al (2006) An intervention to decrease catheter-related bloodstream infections in the ICU. N Engl J Med 355(26):2725–2732
Crnich CJ, Maki DG (2002) The promise of novel technology for the prevention of intravascular device-related bloodstream infection. I. Pathogenesis and short-term devices. Clin Infect Dis 34:1232–1242
Hanazaki K, Shingu K, Adachi W et al (1999) Chlorhexidine dressing for reduction in microbial colonization of the skin with central venous catheters: a prospective randomized controlled trial. J Hosp Infect 42:165–168
Maki DG, Mermel LA, Kluger D et al (2000) The efficacy of a chlorhexidine-impregnated sponge (Biopatch) for the prevention of intravascular catheter-related infection: s prospective randomized, controlled multicenter study (abstract). In: Proceedings of the 40th Interscience Conference on Antimicrobial Agents and Chemotherapy. Washington DC, Sept. 2000, Abstract 1430, p 422
Chambers ST, Sanders J, Patton WN et al (2005) Reduction of exit-site infections in tunnelled intravascular catheters among neutropenic patients by sustained-release chlorhexidine dressings: results from a prospective randomized controlled trial. J Hosp Infect 61:53–61
Ho K, Litton E (2006) Use of chlorhexidine-impregnated dressing to prevent vascular and epidural catheter colonization and infection: a meta-analysis. J Antimicrob Chemother 58:281–287
Warren DK, Quadir WW, Hollenbaek CS et al (2006) Attributable cost of catheter-associated bloodstream infections among intensive care patients in a nonteaching hospital. Crit Care Med 34(8):2084–2089
Rüsch S, Walder B, Tramèr MR (2002) Complications of central venous catheters: internal jugular versus subclavian access—a systematic review. Crit Care Med 30(2):454–460
Kluger M (2003) Anaphylaxis to chlorhexidine-impregnated central venous catheter. Anaesth Intensive Care 31(6):697–698
Jayathillake A, Mason DF, Broome K (2003) Allergy to chlorhexidine gluconate in urethral gel: report of four cases and review of the literature. Urology 61(4):837
Tattawasart U, Maillard JY, Furr JR et al (1999) Development of resistance to chlorhexidine diacetate and cetylpyridinium chloride in Pseudomonas stutzeri and changes in antibiotic susceptibility. J Hosp Infect 42(3):219–229
Acknowledgements
The authors are especially indebted to the nurses at the Department of Haematology, Haemostasis, Oncology, and Stem Cell Transplantation at Hannover Medical School. The authors thank Christian Apfel M.D. for critical review of the manuscript.
Funding and conflict of interest statement
The study was carried out without any external funding. Part of the results of this study was presented as an abstract at the Annual Meeting of the European Society of Anaesthesiologists (ESA) in Madrid in June 2006. H.R. and M.F. received travel grants by Ethicon/Johnson & Johnson.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ruschulte, H., Franke, M., Gastmeier, P. et al. Prevention of central venous catheter related infections with chlorhexidine gluconate impregnated wound dressings: a randomized controlled trial. Ann Hematol 88, 267–272 (2009). https://doi.org/10.1007/s00277-008-0568-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-008-0568-7