Abstract
Purpose
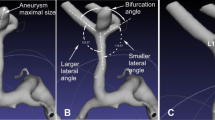
The Cerebral arterial circle presents multiple individual anatomical configurations which are of the highest importance regarding the pathological processes for intracranial aneurysms development. Previous studies highlighted the importance of geometry and especially arterial bifurcations leading to aneurysms development. The primary objective of this study was to determine whether a flow pattern asymmetry of the P1 segments of the posterior cerebral arteries was associated with a higher risk of basilar tip aneurysm.
Material and methods
Two different populations were retrospectively reviewed. The first population, without aneurysm, for which TOF MRI sequences were reviewed. The second population with patients harboring basilar tip aneurysms for whom cerebral angiograms were reviewed. We retrospectively analyzed the flow contribution and symmetry of the two right and left P1 segments of the posterior cerebral arteries and the two posterior communicating arteries (Pcomm). We analyzed the association and risk factors for basilar tip aneurysm.
Results
The anatomical and flow configurations of P1 and Pcomm have been reviewed in 467 patients without aneurysms and 35 patients with aneurysms. We identified a significant association between the flow pattern asymmetry of the P1 segments and the presence of a basilar tip aneurysm (OR = 2.12; IC95% = [1.01–4.36]; p = 0.04). We also confirmed that the male gender was protective against aneurysm (OR = 0.45; IC95% = [0.194–0.961]; p = 0.04).
Conclusion
Non-modal basilar tip bifurcation and flow asymmetry of P1 segments are associated with an increased risk of basilar tip aneurysm. These findings highlight the importance of analyzing MRI-TOF of the posterior configuration of the Cerebral arterial circle to potentially refine the aneurysms risk prediction.



Similar content being viewed by others

Availability of data and materials
Datasets can be available upon request to the corresponding author.
References
Alpers BJ, Berry RG, Paddison RM (1959) Anatomical studies of the circle of Willis in normal brain. AMA Arch Neurol Psychiatry 81:409–418. https://doi.org/10.1001/archneurpsyc.1959.02340160007002
Baptista AG (1964) Studies on the arteries of the brain. 3. Circle of Willis: morphologic features. Acta Neurol Scand 40:398–414. https://doi.org/10.1111/j.1600-0404.1964.tb02765.x
Can A, Mouminah A, Ho AL, Du R (2015) Effect of vascular anatomy on the formation of basilar tip aneurysms. Neurosurgery 76:62–66. https://doi.org/10.1227/NEU.0000000000000564
Claux F, Baudouin M, Bogey C, Rouchaud A (2022) Dense, deep learning-based intracranial aneurysm detection on TOF MRI using two-stage regularized U-Net. J Neuroradiol J Neuroradiol S0150–9861(22):00101–00108. https://doi.org/10.1016/j.neurad.2022.03.005
Fredon F, Baudouin M, Hardy J et al (2021) An MRI study of typical anatomical variants of the anterior communicating artery complex. Surg Radiol Anat SRA 43:1983–1988. https://doi.org/10.1007/s00276-021-02782-x
Frösen J, Cebral J, Robertson AM, Aoki T (2019) Flow-induced, inflammation-mediated arterial wall remodeling in the formation and progression of intracranial aneurysms. Neurosurg Focus 47:E21. https://doi.org/10.3171/2019.5.FOCUS19234
Guerin J, Gouaze A, Lazorthes G (1976) Le polygone de Willis de l’enfant et les facteurs de son modelage. Neurochirurgie 22:217–226
Kirgis HD, Llewellyn RC, Peebles EM (1960) Functional trifurcation of the internal carotid artery and its potential clinical significance. J Neurosurg 17:1062–1072. https://doi.org/10.3171/jns.1960.17.6.1062
Lauric A, Hippelheuser JE, Malek AM (2019) Induction of aneurysmogenic high positive wall shear stress gradient by wide angle at cerebral bifurcations, independent of flow rate. J Neurosurg 131:442–452. https://doi.org/10.3171/2018.3.JNS173128
Li L, Hofmann BB, Fischer I et al (2021) Asymmetry of P1 and vertebral arteries is not related to basilar tip aneurysm development or rupture. Acta Neurochir (Wien) 163:805–812. https://doi.org/10.1007/s00701-020-04593-2
Meng H, Tutino VM, Xiang J, Siddiqui A (2014) High WSS or Low WSS? Complex interactions of hemodynamics with intracranial aneurysm initiation, growth, and rupture: toward a unifying hypothesis. Am J Neuroradiol 35:1254–1262. https://doi.org/10.3174/ajnr.A3558
Moffat DB (1961) The development of the posterior cerebral artery. J Anat 95:485–494
Padget D (1948) The development of the cranial arteries in the human embryo. Contrib Embryol 32:205–262
Padget DH (1945) The Circle of Willis, its embryology and anatomy. DANDY WE 67–90
Puchades-Orts A, Nombela-Gomez M, Ortuño-Pacheco G (1976) Variation in form of circle of Willis: some anatomical and embryological considerations. Anat Rec 185:119–123. https://doi.org/10.1002/ar.1091850112
Riggs HE, Rupp C (1963) Variation in form of circle of Willis. The relation of the variations to collateral circulation: anatomic analysis. Arch Neurol 8:8–14. https://doi.org/10.1001/archneur.1963.00460010024002
Rinkel GJE, Djibuti M, Algra A, van Gijn J (1998) Prevalence and risk of rupture of intracranial aneurysms: a systematic review. Stroke 29:251–256. https://doi.org/10.1161/01.STR.29.1.251
Staarmann B, Smith M, Prestigiacomo CJ (2019) Shear stress and aneurysms: a review. Neurosurg Focus 47:E2. https://doi.org/10.3171/2019.4.FOCUS19225
Stehbens WE (1963) Aneurysms and anatomical variation of cerebral arteries. Arch Pathol 75:45–64
Tütüncü F, Schimansky S, Baharoglu MI et al (2014) Widening of the basilar bifurcation angle: association with presence of intracranial aneurysm, age, and female sex: clinical article. J Neurosurg 121:1401–1410. https://doi.org/10.3171/2014.8.JNS1447
Windle BC (1888) The arteries forming the circle of Willis. J Anat Physiol 22:289–293
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Contributions
PL: contributed to conception and design of the study, contributed to data collection and analysis, drafting the manuscript, contributed to revising the manuscript for important intellectual content. QP: contributed to conception and design of the study, contributed to data collection and analysis, drafting the manuscript, contributed to revising the manuscript for important intellectual content. RC: contributed to data collection and analysis, drafting the manuscript. JC: contributed to data collection and analysis, drafting the manuscript. AR: contributed to conception and design of the study, contributed to data collection and analysis, drafting the manuscript, contributed to revising the manuscript for important intellectual content. All authors approved the final version to be published.
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethical approval
As for all non-interventional retrospective studies of de-identified data in France, written informed consent was waived, but the patients or their proxies were informed that they could oppose the use of their de-identified health-related data for the purpose of research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lapraz, P., Pinsard, Q., Coudert, R. et al. Association between flow patterns of the posterior cerebral arterial circle and basilar-tip aneurysms. Surg Radiol Anat 45, 505–511 (2023). https://doi.org/10.1007/s00276-023-03121-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-023-03121-y