Abstract
Purpose
The aim of this study was to analyze variations in the morphological features of the subparietal sulcus (SPS) and to investigate interhemispheric and gender differences in these variations using multiplanar reconstructed (MPR) magnetic resonance (MR) images.
Methods
Two hundred subjects with normal cranial MR imaging, including high-resolution T1-weighted volumetric data, were enrolled in the study. The sagittal or oblique sagittal MPR images created from high-resolution T1-weighted data were analyzed for the following morphological features: the SPS patterns, the continuity of the SPS with the cingulate sulcus and parieto-occipital sulcus (POS), and the presence of upwardly directed SPS branches reaching to the hemispheric surface. Interindividual variability of the morphologic features, hemispheric asymmetry, and gender differences were investigated.
Results
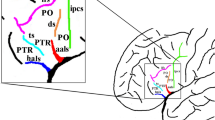
Considerable variations were found in the morphological features of the SPS. The H-pattern, no connection with the cingulate sulcus or the POS, and one upwardly directed branch reaching the hemispheric surface were most commonly observed morphologic features of the SPS in 45.2, 41.8, and 48 % of the all hemispheres, respectively. Furthermore, the connection of the SPS only with the cingulate sulcus and the presence of two upwardly directed branches reaching the hemispheric surface showed the significant leftward asymmetry (P < 0.05).
Conclusions
Our study demonstrated the extensive morphological variability of the SPS and the hemispheric asymmetry for some morphological features. Knowledge of these variations and their hemispheric asymmetry may be helpful for surgical approaches in neurosurgery and structure–function correlations in functional neuroimaging studies involving the posteromedial hemisphere.



Similar content being viewed by others
References
Barkovich AJ, Rowley HA, Andermann F (1995) MR in partial epilepsy: value of high-resolution volumetric techniques. AJNR Am J Neuroradiol 16:339–343
Cauda F, Geminiani G, D’Agata F, Sacco K, Duca S, Bagshaw AP, Cavanna AE (2010) Functional connectivity of the posteromedial cortex. PLoS One 5(9):e13107. doi:10.1371/journal.pone.0013107
Cavanna AE, Trimble MR (2006) The precuneus: a review of its functional anatomy and behavioural correlates. Brain 129:564–583
Eckert MA, Galaburda AM, Karchemskiy A, Liang A, Thompson P, Dutton RA, Lee AD, Bellugi U, Korenberg JR, Mills D, Rose FE, Reiss AL (2006) Anomalous sylvian fissure morphology in Williams syndrome. Neuroimage 33:39–45
Fornito A, Yücel M, Wood S, Stuart GW, Buchanan JA, Proffitt T, Anderson V, Velakoulis D, Pantelis C (2004) Individual differences in anterior cingulate/paracingulate morphology are related to executive functions in healthy males. Cereb Cortex 14:424–431
Fornito A, Yücel M, Wood SJ, Proffitt T, McGorry PD, Velakoulis D, Pantelis C (2006) Morphology of the paracingulate sulcus and executive cognition in schizophrenia. Schizophr Res 88:192–197
Fornito A, Whittle S, Wood SJ, Velakoulis D, Pantelis C, Yücel M (2006) The influence of sulcal variability on morphometry of the human anterior cingulate and paracingulate cortex. Neuroimage 33:843–854
Fransson P, Marrelec G (2008) The precuneus/posterior cingulate cortex plays a pivotal role in the default mode network: evidence from a partial correlation network analysis. Neuroimage 42:1178–1184
Galaburda AM, Rosen GD, Sherman GF (1990) Individual variability in cortical organization: its relationship to brain laterality and implications to function. Neuropsychologia 28:529–546
Gonul Y, Songur A, Uzun I, Uygur R, Alkoc OA, Caglar V, Kucuker H (2014) Morphometry, asymmetry and variations of cerebral sulci on superolateral surface of cerebrum in autopsy cases. Surg Radiol Anat 36:651–661
Good CD, Johnsrude IS, Ashburner J, Henson RN, Friston KJ, Frackowiak RS (2001) A voxel-based morphometric study of ageing in 465 normal adult human brains. Neuroimage 14:21–36
Gürer B, Bozkurt M, Neves G, Cikla U, Hananya T, Antar V, Salamat S, Başkaya MK (2013) The subparietal and parietooccipital sulci: an anatomical study. Clin Anat 26:667–674
Hamasaki T, Imamura J, Kawai H, Kuratsu J (2012) A three-dimensional MRI study of variations in central sulcus location in 40 normal subjects. J Clin Neurosci 19:115–120
Hasnain MK, Fox PT, Woldorff MG (2006) Hemispheric asymmetry of sulcus-function correspondence: quantization and developmental implications. Hum Brain Mapp 27:277–287
Keller SS, Highley JR, Garcia-Finana M, Sluming V, Rezaie R, Roberts N (2007) Sulcal variability, stereological measurement and asymmetry of Broca’s area on MR images. J Anat 211:534–555
Margulies DS, Vincent JL, Kelly C, Lohmann G, Uddin LQ, Biswal BB, Villringer A, Castellanos FX, Milham MP, Petrides M (2009) Precuneus shares intrinsic functional architecture in humans and monkeys. Proc Natl Acad Sci USA 106:20069–20074
Montenegro MA, Li LM, Guerreiro MM, Guerreiro CA, Cendes F (2002) Focal cortical dysplasia: improving diagnosis and localization with magnetic resonance imaging multiplanar and curvilinear reconstruction. J Neuroimaging 12:224–230
Ochiai T, Grimault S, Scavarda D, Roch G, Hori T, Rivière D, Mangin JF, Régis J (2004) Sulcal pattern and morphology of the superior temporal sulcus. Neuroimage 22:706–719
Ono M, Kubik S, Abernathey CD (1990) Atlas of cerebral sulci. Thieme, Stuttgart
Paus T, Otaky N, Caramanos Z, MacDonald D, Zijdenbos A, D’Avirro D, Gutmans D, Holmes C, Tomaiuolo F, Evans AC (1996) In vivo morphometry of the intrasulcal gray matter in the human cingulate, paracingulate, and superior-rostral sulci: hemispheric asymmetries, gender differences and probability maps. J Comp Neurol 376:664–673
Pruessner JC, Köhler S, Crane J, Pruessner M, Lord C, Byrne A, Kabani N, Collins DL, Evans AC (2002) Volumetry of temporopolar, perirhinal, entorhinal and parahippocampal cortex from high-resolution MR images: considering the variability of the collateral sulcus. Cereb Cortex 12:1342–1353
Reckess GZ, Dunn CB, Bauer RM, Leonard CM (2013) Anterior temporobasal sulcal morphology: development of a reliable rating protocol and normative data. Brain Struct Funct 218:889–901
Rhoton AL Jr (2002) The cerebrum. Neurosurgery 51(4 Suppl):S1–S51
Ribas GC, Yasuda A, Ribas EC, Nishikuni K, Rodrigues AJ Jr (2006) Surgical anatomy of micro neurosurgical sulcal key points. Neurosurgery 59:177–211
Tu Z, Zheng S, Yuille AL, Reiss AL, Dutton RA, Lee AD, Galaburda AM, Dinov I, Thompson PM, Toga AW (2007) Automated extraction of the cortical sulci based on a supervised learning approach. IEEE Trans Med Imaging 26:541–552
van der Kouwe AJ, Benner T, Salat DH, Fischl B (2008) Brain morphometry with multiecho MPRAGE. Neuroimage 40:559–569
Winterburn JL, Pruessner JC, Chavez S, Schira MM, Lobaugh NJ, Voineskos AN, Chakravarty MM (2013) A novel in vivo atlas of human hippocampal subfields using high-resolution 3 T magnetic resonance imaging. Neuroimage 74:254–265
Yasargil MG (1996) Intraventricular tumors. Microneurosurgery, vol IV B. Georg Thieme Verlag, Stuttgart, pp 320–323
Yaşargil MG, Abdulrauf SI (2008) Surgery of intraventricular tumors. Neurosurgery 62(6 Suppl):S1029–S1040
Zhang S, Li CS (2012) Functional connectivity mapping of the human precuneus by resting state fMRI. Neuroimage 59:3548–3562
Zhang Y, Fan L, Zhang Y, Wang J, Zhu M, Zhang Y, Yu C, Jiang T (2014) Connectivity-based parcellation of the human posteromedial cortex. Cereb Cortex 24:719–727
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
The study was approved by the institutional ethics committee.
Rights and permissions
About this article
Cite this article
Kacar, E., Nas, O.F., Okeer, E. et al. Pattern, variability, and hemispheric differences of the subparietal sulcus on multiplanar reconstructed MR images. Surg Radiol Anat 38, 89–96 (2016). https://doi.org/10.1007/s00276-015-1525-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-015-1525-5