Abstract
Purpose
To evaluate the relationship between non-tumor liver (NTL) dose and adverse events (AE) in patients with hepatocellular carcinoma (HCC) treated with glass-based Yttrium-90 radioembolization (Y90-RE).
Materials and Methods
A retrospective analysis of patients with HCC treated with Y90-RE between 2013 and 2018 was performed. Baseline characteristics including demographics and Y90-RE treatment approach were captured. Common Terminology Criteria for Adverse Events v5 was assessed at months 3 and 6 post-treatment. Using voxel-based dosimetry with MIM Software V. 6.9, dose-volume histograms of treated area of liver were created. Receiver operator characteristic curve was used to determine NTL dose threshold predicting AEs. Multivariate analysis was used to determine independent clinical factors of predicting severe AEs. Chi-square analysis was used to compare proportions.
Results
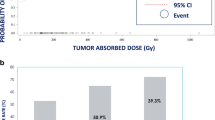
Two hundred and twenty-nine consecutive patients (115(50.2%) lobar and 114(49.8%) segmental) were included. At 3 months, there was a lower rate of any grade AE (55(46%) segmental and 36(31%) lobar, p = 0.009) and increased rate of severe AEs for lobar compared to segmental (2(2%) segmental and 9(8%) lobar, p = 0.029). At 6 months, severe AEs were greater for lobar than segmental (1(1%) segmental vs 10(9%) lobar, p = 0.005). For lobar Y90-RE, mean NTL dose of 112 Gy predicted severe AE (89% sensitivity and 91% specificity (AUC = 0.95, p = < 0.0001) at 3 and 6 months. For the segmental group, no significant association was found between NTL dose and severe treatment-related AE at 3 and 6 months.
Conclusion
In patients with HCC undergoing glass-based lobar Y90-RE, NTL dose of > 112 Gy is associated with severe treatment-related AEs at 3–6 months.


Similar content being viewed by others
References
Garin E, et al. Personalised versus standard dosimetry approach of selective internal radiation therapy in patients with locally advanced hepatocellular carcinoma (DOSISPHERE-01): a randomised, multicentre, open-label phase 2 trial. Lancet Gastroenterol Hepatol. 2021;6(1):17–29.
Shah RM, et al. Prognostic factors of unresectable hepatocellular carcinoma treated with yttrium-90 radioembolization: results from a large cohort over 13 years at a single center. J Gastrointest Oncol. 2021;12(4):1718–31.
Moreno-Luna LE, et al. Efficacy and safety of transarterial radioembolization versus chemoembolization in patients with hepatocellular carcinoma. Cardiovasc Intervent Radiol. 2013;36(3):714–23.
Lance C, et al. Comparative analysis of the safety and efficacy of transcatheter arterial chemoembolization and yttrium-90 radioembolization in patients with unresectable hepatocellular carcinoma. J Vasc Interv Radiol. 2011;22(12):1697–705.
Hilgard P, et al. Radioembolization with yttrium-90 glass microspheres in hepatocellular carcinoma: European experience on safety and long-term survival. Hepatology. 2010;52(5):1741–9.
Salem R, et al. Radioembolization results in longer time-to-progression and reduced toxicity compared with chemoembolization in patients with hepatocellular carcinoma. Gastroenterology. 2011;140(2):497-507.e2.
El Fouly A, et al. In intermediate stage hepatocellular carcinoma: radioembolization with yttrium 90 or chemoembolization? Liver Int. 2015;35(2):627–35.
Gabr A, et al. Correlation of Y90-absorbed radiation dose to pathological necrosis in hepatocellular carcinoma: confirmatory multicenter analysis in 45 explants. Eur J Nucl Med Mol Imaging. 2021;48(2):580–3.
Levillain H, et al. International recommendations for personalised selective internal radiation therapy of primary and metastatic liver diseases with yttrium-90 resin microspheres. Eur J Nucl Med Mol Imaging. 2021;48(5):1570–84.
Camacho JC, et al. (90)Y Radioembolization: multimodality imaging pattern approach with angiographic correlation for optimized target therapy delivery. Radiographics. 2015;35(5):1602–18.
Package Insert: TheraSphere Yttrium-90 Glass Microspheres. [cited 6 February 2022]; https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&ved=2ahUKEwi_h-qxxuz1AhVkjYkEHSDnBocQFnoECAMQAQ&url=https%3A%2F%2Fwww.accessdata.fda.gov%2Fcdrh_docs%2Fpdf%2Fh980006c.pdf&usg=AOvVaw3DTs9oqnqZ43-boZ5FwCzu.
Villalobos A, et al. Tumor-to-normal ratio relationship between planning technetium-99 macroaggregated albumin and posttherapy Yttrium-90 Bremsstrahlung SPECT/CT. J Vasc Interv Radiol. 2021;32(5):752–60.
Bolch WE, et al. MIRD pamphlet No 17: the dosimetry of nonuniform activity distributions–radionuclide S values at the voxel level medical internal radiation dose committee. J Nucl Med. 1999;40(1):11s–36s.
Bolch WE, et al. MIRD pamphlet No 21: a generalized schema for radiopharmaceutical dosimetry–standardization of nomenclature. J Nucl Med. 2009;50(3):477–84.
Elsayed M, et al. Comparison of Tc-99m MAA planar versus SPECT/CT imaging for lung shunt fraction evaluation prior to Y-90 radioembolization: are we overestimating lung shunt fraction? Cardiovasc Intervent Radiol. 2021;44(2):254–60.
Knešaurek K. An estimate of (90)Y dosimetry for bremsstrahlung SPECT/CT imaging in liver therapy with (90)Y microspheres. Eur J Radiol. 2021;139: 109698.
Potrebko PS, et al. SPECT/CT image-based dosimetry for Yttrium-90 radionuclide therapy: application to treatment response. J Appl Clin Med Phys. 2018;19(5):435–43.
Nelson A, Swallen A, Dewaraja Y. Evaluation of a voxel-based yttrium-90 (Y-90) dose calculation method for Bremsstrahlung SPECT using a liver phantom. J Nucl Med. 2016;57(supplement 2):306–306.
Skanjeti A, et al. Selective internal radiation therapy of hepatic tumors: morphologic and functional imaging for voxel-based computer-aided dosimetry. Biomed Pharmacother. 2020;132: 110865.
Sankhla T, et al. Role of resin microsphere y90 dosimetry in predicting objective tumor response, survival and treatment related toxicity in surgically unresectable colorectal liver metastasis: a retrospective single institution study. Cancers (Basel). 2021;13(19):4908.
Program, N.C.T.E. Common terminology criteria for adverse events (CTCAE). 2020 [cited 6 February 2022]; https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_8.5x11.pdf.
Oken MM, et al. Toxicity and response criteria of the Eastern cooperative oncology group. Am J Clin Oncol. 1982;5(6):649–55.
Garin E, et al. Boosted selective internal radiation therapy with 90Y-loaded glass microspheres (B-SIRT) for hepatocellular carcinoma patients: a new personalized promising concept. Eur J Nucl Med Mol Imaging. 2013;40(7):1057–68.
Chiesa C, et al. Radioembolization of hepatocarcinoma with 90Y glass microspheres: development of an individualized treatment planning strategy based on dosimetry and radiobiology. Eur J Nucl Med Mol Imaging. 2015;42(11):1718–38.
Garin E, et al. High impact of macroaggregated albumin-based tumour dose on response and overall survival in hepatocellular carcinoma patients treated with (90) Y-loaded glass microsphere radioembolization. Liver Int official J Int Assoc Study Liver. 2017;37(1):101–10.
Chan KT, et al. Hepatotoxic dose thresholds by positron-emission tomography after yttrium-90 radioembolization of liver tumors: a prospective single-arm observational study. Cardiovasc Intervent Radiol. 2018;41(9):1363–72.
South CD, et al. Yttrium-90 microsphere induced gastrointestinal tract ulceration. World J Surg Oncol. 2008;6:93.
Riaz A, et al. Complications following radioembolization with yttrium-90 microspheres: a comprehensive literature review. J Vasc Interv Radiol. 2009;20(9):1121–30.
Lam M et al. A global evaluation of advanced dosimetry in transarterial radioembolization of hepatocellular carcinoma with Yttrium-90: the TARGET study. Eur J Nucl Med Mol Imaging. 2022;49(10):3340–52.
Padia SA, et al. Comparison of positron emission tomography and bremsstrahlung imaging to detect particle distribution in patients undergoing yttrium-90 radioembolization for large hepatocellular carcinomas or associated portal vein thrombosis. J Vasc Interv Radiol. 2013;24(8):1147–53.
Funding
This article was funded by Sirtex Medical and Boston Scientific Corporation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Nima Kokabi, MD, receives research support from SIRtex Medical. Nima Kokabi, MD receives educational grant from Boston Scientific. Bill S. Majdalany, MD, serves on the scientific advisory board of Balt Medical. The remaining authors have no conflicts of interests to disclose.
Compliance with Ethical Standards
This study was IRB-approved, and, due to the retrospective nature of this work, no formal consent was required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Oral Presentation at the 2022 SIR Annual Scientific Meeting.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Webster, L.A., Villalobos, A., Cheng, B. et al. Correlation of Non-tumoral Liver Dose with Treatment-Related Adverse Events in Patients with Hepatocellular Carcinoma Treated with Glass-Based Yttrium-90 Radioembolization. Cardiovasc Intervent Radiol 46, 60–68 (2023). https://doi.org/10.1007/s00270-022-03314-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-022-03314-9