Abstract
Purpose
Performing a systematic review and meta-analysis to assess the evidence of intra-arterial therapies in liver metastatic breast cancer (LMBC) patients.
Methods
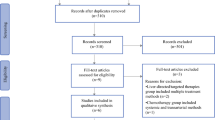
A systemic literature search was performed in PubMed, EMBASE, SCOPUS for studies regarding intra-arterial therapies in LMBC patients. Full text studies of LMBC patients (n ≥ 10) published between January 2010 and December 2020 were included when at least one outcome among response rate, adverse events or survival was available. Response rates were pooled using generalized linear mixed models. A weighted estimate of the population median overall survival (OS) was obtained under the assumption of exponentially distributed survival times.
Results
A total of 26 studies (1266 patients) were included. Eleven articles reported on transarterial radioembolization (TARE), ten on transarterial chemoembolization (TACE) and four on chemo-infusion. One retrospective study compared TARE and TACE. Pooled response rates were 49% for TARE (95%CI 32–67%), 34% for TACE (95%CI 22–50%) and 19% for chemo-infusion (95%CI 14–25%). Pooled median survival was 9.2 months (range 6.1–35.4 months) for TARE, 17.8 months (range 4.6–47.0) for TACE and 7.9 months (range 7.0–14.2) for chemo-infusion. No comparison for OS was possible due to missing survival rates at specific time points (1 and 2 year OS) and the large heterogeneity.
Conclusion
Although results have to be interpreted with caution due to the large heterogeneity, the superior response rate of TARE and TACE compared to chemo-infusion suggests first choice of TARE or TACE in chemorefractory LMBC patients. Chemo-infusion could be considered in LMBC patients not suitable for TARE or TACE.
Level of Evidence
3a-




Similar content being viewed by others
Abbreviations
- MBC:
-
Metastatic breast cancer
- OS:
-
Overall survival
- LMBC:
-
Liver metastatic breast cancer
- PFS:
-
Progression-free survival
- TARE:
-
Transarterial radioembolization
- TAE:
-
Transarterial embolization
- TACE:
-
Transarterial chemoembolization
- DEBDOX:
-
Drug-eluting beads loaded with doxorubicin
- 18F-FDG PET:
-
18F-FDG positron emission tomography
- CI:
-
Confidence interval
- RECIST:
-
Response evaluation criteria in solid tumors
References
Wu S-G, Li H, Tang L-Y, et al. The effect of distant metastases sites on survival in de novo stage-IV breast cancer: A SEER database analysis. Tumor Biology. 2017;39(6):1010428317705082.
Eng LG, Dawood S, Sopik V, et al. Ten-year survival in women with primary stage IV breast cancer. Breast Cancer Res Treat. 2016;160(1):145–52.
Kwapisz D. Oligometastatic breast cancer. Breast Cancer. 2019;26(2):138–46.
Lim B, Hortobagyi GN. Current challenges of metastatic breast cancer. Cancer Metastasis Rev. 2016;35(4):495–514.
Breedis C, Young G. The blood supply of neoplasms in the liver. Am J Pathol. 1954;30(5):969–77.
Levy J, Zuckerman J, Garfinkle R, et al. Intra-arterial therapies for unresectable and chemorefractory colorectal cancer liver metastases: A systematic review and meta-analysis. HPB (Oxford). 2018;20(10):905–15.
Duran R, Chapiro J, Schernthaner RE, Geschwind JF. Systematic review of catheter-based intra-arterial therapies in hepatocellular carcinoma: state of the art and future directions. Br J Radiol. 2015;88(1052):20140564.
Gordon AC, Uddin OM, Riaz A, Salem R, Lewandowski RJ. Making the case: Intra-arterial therapy for less common metastases. Semin Intervent Radiol. 2017;34(2):132–9.
Shamseer L, Moher D, Clarke M, et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: Elaboration and explanation. BMJ. 2015;349(jan02 1):g7647–g7647.
Munn Z PK, Aromataris E, Lockwood C, Peters M et al. Supporting Document for the Joanna Briggs Institute Levels of Evidence and Grades of Recommendation. The Joanna Briggs Institute Levels of Evidence and Grades of Recommendation Working Party. 2014 The Joanna Briggs Institute
Warton DI, Hui FK. The arcsine is asinine: The analysis of proportions in ecology. Ecology. 2011;92(1):3–10.
Schwarzer G, Chemaitelly H, Abu-Raddad LJ, Rücker G. Seriously misleading results using inverse of Freeman-Tukey double arcsine transformation in meta-analysis of single proportions. Res Synth Methods. 2019;10(3):476–83.
Gillen S, Schuster T, Meyer Zum Büschenfelde C, Friess H, Kleeff J. Preoperative/neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of response and resection percentages. PLoS Med. 2010;7(4):1000267.
Cianni R, Pelle G, Notarianni E, et al. Radioembolisation with (90)Y-labelled resin microspheres in the treatment of liver metastasis from breast cancer. Eur Radiol. 2013;23(1):182–9.
Helmberger T, Golfieri R, Pech M, et al. Clinical application of trans-arterial radioembolization in Hepatic malignancies in Europe: First results from the prospective multicentre observational study CIRSE registry for SIR-Spheres Therapy (CIRT). Cardiovasc Intervent Radiol. 2020;44:21–35.
Aarts BM, Klompenhouwer EG, Dresen RC, et al. Sequential intra-arterial infusion of 90Y-resin microspheres and mitomycin C in chemo refractory liver metastatic breast cancer patients: A single centre pilot study. Radiol Oncol. 2020;54(1):33–9.
Bagni O, Filippi L, Pelle G, Cianni R, Schillaci O. Total lesion glycolysis and sequential (90)Y-selective internal radiation therapy in breast cancer liver metastases: Preliminary results. Cancer Biother Radiopharm. 2015;30(10):421–6.
Davisson NA, Bercu ZL, Friend SC, et al. Predictors of survival after yttrium-90 radioembolization of chemotherapy-refractory hepatic metastases from breast cancer. J Vasc Interv Radiol. 2020;31(6):925–33.
Gordon AC, Gradishar WJ, Kaklamani VG, et al. Yttrium-90 radioembolization stops progression of targeted breast cancer liver metastases after failed chemotherapy. J Vasc Interv Radiol. 2014;25(10):1523–1532
Saxena A, Kapoor J, Meteling B, Morris DL, Bester L. Yttrium-90 radioembolization for unresectable, chemoresistant breast cancer liver metastases: A large single-center experience of 40 patients. Ann Surg Oncol. 2014;21(4):1296–303.
Fendler WP, Lechner H, Todica A, et al. Safety, efficacy, and prognostic factors after radioembolization of hepatic metastases from breast cancer: a large single-center experience in 81 patients. J Nucl Med. 2016;57(4):517–23.
Pieper CC, Meyer C, Wilhelm KE, et al. Yttrium-90 radioembolization of advanced, unresectable breast cancer liver metastases-a single-center experience. J Vasc Interv Radiol. 2016;27(9):1305–15.
Deipolyi AR, Riedl CC, Bromberg J, et al. Association of PI3K pathway mutations with early positron-emission tomography/CT imaging response after radioembolization for breast cancer liver metastases: Results of a single-center retrospective pilot study. J Vasc Interv Radiol. 2018;29(9):1226–35.
Sabet A, Ries M, Al-Khalaf Y, et al. Early metabolic response assessment of breast cancer liver metastases: 4-week posttreatment FDG PET predicts survival after 90Y microsphere radioembolization. Nuklearmedizin. 2019;58(3):242–8.
Martin RC, Robbins K, Fages JF, et al. Optimal outcomes for liver-dominant metastatic breast cancer with transarterial chemoembolization with drug-eluting beads loaded with doxorubicin. Breast Cancer Res Treat. 2012;132(2):753–63.
Joshi J, Robbins K, Valek V, et al. Transarterial chemoembolization with drug-eluting beads loaded with doxorubicin for the treatment of metastatic breast cancer to the liver: Results from a multiinstitutional registry. J Interventional Oncol. 2010;3(2):39–47.
Eichler K, Jakobi S, Gruber-Rouh T, Hammerstingl R, Vogl TJ, Zangos S. Transarterial chemoembolisation (TACE) with gemcitabine: phase II study in patients with liver metastases of breast cancer. Eur J Radiol. 2013;82(12):e816-822.
Lindgaard SC, Brinch CM, Jensen BK, et al. Hepatic arterial therapy with oxaliplatin and systemic capecitabine for patients with liver metastases from breast cancer. Breast. 2019;43:113–9.
Vogl TJ, Naguib NN, Nour-Eldin NE, Eichler K, Zangos S, Gruber-Rouh T. Transarterial chemoembolization (TACE) with mitomycin C and gemcitabine for liver metastases in breast cancer. Eur Radiol. 2010;20(1):173–80.
Lin YT, Medioni J, Amouyal G, Dean C, Sapoval M, Pellerin O. Doxorubicin-loaded 70–150 mum microspheres for liver-dominant metastatic breast cancer: Results and outcomes of a pilot study. Cardiovasc Intervent Radiol. 2017;40(1):81–9.
Cho SW, Kitisin K, Buck D, et al. Transcatheter arterial chemoembolization is a feasible palliative locoregional therapy for breast cancer liver metastases. Int J Surg Oncol. 2010;2010:251621.
Duan XF, Dong NN, Zhang T, Li Q. Treatment outcome of patients with liver-only metastases from breast cancer after mastectomy: A retrospective analysis. J Cancer Res Clin Oncol. 2011;137(9):1363–70.
Farshid P, Darvishi A, Naguib N, et al. Repetitive chemoembolization of hypovascular liver metastases from the most common primary sites. J Future Oncology. 2013;9(3):419–26.
Nielsen DL, Norgaard H, Vestermark LW, et al. Intrahepatic and systemic therapy with oxaliplatin combined with capecitabine in patients with hepatic metastases from breast cancer. Breast. 2012;21(4):556–61.
Tewes M, Peis MW, Bogner S, et al. Hepatic arterial infusion chemotherapy for extensive liver metastases of breast cancer: efficacy, safety and prognostic parameters. J Cancer Res Clin Oncol. 2017;143(10):2131–41.
Fu S, Naing A, Moulder SL, et al. Phase I trial of hepatic arterial infusion of nanoparticle albumin-bound paclitaxel: Toxicity, pharmacokinetics, and activity. Mol Cancer Ther. 2011;10(7):1300–7.
Tsimberidou AM, Vaklavas C, Fu S, et al. Hepatic arterial infusion therapy in advanced cancer and liver-predominant disease: the MD anderson experience. Hepatogastroenterology. 2013;60(127):1611–23.
Aarts BM, Klompenhouwer EG, Dresen RC, et al. Intra-arterial Mitomycin C infusion in a large cohort of advanced liver metastatic breast cancer patients: Safety, efficacy and factors influencing survival. Breast Cancer Res Treat. 2019;176(3):597–605.
Chang J, Charalel R, Noda C, et al. Liver-dominant Breast cancer metastasis: A comparative outcomes study of chemoembolization versus radioembolization. Anticancer Res. 2018;38(5):3063–8.
Haug AR, Tiega Donfack BP, Trumm C, et al. 18F-FDG PET/CT predicts survival after radioembolization of hepatic metastases from breast cancer. J Nucl Med. 2012;53(3):371–7.
Barabasch A, Heinzel A, Bruners P, Kraemer NA, Kuhl CK. Diffusion-weighted MRI Is superior to PET/CT in predicting survival of patients undergoing (90)Y radioembolization of hepatic metastases. Radiology. 2018;288(3):764–73.
Li XP, Meng ZQ, Guo WJ, Li J. Treatment for liver metastases from breast cancer: Results and prognostic factors. World J Gastroenterol. 2005;11(24):3782–7.
Claessens AKM, Ramaekers BLT, Lobbezoo DJA, et al. Quality of life in a real-world cohort of advanced breast cancer patients: A study of the SONABRE Registry. Qual Life Res. 2020;29(12):3363–74.
Schrijver W, Suijkerbuijk KPM, van Gils CH, van der Wall E, Moelans CB, van Diest PJ. Receptor conversion in distant breast cancer metastases: A systematic review and meta-analysis. J Natl Cancer Inst. 2018;110(6):568–80.
Angus L, Smid M, Wilting SM, et al. The genomic landscape of metastatic breast cancer highlights changes in mutation and signature frequencies. Nat Genet. 2019;51(10):1450–8.
Funding
This study was not supported by any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants performed by any of the authors.
Informed Consent
For this type of study, formal consent is not required.
Consent for Publication
For this type of study, consent for publication is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix 1: Full Search Pubmed
Appendix 1: Full Search Pubmed
((“Breast Neoplasms”[Mesh] OR ((breast*[tiab] OR mammary[tiab] OR mamma[tiab]) AND (neoplasm*[tiab] OR tumor[tiab] OR tumors[tiab] OR tumour*[tiab] OR cancer*[tiab] OR malign*[tiab] OR oncolog*[tiab] OR carcinom*[tiab])) OR (mammacarcinom*[tiab] OR mamma-carcinom*[tiab])) AND (“Liver Neoplasms”[Mesh] OR liver[tiab] OR hepatic[tiab] OR hepatocellular[tiab]) AND (“Neoplasm Metastasis”[MeSH] OR metasta*[tiab] OR metasti*[tiab]) OR LMBC[tiab]) AND (“Infusions, Intra-Arterial”[MeSH] OR intra arterial infusion*[tiab] OR intraarterial infusion*[tiab] OR intraarterial therap*[tiab] OR regional arterial infusion*[tiab] OR intra arterial therap*[tiab] OR hepatic arterial infusion*[tiab] OR hepatic arterial catheter*[tiab] OR (intra-hepatic[tiab] AND infusion*[tiab]) OR HAI[tiab] OR HAIC[tiab] OR chemo emboliz*[tiab] OR chemoemboliz*[tiab] OR chemoembolis*[tiab] OR chemo-embolis*[tiab] OR TACE[tiab] OR TAE[tiab] OR radioemboliz*[tiab] OR TARE[tiab] OR radioembolis*[tiab] OR radio-emboliz*[tiab] OR radio-embolis*[tiab] OR selective internal radiation therap*[tiab] OR selective internal radiotherap*[tiab] OR selective internal RT[tiab] OR selective internal radionuclide therap*[tiab] OR selective intraarterial radionuclide therap*[tiab] OR SIRT[tiab] OR intra-arterial chemotherap*[tiab] OR intraarterial chemotherap*[tiab] OR radiofrequency ablat*[tiab] OR regional infusion*[tiab] OR hepatic infusion*[tiab] OR arterial infusion*[tiab] OR arterial cytostatic infusion*[tiab] OR locoregional chemotherap*[tiab] OR intra-arterial catheter*[tiab] OR intraarterial catheter*[tiab] OR intrahepatic arterial chemo*[tiab] OR regional chemotherap*[tiab] OR transarterial immunochemotherap*[tiab] OR transarterial immuno-chemotherap*[tiab] OR local immunochemotherap*[tiab] OR local immuno-chemotherap*[tiab] OR intra-arterial chemo-infusion*[tiab] OR chemofiltration*[tiab] OR locoregional chemotherap*[tiab] OR arterial infusion chemotherap*[tiab] OR loco-regional cancer therap*[tiab] OR locoregional cancer therap*[tiab] OR hepatic artery infusion*[tiab] OR transarterial emboliz*[tiab] OR transarterial embolis*[tiab] OR bland emboliz*[tiab] OR bland embolis*[tiab]).
Rights and permissions
About this article
Cite this article
Aarts, B.M., Muñoz, F.M.G., Wildiers, H. et al. Intra-Arterial Therapies for Liver Metastatic Breast Cancer: A Systematic Review and Meta-Analysis. Cardiovasc Intervent Radiol 44, 1868–1882 (2021). https://doi.org/10.1007/s00270-021-02906-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-021-02906-1