Abstract
Purpose
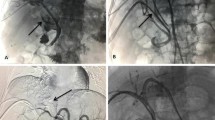
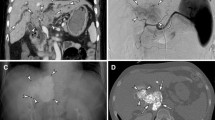
This study was performed to evaluate the accumulation of lipiodol emulsion (LE) and adverse events during our initial experience of balloon-occluded trans-catheter arterial chemoembolization (B-TACE) for hepatocellular carcinoma (HCC) compared with conventional TACE (C-TACE).
Methods
B-TACE group (50 cases) was compared with C-TACE group (50 cases). The ratio of the LE concentration in the tumor to that in the surrounding embolized liver parenchyma (LE ratio) was calculated after each treatment. Adverse events were evaluated according to the Common Terminology Criteria for Adverse Effects (CTCAE) version 4.0.
Results
The LE ratio at the level of subsegmental showed a statistically significant difference between the groups (t test: P < 0.05). Only elevation of alanine aminotransferase was more frequent in the B-TACE group, showing a statistically significant difference (Mann–Whitney test: P < 0.05). While B-TACE caused severe adverse events (liver abscess and infarction) in patients with bile duct dilatation, there was no statistically significant difference in incidence between the groups. Multivariate logistic regression analysis suggested that the significant risk factor for liver abscess/infarction was bile duct dilatation (P < 0.05).
Conclusion
The LE ratio at the level of subsegmental showed a statistically significant difference between the groups (t test: P < 0.05). B-TACE caused severe adverse events (liver abscess and infarction) in patients with bile duct dilatation.

Similar content being viewed by others
References
Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: chemoembolization improves survival. Hepatology. 2003;37:429–42.
Llovet JM, Real MI, Montana X, et al. Arterial embolization or chemoembolization verses systemic treatment in patients with unresectable hepatocellular carcinoma: a randomized controlled trial. Lancet. 2002;359:1734–9.
Yamada R, Sato M, Kawabata M, et al. Hepatic artery embolization in 120 patients with unresectable hepatoma. Radiology. 1983;148:397–401.
Lin DY, Liaw YF, Lee TY, Lai CM. Hepatic arterial embolization in patients with unresectable hepatocellular carcinoma—a randomized controlled trial. Gastroenterology. 1998;94:453–6.
Ikeda K, Kumada H, Saitoh S, et al. Effect of repeated transcatheter arterial embolization on the survival time in patients with hepatocellular carcinoma. An analysis by the Cox proportional hazard model. Cancer. 1991;68:2150–4.
Llovet JM, Real MI, Montaña X, et al. Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet. 2002;359:1734–9.
Lo CM, Ngan H, Tso WK, et al. Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology. 2002;35:1164–71.
Cammà C, Schepis F, Orlando A, et al. Transarterial chemoembolization for unresectable hepatocellular carcinoma: meta-analysis of randomized controlled trials. Radiology. 2002;224:47–54.
Takayasu K, Arii S, Ikai I, et al. Prospective cohort study of transarterial chemoembolization for unresectable hepatocellular carcinoma in 8510 patients. Gastroenterology. 2006;131:461–9.
Ikeda M, Arai Y, Park SJ, et al. Prospective study of transcatheter arterial chemoembolization for unresectable hepatocellular carcinoma: an Asian cooperative study between Japan and Korea. J Vasc Intervent Radiol. 2013;24:490–500.
Irie T, Kuramochi M, Takahashi N. Dense accumulation of lipiodol emulsion in hepatocellular carcinoma nodule during selective balloon-occluded transarterial chemoembolisation: measurement of balloon-occluded arterial stump pressure. Cardiovasc Intervent Radiol. 2013;36:706–13.
Irie T, Kuramochi M, Takahashi N. Improved accumulation of lipiodol under balloon-occluded transarterial chemoembolisation (B-TACE)for hepatocellular carcinoma: measurement of blood pressure at the embolized artery before and after balloon inflation. Jpn J Intervent Radiol. 2011;26:49–54.
Irie T, Kuramochi M, Ishikawa A. Safety of balloon-occluded transarterial chemoembolization (B-TACE) for hepatocellular carcinoma: analysis of adverse event in 82 cases (in Japanese). Jpn J Intervent Radiol. 2011;26:175–81.
Llovet JM, DiBisceglie AM, Bruix J, et al. Design and endpoints of clinical trials in hepatocellular carcinoma. J Natl Cancer Inst. 2008;100(10):698–711.
Kokudo N, Hasegawa K, Akahane M, et al. Evidence-based Clinical Practice Guidelines for Hepatocellular Carcinoma: the Japan Society of Hepatology 2013 update (3rd JSH-HCC Guidelines). Hepatol Res. 2015;45:128–246.
Eijun S, Hayashida T, Sakamoto I, et al. Vascular complications of hepatic artery after transcatheter arterial chemoembolization in patients with hepatocellular carcinoma. Am J Roentgenol. 2010;195:245–51.
Chen C, Chen PJ, Yang PM, et al. Meta-analysis comparing TACE/TAE/TOCE with conservative or suboptimal treatment. Transarterial therapies significantly decreased mortality. TACE and TAE achieved the same survival benefit. Clinical and microbiological features of liver abscess after transarterial embolization for hepatocellular carcinoma. Am J Gastroenterol. 1997;92(12):2257–9.
Cohen SE, Safadi R, Verstandig A, et al. Liver-spleen infarcts following transcatheter chemoembolization: a case report and review of the literature on adverse effects. Dig Dis Sci. 1997;42(5):938–43.
Kobayashi S, Nakanuma Y, Terada T, et al. Postmortem survey of bile duct necrosis and biloma in hepatocellular carcinoma after transcatheter arterial chemoembolization therapy: relevance to microvascular damages of peribiliary capillary plexus. Am J Gastroenterol. 1993;88:1410–5.
Choi BI, Kim HC, Han JK, et al. Therapeutic effect of transcatheter oily chemoembolization therapy for encapsulated nodular hepatocellular carcinoma: CT and pathologic findings. Radiology. 1992;182:709–13.
Bhattacharya S, Novell JR, Winslet MC, et al. Iodized oil in the treatment of hepatocellular carcinoma. Br J Surg. 1994;81:1563–71.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author and co-authors have no conflict of interest to disclose with respect to this study.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in this study to the B-TACE or C-TACE procedure.
Rights and permissions
About this article
Cite this article
Maruyama, M., Yoshizako, T., Nakamura, T. et al. Initial Experience with Balloon-Occluded Trans-catheter Arterial Chemoembolization (B-TACE) for Hepatocellular Carcinoma. Cardiovasc Intervent Radiol 39, 359–366 (2016). https://doi.org/10.1007/s00270-015-1237-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-015-1237-6