Abstract
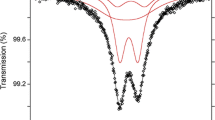
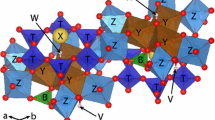
An Fe-rich fluor-elbaite was thermally treated in air and hydrogen atmosphere up to 800 °C to study potential changes in Fe- and Al-ordering over the octahedrally coordinated Y and Z sites. Overall, the experimental data (structural refinement, electron and ion microprobe, Mössbauer, infrared and optical absorption spectroscopy) show that thermal treatment of fluor-elbaite results in an increase of Fe contents at the Z site balanced by an increase of Al at the Y site. On the basis of this and previous experimental studies on Fe–Mg–Al-bearing tourmalines, it can be stated that the intersite Fe–Mg–Al exchange rates are significant at temperatures around 700–800 °C. Thermal treatment results in an increase of ca. 0.30 Fe atoms per formula unit at the Z site compensated by a similar increase of (Mg + Al) at the Y site, following the exchange reaction YFe + Z(Mg + Al) → ZFe + Y(Mg + Al). Since the tourmaline nomenclature is based on the occupancy of ions at each structural site, the intersite Fe–Mg–Al ordering may determine the tourmaline species. This means that effectively the name associated with a given composition may be a function of the sample thermal history.




Similar content being viewed by others
References
Andreozzi GB, Bosi F, Longo M (2008) Linking Mössbauer and structural parameters in elbaite-schorl-dravite tourmalines. Am Mineral 93:658–666
Bosi F (2013) Bond-valence constraints around the O1 site of tourmaline. Mineral Mag 77:343–351
Bosi F (2018) Tourmaline crystal chemistry. Am Mineral 103:298–306
Bosi F, Lucchesi S (2007) Crystal chemical relationships in the tourmaline group: Structural constraints on chemical variability. Am Mineral 92:1054–1063
Bosi F, Andreozzi GB, Federico M, Graziani G, Lucchesi S (2005) Crystal chemistry of the elbaite-schorl series. Am Mineral 90:1784–1792
Bosi F, Andreozzi GB, Skogby H, Lussier AJ, Abdu Y, Hawthorne FC (2013) Fluor-elbaite, Na(Li1.5Al1.5)Al6(Si6O18)(BO3)3(OH)3F, a new mineral species of the tourmaline supergroup. Am Mineral 98:297–303
Bosi F, Andreozzi GB, Hålenius U, Skogby H (2015a) Experimental evidence for partial Fe2+ disorder at the Y and Z sites of tourmaline: a combined EMP, SREF, MS, IR and OAS study of schorl. Mineral Mag 79:515–528
Bosi F, Skogby H, Lazor P, Reznitskii L (2015b) Atomic arrangements around the O3 site in Al- and Cr-rich oxy-tourmalines: a combined EMP, SREF, FTIR and Raman study. Phys Chem Miner 42:441–453
Bosi F, Skogby H, Balić-Žunić T (2016a) Thermal stability of extended clusters in dravite: a combined EMP, SREF and FTIR study. Phys Chem Mineral 43:395–407
Bosi F, Skogby H, Hålenius U (2016b) Thermally induced cation redistribution in Fe-bearing oxy-dravite and potential geothermometric implications. Contrib Mineral Petrol 171:47
Bosi F, Reznitskii L, Hålenius U, Skogby H (2017a) Crystal chemistry of Al-V-Cr oxy-tourmalines from Sludyanka complex, Lake Baikal, Russia. Eur J Mineral 29:457–472
Bosi F, Cámara F, Ciriotti ME, Hålenius U, Reznitskii L, Stagno V (2017b) Crystal-chemical relations and classification problems of tourmalines belonging to the oxy-schorl–oxy-dravite–bosiite–povondraite series. Eur J Mineral 29:445–455
Bosi F, Naitza S, Skogby H, Secchi F, Conte AM, Cuccuru S, Hålenius U, De La Rosa N, Kristiansson P, Nilsson EJC, Ros L, Andreozzi GB (2018a) Late magmatic controls on the origin of schorlitic and foititic tourmalines from late-Variscan peraluminous granites of the Arbus pluton (SW Sardinia, Italy): crystal-chemical study and petrological constraints. Lithos 308–309:395–411
Bosi F, Skogby H, Hålenius U, Ciriotti M (2018b) Experimental cation redistribution in the tourmaline lucchesiite, CaFe2 + 3Al6(Si6O18)(BO3)3(OH)3O. Phys Chem Mineral 45:621–632
Burns PC, MacDonald DJ, Hawthorne FC (1994) The crystal chemistry of manganese-bearing elbaite. Can Mineral 32:31–41
Deloule E, Chaussidon M, Allé P (1992) Instrumental limitations for isotope ratios measurements with a Cameca IMS 3f ion microprobe: the example of H, B, S, Sr. Chem Geol 101:187–192
Dutrow BL, Henry DJ (2011) Tourmaline: a geologic DVD. Elements 7:301–306
Ertl A, Tillmanns E, Ntaflos T, Francis C, Giester G, Körner W, Hughes JM, Lengauer C, Prem M (2008) Tetrahedrally coordinated boron in Al-rich tourmaline and its relationship to the pressure–temperature conditions of formation. Eur J Mineral 20:881–888
Ertl A, Rossman GR, Hughes JM, London D, Wang Y, O’Leary JA, Dyar MD, Prowatke S, Ludwig T, Tillmanns E (2010) Tourmaline of the elbaite-schorl series from the Himalaya Mine, Mesa Grande, California, USA: A detailed investigation. Am Mineral 95:24–40
Ertl A, Kolitsch U, Dyar MD, Hughes JM, Rossman GR, Pieczka A, Henry DJ, Pezzotta F, Prowatke S, Lengauer CL, Körner W, Brandstatter F, Francis CA, Prem M, Tillmans E (2012a) Limitations of Fe2+ and Mn2+ site occupancy in tourmaline: evidence from Fe2+- and Mn2+-rich tourmaline. Am Mineral 97:1402–1416
Ertl A, Schuster R, Hughes JM, Ludwig T, Meyer H-P, Finger F, Dyar MD, Ruschel K, Rossman GR, Klötzli U, Brandstätter F, Lengauer CL, Tillmanns E (2012b) Li-bearing tourmalines in Variscan pegmatites from the Moldanubian nappes, Lower Austria. Eur J Mineral 24:695–715
Ertl A, Henry DJ, Tillmanns E (2018) Tetrahedral substitutions in tourmaline: a review. Eur J Mineral 30:465–470
Federico M, Andreozzi GB, Lucchesi S, Graziani G, César-Mendes J (1998) Crystal chemistry of tourmalines. I. Chemistry, compositional variations and coupled substitutions in the pegmatite dikes of the Cruzeiro mine, Minas Gerais, Brazil. Can Mineral 36:415–431
Ferrow E (2009) Non-integral hybrid ions in tourmaline: buffering and geo-thermometry. Eur J Mineral 21:241–250
Filip J, Bosi F, Novák M, Skogby H, Tuček J, Čuda J, Wildner M (2012) Redox processes of iron in the tourmaline structure: example of the high-temperature treatment of Fe3+-rich schorl. Geochim Cosmochim Acta 86:239–256
Fuchs Y, Lagache M, Linares J (1998) Fe-tourmaline synthesis under different T and ƒO2 conditions. Am Mineral 83:525–534
Gatta GD, Bosi F, McIntyre GJ, Skogby H (2014) First accurate location of two proton sites in tourmaline: a single-crystal neutron diffraction study of oxy-dravite. Mineral Mag 78:681–692
Gonzalez-Carreño T, Fernandez M, Sanz J (1988) Infrared and electron microprobe analysis of tourmalines. Phys Chem Mineral 15:452–460
Grew ES, Krivovichev SV, Hazen RM, Hystad G (2016) Evolution of structural complexity in boron minerals. Can Mineral 54:125–143
Hawthorne FC (2016) Short-range atomic arrangements in minerals. I: the minerals of the amphibole, tourmaline and pyroxene supergroups. Eur J Mineral 28:513–536
Henry DJ, Dutrow BL (1992) Tourmaline in a low grade clastic metasedimentary rock: an example of the petrogenetic potential of tourmaline. Contrib Mineral Petrol 112:203–218
Henry DJ, Dutrow BL (1996) Metamorphic tourmaline and its petrologic applications. In: Grew ES, Anvitz LM (eds) Boron: mineralogy, petrology and geochemistry, reviews in mineralogy and geochemistry, vol 33. Mineralogical Society of America, Chantilly, Virginia, pp 503–557
Henry DJ, Novák M, Hawthorne FC, Ertl A, Dutrow B, Uher P, Pezzotta F (2011) Nomenclature of the tourmaline supergroup minerals. Am Mineral 96:895–913
Henry DJ, Novák M, Hawthorne FC, Ertl A, Dutrow B, Uher P, Pezzotta F (2013) Erratum Am Mineral 98:524
Kutzschbach M, Wunder B, Rhede D, Koch-Müller M, Ertl A, Giester G, Heinrich W, Franz G (2016) Tetrahedral boron in natural and synthetic HP/UHP tourmaline: evidence from Raman spectroscopy, EMPA, and single-crystal XRD. Am Mineral 101:93–104
Libowitzky E (1999) Correlation of O-H stretching frequencies and O–H⋯O hydrogen bond lengths in minerals. Monatsh Chemie 130:1047–1059
Lussier A, Ball NA, Hawthorne FC, Henry DJ, Shimizu R, Ogasawara Y, Ota T (2016) Maruyamaite, K(MgAl2)(Al5Mg)Si6O18(BO3)3(OH)3O, a potassium-dominant tourmaline from the ultrahigh-pressure Kokchetav massif, northern Kazakhstan: description and crystal structure. Am Mineral 101:355–361
Marschall HR, Korsakov AV, Luvizotto GL, Nasdala L, Ludwig T (2009) On the occurrence and boron isotopic composition of tourmaline in (ultra)high-pressure metamorphic rocks. J Geol Soc 166:811–823
Mattson SM, Rossman GR (1984) Ferric iron in tourmaline. Phys Chem Mineral 11:225–234
Mattson SM, Rossman GR (1987) Fe2+-Fe3+ interactions in tourmaline. Phys Chem Mineral 14:163–171
Pieczka A, Kraczka J (2004) Oxidized tourmalines—a combined chemical, XRD and Mossbauer study. Eur J Mineral 16:309–321
Pouchou JL, Pichoir F (1991) Quantitative analysis of homogeneous or stratified microvolumes applying the model “PAP”. In: Heinrich KFJ, Newbury DE (eds) Electron Probe Quantitation. Plenum, New York, pp 31–75
Prescher C, McCammon C, Dubrowinsky L (2012) MossA: a program for analyzing energy-domain Mössbauer spectra from conventional and synchrotron sources. J Appl Cryst 45:329–331
Sheldrick GM (2013) SHELXL2013. University of Göttingen, Germany
Skogby H, Bosi F, Lazor P (2012) Short-range order in tourmaline: a vibrational spectroscopic approach to elbaite. Phys Chem Mineral 39:811–816
Taran MN, Rossman GR (2002) High-temperature, high-pressure optical spectroscopic study of ferric-iron-bearing tourmaline. Am Mineral 87:1148–1153
Taran MN, Lebedev AS, Platonov AN (1993) Optical absorption spectroscopy of synthetic tourmalines. Phys Chem Mineral 20:209–220
van Hinsberg VJ, Schumacher JC (2009) The geothermobarometric potential of tourmaline, based on experimental and natural data. Am Mineral 94:761–770
van Hinsberg VJ, Henry DJ, Marschall HR (2011) Tourmaline: an ideal indicator of its host environment. Can Mineral 49:1–16
Watenphul A, Burgdorf M, Schlüter J, Horn I, Malcherek T, Mihailova B (2016) Exploring the potential of Raman spectroscopy for crystallochemical analyses of complex hydrous silicates: II. Tourmalines. Am Mineral 101:970–985
Acknowledgements
Funding by Sapienza University of Rome (Prog. Università 2017 to F.B.) and the Swedish Research Council (H.S.) is gratefully acknowledged. E. Tillmanns and D.J. Henry are thanked for their constructive comments.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bosi, F., Skogby, H. & Hålenius, U. Thermally induced cation redistribution in fluor-elbaite and Fe-bearing tourmalines. Phys Chem Minerals 46, 371–383 (2019). https://doi.org/10.1007/s00269-018-1009-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00269-018-1009-3