Abstract
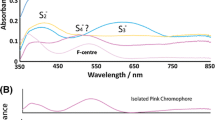
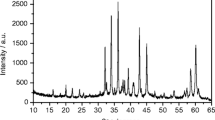
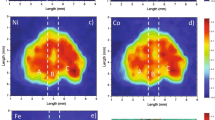
Natural hydrothermal ZnCO3 crystal aggregates are nominally anhydrous phases with interfacial water, with substitutional divalent cations and decarbonation c. 300°C. All these common features must be involved during the experimental heating of a thermoluminescence (TL) glow curve up to 500°C: dehydration–dehydroxylation, phase transition and ion transition of point defects in Zn2+ positions. A representative specimen of natural smithsonite was analysed by X-ray fluorescence spectrometry, field emission scanning electron microscopy (FESEM) with a chemical probe of energy dispersive spectrometer, high temperature in situ X-ray diffraction, differential scanning calorimetry, differential thermal analyses coupled to thermogravimetry, TL, radioluminescence and high resolution spectra thermoluminescence (3DTL), to gain an overview of the spectra emission and defects linkages modified by heating from room temperature up to 500°C. The ZnCO3 specimen contains minor amounts of Ca, Cu, Cr, Cd, Pb, Ce, Co, Ni, Mn and Fe. Under FESEM, it displays CaCO3 clusters and oscillatory zoning distribution with lamellae ranging from Ca0.11Zn0.89 to Ca0.19Zn0.81. The analytical results suggest assignments of defects and processes to measured 3DTL emission bands, as follows: (1) peak at ~260°C, ~360 nm bonds, breaking during the thermal decarbonation process; (2) peak ~120°C, ~340 nm: non-bridging oxygen centres associated to a complex dehydration–dehydroxylation process and (3) peak at ~170°C, ~650 nm, crystal field effects on the thermoluminescence of Mn2+ centres and associated transitional elements in the ZnCO3 phase.




Similar content being viewed by others
References
Bøtter-Jensen L, Duller GAT (1992) A new system for measuring OSL from quartz samples. Nucl Tracks Radiat Meas 20:549–553. doi:10.1016/1359-0189(92)90003-E
Calderon T, Townsend PD, Beneitez P, Garcia-Guinea J, Millan A, Rendell HM, Tookey A, Urbina M, Wood RA (1996) Crystal field effects on the thermoluminescence of manganese in carbonate lattices. Radiat Meas 26(5):719–731. doi:10.1016/S1350-4487(97)82886-7
Correcher V, Delgado A (1998) On the use of natural quartz as transfer dosimeter in retrospective dosimetry. Radiat Meas 29(3–4):411–414. doi:10.1016/S1350-4487(98)00040-7
Frost RL, Hales MC, Wain DL (2008) Raman spectroscopy of smithsonite. J Raman Spectrosc 39:108–114. doi:10.1002/jrs.1835
Gadalla AM (1984) Kinetics of non-isothermal calcination of ZnCO3 in air. Int J Chem Kinet 16:1471–1479. doi:10.1002/kin.550161202
Galloway RB (2003) Limestone: some observations on luminescence in the region of 360 nm. Radiat Meas 37(2):177–185. doi:10.1016/S1350-4487(02)00185-3
Garcia-Guinea J, Correcher V (2000) Luminescence spectra of alkali feldspars: influence of crushing on the ultraviolet emission band. Spectrosc Lett 33:103–113. doi:10.1080/00387010009350062
Garcia-Guinea J, Correcher V, Sanchez-Munoz L, Finch AA, Hole DE, Townsend PD (2007) On the luminescence emission band at 340 nm of stressed tectosilicate phases. Nucl Instrum Methods A 580:648–651. doi:10.1016/j.nima.2007.05.111
Gmelin E, Sarge SM (1995) Calibration of differential scanning calorimeters. Pure Appl Chem 67:1789–1800. doi:10.1351/pac199567111789
Gmelin E, Sarge SM (2000) Temperature, heat and heat flow rate calibration of differential scanning calorimeters. Thermochim Acta 347:9–13. doi:10.1016/S0040-6031(99)00424-4
Gómez-Fernández F, Both RA, Mangas J, Arribas A (2000) Metallogenesis of Zn–Pb carbonate-hosted mineralization in the southeastern region of the Picos de Europa (Central Northern Spain) Province: geologic, fluid inclusion, and stable isotope studies. Econ Geol 95(1):19–39
Hales MC, Frost RL (2008) Thermal analysis of smithsonite and hydrozincite. J Therm Anal Calorim 91(3):855–860. doi:10.1007/s10973-007-8571-0
Haselton HT, Goldsmith JR (1987) The decarbonation and heat-capacity of ZnCO3. Geochim Cosmochim Acta 51:261–265. doi:10.1016/0016-7037(87)90238-9
Luff BJ, Townsend PD (1993) High-sensitivity thermoluminescence spectrometer. Meas Sci Technol 4:65–71. doi:10.1088/0957-0233/4/1/011
Reddy BJ, Yamauchi J, Ravikumar RVSSN, Chandrasekhar AV, Venkataramanaiah M (2004) Optical and EPR investigations on smithsonite minerals. Radiat Eff Defect S 159(3):141–147. doi:10.1080/10420150410001669587
Sabbah R, Xu-Wu A, Chickos JS, Planas Leitão ML, Roux MV, Torres LA (1999) Reference materials for calorimetry and differential thermal analysis. Thermochim Acta 331:93–204. doi:10.1016/S0040-6031(99)00009-X
Sarge SM, Gmelin E, Höhne GWH, Cammenga HK, Hemminger W, Eysel W (1994) The caloric calibration of scanning calorimeters. Thermochim Acta 247(2):129–168. doi:10.1016/0040-6031(94)80118-5
Stipp SLS, Gutmannsbauer W, Lehmann T (1996) The dynamic nature of calcite surfaces in air. Am Min 81(1–2):1–8
Temprado M, Roux MV, Jimenez P, Guzman-Mejia R, Juaristi E (2006) Thermophysical properties of sulfur heterocycles: thiane and thiophene derivatives. Thermochim Acta 441:20–26. doi:10.1016/j.tca.2005.11.024
Yao BD, Shi HZ, Zhang XY, Zhang LD (2001) Ultraviolet photoluminescence from nonbridging oxygen hole centers in porous silica. Appl Phys Lett 78(2):174–176. doi:10.1063/1.1339844
Acknowledgments
This work was supported in part by PROJETS: C·I.C.Y.T. CGL2008-03842, Comunidad Autonoma de Madrid MATERNAS (CAM) S0505/MAT/0094, CTQ2007-60895/BQU and a Contract Post-PhD CSIC JAE-DOC.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Garcia-Guinea, J., Crespo-Feo, E., Correcher, V. et al. Thermo-optical detection of defects and decarbonation in natural smithsonite. Phys Chem Minerals 36, 431–438 (2009). https://doi.org/10.1007/s00269-009-0289-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00269-009-0289-z