Abstract
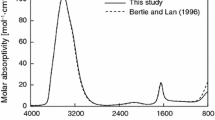
The theoretical infrared (IR) and Raman spectra of bayerite (β-Al(OH)3) are computed in the density functional theory framework, using the linear response theory. The results are consistent with the occurrence of six non-equivalent OH groups in a bayerite structure with space group P21/n. Similar to gibbsite, the transmission powder IR spectrum of bayerite in the region of the OH stretching bands is found to depend on the shape of particles. In particular, the broadening of the strong band observed at about 3,460 cm−1 in the spectrum of Al hydroxides is related to the electrostatic charges occurring at the surface of the polarized dielectric particles. The experimental correlation observed between the shape of this band and morphological parameters has therefore a physical, instead of chemical, origin.




Similar content being viewed by others
References
Balan E, Saitta AM, Mauri F, Calas G (2001) First-principles modeling of the infrared spectrum of kaolinite. Am Mineral 86:1321-1330
Balan E, Saitta AM, Mauri F, Lemaire C, Guyot F (2002a) First-principles calculation of the infrared spectrum of lizardite. Am Mineral 87:1286–1290
Balan E, Mauri F, Lemaire C, Brouder C, Guyot F, Saitta AM, Devouard B (2002b) Multiple ionic plasmon resonances in naturally-occurring multiwall nanotubes: infrared spectra of chrysotile asbestos. Phys Rev Lett 89:177401
Balan E, Lazzeri M, Saitta AM, Allard T, Fuchs Y, Mauri F (2005) First-principles study of OH stretching modes in kaolinite, dickite and nacrite. Am Mineral 90:50–60
Balan E, Lazzeri M, Morin G, Mauri F (2006) First-principles study of the OH stretching modes of gibbsite. Am Mineral 91:115–119
Baroni S, de Gironcoli S, Dal Corso A, Giannozzi P (2001) Phonons and related crystal properties from density-functional perturbation theory. Rev Modern Phys 73:515–561
Digne M, Sautet P, Raybaud P, Toulhoat H, Artacho E (2002) Structure and stability of aluminum hydroxides: a theoretical study. J Phys Chem B 106:5155–5162
Farmer VC (1974) The infrared spectra of minerals. Mineralogical Society, London
Farmer VC (1998) Differing effect of particle size and shape in the infrared and Raman spectra of kaolinite. Clay Miner 33:601–604
Farmer VC (2000) Transverse and longitudinal crystal modes associated with OH stretching vibrations in single crystals of kaolinite and dickite. Spectrochim Acta Part A 56:927–930
Fuchs R (1975) Theory of the optical properties of ionic crystal cubes. Phys Rev B 11:1732–1740
Gale JD, Rohl AL, Milman V, Warren MC (2001) An ab initio study of the structure and properties of aluminium hydroxide: gibbsite and bayerite. J Phys Chem B 105:10236–10242
Hochepied J-F, Ilioukhina O, Berger M-H (2003) Effect of the mixing procedure on aluminium (oxide)-hydroxide obtained by precipitation of aluminium nitrate with soda. Mater Lett 57:2817–2822
Jodin M-C, Gaboriaud F, Humbert B (2004) Repercussions of size heterogeneity on the measurement of specific surface areas of colloidal minerals: combination of macroscopic and microscopic analyses. Am Mineral 89:1456–1462
Jones RC (1945) A generalization of the dielectric ellipsoid problem. Phys Rev 68:93–96
Kleinman L, Bylander DM (1982) Efficacious form for model pseudopotentials. Phys Rev Lett 48:1425–1428
Lazzeri M, Mauri F (2003) First principles calculation of vibrational Raman spectra in large systems: signature of small rings in crystalline SiO2. Phys Rev Lett 90:036401
Lefèvre G, Duc M, Lepeut P, Caplain R, Fédoroff M (2002) Hydration of γ-alumina in water and its effects on surface reactivity. Langmuir 18:7530–7537
Lefèvre G, Pichot V, Fédoroff M (2003) Controlling particle morphology during growth of bayerite in aluminate solutions. Chem Mater 15:2584–2592
Perdew JP, Burke K, Ernzerhof M (1996) Generalized gradient approximation made simple. Phys Rev Lett 77:3865–3868
Phambu N (2003) Characterization of aluminum hydroxide thin film on metallic aluminum powder. Mater Lett 57:2907–2913
Phambu N, Humbert B, Burneau A (2000) Relation between the infrared spectra and the lateral specific surface areas of gibbsite samples. Langmuir 16:6200–6207
Rodgers KA, Gregory MR, Cooney RP (1989) Bayerite, Al(OH)3, from Raoul Island, Kermadec Group, South Pacific. Clay Miner 24:531–538
Rothbauer R, Zigan F, O’Daniel H (1967) Verfeinerung der struktur des bayerits. Al(OH)3. Zeit Kristall 125:317–331
Ruan HD, Frost RL, Kloprogge JT (2001) Comparison of Raman spectra in characterizing gibbsite, bayerite, diaspore and boehmite. J Raman Spectro 32:745–750
Rzhevskii AM, Ribeiro FH (2001) UV Raman spectroscopic study of hydrogen bonding in gibbsite and bayerite between 93 and 453 K. J Raman Spectro 32:923–928
Schoen R, Roberson CE (1970) Structures of aluminum hydroxide and geochemical implications. Am Mineral 55:43–77
Serna JC, Ocana M, Iglesias JE (1987) Optical properties of α-Fe2O3 microcrystals in the infrared. J Phys C Sol State Phys 20:473–484
Troullier N, Martins JL (1991) Efficient pseudopotentials for plane-wave calculations. Phys Rev B 43:1993–2006
Van de Hulst HC (1981) Light scattering by small particles. Dover Publications, New York
Wang S-L, Johnston CT (2000) Assignment of the structural OH stretching bands of gibbsite. Am Mineral 85:739–744
Zigan F, Joswig W, Burger N (1978) Die wasserstoffpositionen im bayerit Al(OH)3. Zeit Kristall 148:255–273
Acknowledgments
We thank S. Borensztajn (LISE, University Paris VI) for the SEM observations. Calculations were performed at the IDRIS institute (Institut du Développement et des Ressources en Informatique Scientifique) of CNRS (Centre National de la Recherche Scientifique) within the project 060411519. This work has been supported by the French National Research Agency (ANR, projet “SPIRSE”) and CNRS-INSU through the ESF Eurocores program EuroMinSci (CRP “Hydromin”). This work is IPGP contribution 2321.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Balan, E., Blanchard, M., Hochepied, JF. et al. Surface modes in the infrared spectrum of hydrous minerals: the OH stretching modes of bayerite. Phys Chem Minerals 35, 279–285 (2008). https://doi.org/10.1007/s00269-008-0221-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00269-008-0221-y