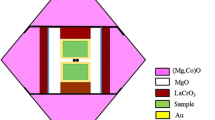
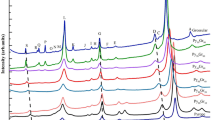
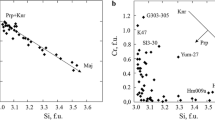
Abstract
Germanate garnets are often used as isostructural analogues of silicate garnets to provide insight into the crystal chemistry and symmetry of the less accessible natural garnet solid solutions. We synthesised two series of germanate garnets at 3 GPa along the joinVIIICa VI3 (CaGe)IVGe3O12–VIIICa VI3 Fe IV2 Ge3O12 at 900 °C and 1,100 °C. Samples with compositions close to the CaGeO3 end-member consist of tetragonal garnet with a small amount of triclinic CaGe2O5. Samples with nominal compositions between XFe=0.4 and 1.0 consist of a mixture of tetragonal and cubic garnets; whereas, single-phase cubic garnets were obtained for compositions with XFe>1.2 (XFe gives the iron content expressed in atoms per formula unit, and varies between 0 and 2 along the join). Run products which were primarily single-phase garnet were investigated using Mössbauer spectroscopy. Spectra from samples synthesised at 1,100°C consist of one well-resolved doublet that can be assigned to Fe3+ in the octahedral site of the garnet structure. A second doublet, present primarily in samples synthesised at 900°C, can be assigned to Fe2+ at the octahedral sites of the garnet structure. The relative abundance of Fe2+ decreases with increasing iron content. Transmission electron microscopy analyses confirm this tendency and show that the garnets are essentially defect-free. The unit-cell parameters of tetragonal VIIICa VI3 (CaGe)IVGe3O3 garnet decrease with increasing synthesis temperature, and the deviation from cubic symmetry becomes smaller. Cubic garnets show a linear decrease of unit-cell parameter with increasing iron content. The results are discussed in the context of iron incorporation into VIIIMg VI3 (MgSi)IVSi3O3 majorite.






Similar content being viewed by others
References
Akaogi M, Tanaka A, Ito E (2002) Garnet–ilmenite–perovskite transitions in the system Mg4Si4O12–Mg3Al2Si4O12 at high pressures and high temperatures: phase equilibria, calorimetry and implications for mantle structure. Phys Earth Planet Int 132:303–324
van Aken PA, Liebscher B, Styrsa VS (1998) Quantitative X-ray determination of iron oxidation states in minerals using Fe L2,3 electron energy-loss near-edge structure spectroscopy. Phys Chem Miner 5:323–327
Amthauer G, Annersten H, Hafner SS (1976) The Mössbauer spectrum of 57 Fe in silicate garnets. Zeit Krist 143:14–55
Angel RJ, Finger LW, Hazen RM, Kanzaki M, Weidner DJ, Liebermann RC, Veblen DR (1989) Structure and twinning of single-crystal MgSiO3 garnet synthesized at 17 GPa and 1800°C. Am Miner 74:509–512
Aust H, Völlenkle H, Wittmann A (1976) Die Kristallstruktur der Hoch-und der Tieftemperaturform von CaGe2O5. Zeit Krist 144:82–90
Chaplin TD, Ross NL, Reynard B (2000) A high-temperature and high-pressure Raman spectroscopy study of CaGeO3 garnet. Phys Chem Miner 27:213–219
Chou IM (1987) Oxygen buffer and hydrogen sensor technique at elevated pressures and temperatures. In: Barnes HL, Ulmer GC (eds) Hydrothermal experimental techniques. Wiley, New York, pp 61–99
Corgne A, Wood BJ (2003) Trace element partitioning between majorite garnet and silicate melt at lower mantle conditions. Geochim Cosmochim Acta 67(1):A70
Eugster HP (1957) Heterogeneous reactions involving oxidation and reduction at high pressures and temperature. J Chem Phys 26:1760–1761
Frost DJ, Langenhorst F (2002) The effect of Al2O3 on Fe-Mg partitioning between magnesiowüstite and magnesium silicate perovskite. Earth Planet Sci Lett 199:227–241
Ganguly J, Newton RC (1968) Thermal stability of chloritoid at high pressure and relatively high oxygen fugacity. J Petrol 9:444–466
Ganguly J, Cheng W, O′Neill HSC (1993) Syntheses, volume, and structural changes of garnets in the pyrope-grossular join: Implications for stability and mixing properties. Am Miner 78:583–593
Ganguly J, Cheng W, Tirone M (1996) Thermodynamics of aluminosilicate garnet solid solution: New experimental data, an optimized model, and thermometric applications. Contrib Mineral Petrol 126:137–151
Gasparik T (2000) Evidence for immiscibility in majorite garnet from experiments at 13–15 GPa. Geochim Cosmochim Acta 64(9):1641–1650
Geiger CA, Grodzicki M, Amthauer G (2003) The crystal chemistry and FeII-site properties of aluminosilicate garnet solid solutions as revealed by Mössbauer spectroscopy and electronic structure calculations. Phys Chem Minerals 30:280–292
Geller S, Miller CE, Treuting RG (1960) New synthetic garnets. Acta Crystallogr (A) 13:179–186
Hatch DM, Ghose S (1989) Symmetry analysis of the phase transition and twinning in MgSiO3 garnet: implications to mantle mineralogy. Am Miner 74:1221–1224
Heinemann S, Sharp TG, Seifert F, Rubie D (1997) The cubic-tetragonal phase transition in the system majorite (Mg4Si4O12)-pyrope (Mg3Al2Si3O12), and garnet symmetry in the Earth′s transition zone. Phys Chem Miner 24:206–221
Langenhorst F, Joreau P, Doukhan JC (1995) Thermal and shock metamorphism of the Tenham chondrite: A TEM examination. Geochim Cosmochim Acta 59:1835–1845
Larson AC, Von Dreele RB (1997) GSAS: General Structure Analysis System. Los Alamos National Laboratory, Document LAUR, pp 86–748
Lévy D, Barbier J (1999) Normal and inverse garnets: Ca3Fe2Ge3O12, CaGeO3–Ca3Y2Ge3O12and Mg3Y2Ge3O12. Acta Crystallogr C55:1611–1614
Long GL, Cranshaw TE, Longworth G (1983) The ideal Mössbauer effect absorber thickness. Möss Effect Ref Data J 6:42–49
McCammon CA, Ross NL (2003) Crystal chemistry of ferric iron in (Mg,Fe)(Si,Al)O3 majorite with implications for the transition zone. Phys Chem Miner 30:206–216
McCammon CA, Frost DJ, Smyth JR, Laustsen HM, Kawamoto T, Ross NL, van Aken PA (2004) Oxidation state of iron in hydrous mantle phases: Implications for subduction and mantle oxygen fugacity. Phys Earth Planet Int 143–144:157–169
Merli M, Callegari A, Cannillo E, Caucia F, Leona M, Oberti R, Ungaretti L (1995) Crystal-chemical complexity in natural garnets: structural constraints on chemical variability. Eur J Mineral 7:1239–1249
Murad E, Cashion JD (2004) Mössbauer Spectroscopy of environmental materials and their industrial utilization. Kluwer, Boston
Naimo D, Balassone G, Beran A, Amalfitano C, Imperato M, Stanzione D (2003) Garnets in volcanic breccias of the Phlegraean Fields (southern Italy): mineralogical, geochemical and genetic features. Miner Petrol 77:259–270
Newton RC (1967) Kyanite-sillimanite equilibrium at 750°C. Science 151:1222–1225
Pattison DRM (1994) Are reversed Fe-Mg exchange and solid solution experiments really reversed? Am Mineral 79:938–950
Plakhty VP, Gukasov AG, Papoular RJ, Smirnov OP (1999) Spin density on ligands O2− and covalency of Fe3+ ions in octahedral sites of the Ca3Fe2Ge3O12 garnet: polarised neutron diffraction study. Europhys Lett 48:233–239
Pouchou JL, Pichoir F (1985) “PAP” ϕ(ρZ) procedure for improved quantitative micro-analysis. In: Armstrong JT (ed) Microbeam Analysis 1985. San Francisco Press, San Francisco, pp 104–160
Prewitt CT, Sleight AW (1969) Garnet-like structures of high-pressure cadmium germanate and calcium germanate. Science 163:386–387
Price GD, Putnis A, Agrell SO (1979) Electron petrography of shock-produced veins in the Tenham chondrite. Contrib Miner Petrol 71:211–218
Ringwood AE, Seabrook M (1963) High-pressure transformations in germanate pyroxene and related compounds. J Geophys Res 68:4601–4609
Ross N, Akaogi M, Navrotsky A, Susaki JI, McMillan P (1986) Phase transitions among the CaGeO3 polymorphs (wollastonite, garnet and perovskite structures): studies by high-pressure synthesis, high-temperature calorimetry, and vibrational spectroscopy and calculation. J Geophys Res 91:4685–4696
Smith JV, Mason B (1970) Pyroxene-garnet transformation in Coorara meteorite. Science 168:832–833
Susaki J, Akaogi M, Akimoto S, Shimomura O (1985) Garnet-perovskite transformation in CaGeO3: in-situ X-ray measurements using synchrotron radiation. Geophys Res Lett 12:729–732
Tauber A, Banks E, Kedesdy H (1958). Synthesis of germanate garnets. Acta Cryst 11:893–894
Toby BH (2001) EXPGUI, a graphical user interface for GSAS. J Appl Crystallogr 34:210–213
Tomioka N, Fujino K, Ito E, Katsura T, Sharp T, Kato T (2002) Microstructures and structural phase transition in (Mg,Fe)SiO3 majorite. Eur J Mineral 14:7–14
Ungaretti L, Leona M, Merli M, Oberti R, (1995) Non-ideal solid solution in garnet: crystal-structure evidence and modelling. Eur J Mineral 7:1299–1312
Van Cappellen E, Doukhan JC (1994) Quantitative X-ray microanalysis of ionic compounds. Ultramicroscopy 53:343–349
Vinograd V, Sluiter M, Winkler B, Putnis A, Hålenius U, Gale J, Becker U (2004) Thermodynamics of mixing and ordering in pyrope-grossular solid solution. Miner Mag 68:101–121
Wang Y, Gasparik T, Liebermann RC (1993) Modulated microstructure in synthetic majorite. Am Miner 78:1165–1173
Weidner D, Wang Y (2000) Phase transformations: implications for mantle structure. In: Karato S, Forte AM, Liebermann RC, Masters G, Stixrude L (eds) Earth’s deep interior: mineral physics and tomography from the Atomic to the global scale. Geophys Monogr 117. Amer Geophys Union, Washington DC, pp 215–235
Wildner M, Andrut M (2001) The crystal chemistry of birefringent natural uvarovites: Part II. Single crystal X-ray structures. Am Miner 86:1231–1251
Woodland AB, Ross CR II (1994) A crystallographic and Mössbauer spectroscopy study of Fe2+3Al2Si3O12–Fe2+3Fe3+2Si3O12, (almandine-”skiagite”) and Ca3Fe3+2Si3O12–Fe2+3Fe3+2Si3O12 (andradite-”skiagite”) garnet solid solutions. Phys Chem Miner 21:117–132
Acknowledgments
This project benefited through discussions with Friedrich Seifert. Geoffrey Bromiley assisted with the piston-cylinder syntheses, Detlef Krauße with collection of the electron microprobe data, Burkhardt Schmidt with analytical techniques, and Georg Hermannsdorfer with technical aspects of the high-pressure runs. The manuscript was improved through careful reviews by J. Ganguly and an anonymous reviewer. This study was supported by the Sofja Kovalevskaja Award to TBB.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iezzi, G., Boffa-Ballaran, T., McCammon, C. et al. The CaGeO3–Ca3Fe2Ge3O12 garnet join: an experimental study. Phys Chem Minerals 32, 197–207 (2005). https://doi.org/10.1007/s00269-005-0460-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00269-005-0460-0