Abstract
Background
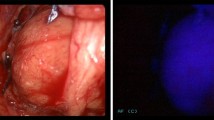
There are scant data in the literature regarding whether parathyroid autofluorescence (AF) signal patterns vary based on the etiology of hyperparathyroidism. The aim of this study was to compare AF signals of parathyroid glands across different etiologies of hyperparathyroidism.
Methods
As a prospective institutional review board-approved study between 2016 and 2019, AF intensities and heterogeneity indexes (HIs) of parathyroid glands in patients who underwent parathyroidectomy using AF were calculated and compared using Chi-square, Kruskal Wallis, Mann Whitney U, and logistic regression tests.
Results
Of the total of 183 patients, 127 patients had sporadic classic primary hyperparathyroidism, 30 patients had sporadic normohormonal primary hyperparathyroidism, 10 patients had sporadic normocalcemic primary hyperparathyroidism, 12 patients had tertiary hyperparathyroidism, and 4 patients had familial primary hyperparathyroidism related to multiple endocrine neoplasia (MEN) 2A. There were no statistical differences in AF signals of abnormal parathyroid glands in classic, normohormonal or normocalcemic sporadic hyperparathyroidism. Parathyroid glands in patients with tertiary hyperparathyroidism were similar in intensity, but more homogenous compared to those in sporadic primary hyperparathyroidism.
Conclusions
The pattern of AF exhibited by abnormal parathyroid glands was similar across different spectrums of primary hyperparathyroidism, in accordance with observations in the literature. However, parathyroid glands in tertiary hyperparathyroidism were more homogeneous, despite exhibiting a similar intensity of AF compared to those in sporadic primary hyperparathyroidism. These differences should be kept in mind when using the AF pattern as an adjunct to visual assessment in parathyroid exploration.





Similar content being viewed by others
Data availability
Data are not shared due to institutional policy.
References
Kose E, Rudin AV, Kahramangil B et al (2020) Autofluorescence imaging of parathyroid glands: an assessment of potential indications. Surgery 167:173–179
Akbulut S, Erten O, Gokceimam M et al (2021) Intraoperative near-infrared imaging of parathyroid glands: a comparison of first- and second-generation technologies. J Surg Oncol 123:866–871
Benmiloud F, Godiris-Petit G, Gras R et al (2020) Association of autofluorescence-based detection of the parathyroid glands during Total thyroidectomy with postoperative hypocalcemia risk: results of the PARAFLUO multicenter randomized clinical trial. JAMA Surg 155:106–112
Ladurner R, Lerchenberger M, Al Arabi N et al (2019) Parathyroid autofluorescence-how does it affect parathyroid and thyroid surgery? A 5 year experience. Molecules 24:2560
Kim YS, Erten O, Kahramangil B et al (2020) The impact of near infrared fluorescence imaging on parathyroid function after total thyroidectomy. J Surg Oncol 122:973–979
Serra C, Silveira L, Canudo A (2020) Identification of inadvertently removed parathyroid glands during thyroid surgery using autofluorescence. Gland Surg 9:893–898
Dip F, Falco J, Verna S et al (2019) Randomized controlled trial comparing white light with near-infrared autofluorescence for parathyroid gland identification during total thyroidectomy. J Am Coll Surg 228:744–751
Solórzano CC, Thomas G, Baregamian N et al (2020) Detecting the near infrared autofluorescence of the human parathyroid: hype or opportunity? Ann Surg 272:973–985
Akbulut S, Erten O, Kim YS et al (2021) Development of an algorithm for intraoperative autofluorescence assessment of parathyroid glands in primary hyperparathyroidism using artificial intelligence. Surgery 170:454
Kose E, Kahramangil B, Aydin H et al (2019) Heterogeneous and low-intensity parathyroid autofluorescence: patterns suggesting hyperfunction at parathyroid exploration. Surgery 165:431–437
Solórzano CC, Thomas G, Berber E et al (2021) Current state of intraoperative use of near infrared fluorescence for parathyroid identification and preservation. Surgery 169:868–878
McWade MA, Paras C, White LM et al (2013) A novel optical approach to intraoperative detection of parathyroid glands. Surgery 154:1371–1377 (discussion 1377)
McWade MA, Sanders ME, Broome JT et al (2016) Establishing the clinical utility of autofluorescence spectroscopy for parathyroid detection. Surgery 159:193–202
DiMarco A, Chotalia R, Bloxham R et al (2019) Autofluorescence in parathyroidectomy: signal intensity correlates with serum calcium and parathyroid hormone but routine clinical use is not justified. World J Surg 43:1532–1537
Takeuchi M, Takahashi T, Shodo R et al (2021) Comparison of autofluorescence with near-infrared fluorescence imaging between primary and secondary hyperparathyroidism. Laryngoscope 131:E2097-e2104
Paras C, Keller M, White L et al (2011) Near-infrared autofluorescence for the detection of parathyroid glands. J Biomed Opt 16:067012
McWade MA, Paras C, White LM et al (2014) Label-free intraoperative parathyroid localization with near-infrared autofluorescence imaging. J Clin Endocrinol Metab 99:4574–4580
Squires MH, Shirley LA, Shen C et al (2019) Intraoperative autofluorescence parathyroid identification in patients with multiple endocrine neoplasia type 1. JAMA Otolaryngol Head Neck Surg 145:897–902
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors have no conflict of interest to declare.
Ethical approval
Institutional review board-approved study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Berber, E., Akbulut, S., Avci, S. et al. Comparison of Parathyroid Autofluorescence Signals in Different Types of Hyperparathyroidism. World J Surg 46, 807–812 (2022). https://doi.org/10.1007/s00268-021-06422-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-021-06422-8