Abstract
Background
Sentinel lymph node biopsy is the gold standard for axillary assessment of patients with clinically node negative breast cancer. The current internationally accepted methods comprise of the usage of either a radioactive tracer, vital stains or the combination of both. However, in developing countries radioactive tracer is not widely used due to its high cost and limited availability. In addition, the classic retro-areolar blue dye injection has a high failure rate.
Objective
Our study aimed to assess the efficacy of patent blue dye injection in the upper outer quadrant of the breast after validation by concurrent usage of radioactive nanocolloid, in comparison with the classic retro-areolar injection in identifying the sentinel node.
Methods
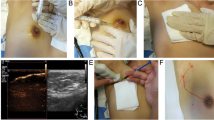
A randomized control study involving 279 patients randomly divided into two groups. In group A lateral injection of 1% patent blue dye (validated by radioactive nanocolloid) was used, while subdermal patent blue dye injection in the retro-areolar space was performed in group B.
Results
The new technique showed the promising results with lower failure rate (3.4%) in comparison with the classic retro-areolar patent blue injection (13.7%).
Conclusions
The lateral injection technique can be result in comparable results to the combination technique with the added benefit of being widely available and a cheaper option especially in developing countries.



Similar content being viewed by others
Data availability
All data and material can be obtained after author approval.
References
Chung A, Giuliano AE (2018) Lymphatic mapping and sentinel lymphadenectomy for breast cancer. Breast Compr Manag Benign Malig Dis 220(3):604-630.e6. https://doi.org/10.1016/B978-0-323-35955-9.00042-8
Chagpar A, Martin RC, Chao C et al (2004) Validation of subareolar and periareolar injection techniques for breast sentinel lymph node biopsy. Arch Surg 139(6):614–620. https://doi.org/10.1001/archsurg.139.6.614
Gray JH (1939) The relation of lymphatic vessels to the spread of cancer. Br J Surg 26(103):462–495. https://doi.org/10.1002/bjs.18002610303
Lyman GH, Giuliano AE, Somerfield MR et al (2005) American Society of Clinical Oncology guideline recommendations for sentinel lymph node biopsy in early-stage breast cancer. J Clin Oncol 23(30):7703–7720. https://doi.org/10.1200/JCO.2005.08.001
Chagpar AB, Martin RC, Scoggins CR et al (2005) Factors predicting failure to identify a sentinel lymph node in breast cancer. Surgery 138(1):56–63. https://doi.org/10.1016/j.surg.2005.03.003
Syme DBY, Collins JP, Mann GB (2005) Comparison of blue dye and isotope with blue dye alone in breast sentinel node biopsy. ANZ J Surg 75(9):817–821. https://doi.org/10.1111/j.1445-2197.2005.03531.x
Hung WK, Chan CM, Ying M, Chong SF, Mak KL, Yip AWC (2005) Randomized clinical trial comparing blue dye with combined dye and isotope for sentinel lymph node biopsy in breast cancer. Br J Surg 92(12):1494–1497. https://doi.org/10.1002/bjs.5211
Pesek S, Ashikaga T, Krag LE, Krag D (2012) The false-negative rate of sentinel node biopsy in patients with breast cancer: a meta-analysis. World J Surg 36(9):2239–2251. https://doi.org/10.1007/s00268-012-1623-z
Li J, Chen X, Qi M, Li Y (2018) Sentinel lymph node biopsy mapped with methylene blue dye alone in patients with breast cancer: A systematic review and metaanalysis. PLoS ONE 13(9):1–18. https://doi.org/10.1371/journal.pone.0204364
Siddique M, Hassan A, Nawaz M, Bashir H, Chaudhry M (2020) Comparison between sentinel lymph node hybrid scintigraphy and blue dye technique in breast cancer patients: An institutional experience. World J Nucl Med 19(1):21. https://doi.org/10.4103/wjnm.wjnm_23_19
Macdonald S, Oncology R, General M (2016) Breast Cancer Breast Cancer. J R Soc Med. 70(8):515–517
Ashikaga T, Krag DN, Land SR et al (2010) Morbidity results from the NSABP B-32 trial comparing sentinel lymph node dissection versus axillary dissection. J Surg Oncol 102(2):111–118. https://doi.org/10.1002/jso.21535
Land SR, Kopec JA, Julian TB et al (2010) Patient-reported outcomes in sentinel node-negative adjuvant breast cancer patients receiving sentinel-node biopsy or axillary dissection: National Surgical Adjuvant Breast and Bowel Project phase III protocol B-32. J Clin Oncol 28(25):3929–3936. https://doi.org/10.1200/JCO.2010.28.2491
Mansel RE, Fallowfield L, Kissin M et al (2006) Randomized multicenter trial of sentinel node biopsy versus standard axillary treatment in operable breast cancer: The ALMANAC trial. J Natl Cancer Inst 98(9):599–609. https://doi.org/10.1093/jnci/djj158
Qiu SQ, Zhang GJ, Jansen L et al (2017) Evolution in sentinel lymph node biopsy in breast cancer. Crit Rev Oncol Hematol 2018(123):83–94. https://doi.org/10.1016/j.critrevonc.2017.09.010
Wong SL, Edwards MJ, Chao C et al (2001) Sentinel lymph node biopsy for breast cancer: Impact of the number of sentinel nodes removed on the false-negative rate. J Am Coll Surg 192(6):684–689. https://doi.org/10.1016/S1072-7515(01)00858-4
Chung A, Yu J, Stempel M, Patil S, Cody H, Montgomery L (2008) Is the “10% rule” equally valid for all subsets of sentinel-node-positive breast cancer patients? Ann Surg Oncol 15(10):2728–2733. https://doi.org/10.1245/s10434-008-0050-8
Jakub JW, Cox CE, Pippas AW, Gardner M, Pendas S, Reintgen DS (2004) Controversial topics in breast lymphatic mapping. Semin Oncol 31(3):324–332. https://doi.org/10.1053/j.seminoncol.2004.03.014
Woznick A, Franco M, Bendick P, Benitez PR (2006) Sentinel lymph node dissection for breast cancer: How many nodes are enough and which technique is optimal? Am J Surg 191(3):330–333. https://doi.org/10.1016/j.amjsurg.2005.10.031
Ban EJ, Lee JS, Koo JS, Park S, Il Kim S, Park BW (2011) How many sentinel lymph nodes are enough for accurate axillary staging in T1–2 breast cancer? J Breast Cancer 14(4):296–300
Cody HS (2001) Clinical aspects of sentinel node biopsy. Breast Cancer Res 3(2):104–108. https://doi.org/10.1186/bcr280
Guo J, Yang H, Wang S et al (2017) Comparison of sentinel lymph node biopsy guided by indocyanine green, blue dye, and their combination in breast cancer patients: a prospective cohort study. World J Surg Oncol 15(1):196
Gupta V, Raju K, Rao TS et al (2020) A Randomized Trial Comparing the Efficacy of Methylene Blue Dye Alone Versus Combination of Methylene Blue Dye and Radioactive Sulfur Colloid in Sentinel Lymph Node Biopsy for Early Stage Breast Cancer Patients. Indian J Surg Oncol 11(2):216–222. https://doi.org/10.1007/s13193-019-01023-3
Shen S, Xu Q, Zhou Y, Mao F, Guan J, Sun Q (2018) Comparison of sentinel lymph node biopsy guided by blue dye with or without indocyanine green in early breast cancer. J Surg Oncol 117(8):1841–1847. https://doi.org/10.1002/jso.25058
Brahma B, Putri RI, Sari L et al (2019) The Application of 1% Methylene Blue Dye As a Single Technique in Breast Cancer Sentinel Node Biopsy. J Vis Exp. https://doi.org/10.3791/57201
He PS, Li F, Li GH, Guo C, Chen TJ (2016) The combination of blue dye and radioisotope versus radioisotope alone during sentinel lymph node biopsy for breast cancer: a systematic review. BMC Cancer. https://doi.org/10.1186/s12885-016-2137-0
Goyal A (2018) New Technologies for Sentinel Lymph Node Detection. Breast Care (Basel) 13(5):349–353. https://doi.org/10.1159/000492436
Mok CW, Tan SM, Zheng Q, Shi L (2019) Network meta-analysis of novel and conventional sentinel lymph node biopsy techniques in breast cancer. BJS Open. 3(4):445–452
Krag DN, Anderson SJ, Julian TB et al (2007) Technical outcomes of sentinel-lymph-node resection and conventional axillary-lymph-node dissection in patients with clinically node-negative breast cancer: results from the NSABP B-32 randomised phase III trial. Lancet Oncol 8(10):881–888. https://doi.org/10.1016/S1470-2045(07)70278-4
Nowikiewicz T, Głowacka-Mrotek I, Tarkowska M, Nowikiewicz M, Zegarski W (2020) Failure of sentinel lymph node mapping in breast cancer patients qualified for treatment sparing axillary lymph nodes—Clinical importance and management strategy—One-center analysis. Breast J 26(5):873–881. https://doi.org/10.1111/tbj.13769
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors indicate that they do not have conflicts of interest.
Ethics approval
Approval of Ethical committee and institutional review board were obtained.
Consent for publication
All authors approved for publishing the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fouad, M.M.B., Fouad, K. & Ibraheim, S.M.N. Assessment of Intraparenchymal Injection of 1% Patent Blue Dye in the Upper Outer Quadrant of the Breast to Identify Sentinel Lymph Node in Early Retro-Areolar Breast Cancer in Women: A Tertiary Centre Experience in Egypt. World J Surg 46, 829–835 (2022). https://doi.org/10.1007/s00268-021-06421-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-021-06421-9