Abstract
Background
Immunotherapy advances for the treatment of cutaneous melanoma question its efficacy in treating anorectal mucosal melanoma (ARMM). We aimed to identify the prevalence, current management, and overall survival (OS) for ARMM.
Methods
Review of patients with ARMM from 2004 to 2015 National Cancer Database. Factors associated with immunotherapy were identified using multivariable logistic regression. The primary outcome was 2- and 5-year OS. Subgroup analysis by treatment type was performed.
Results
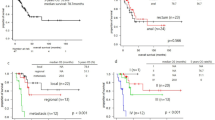
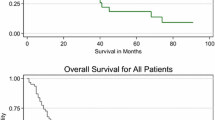
A total of 1331 patients were identified with a significant increase in prevalence (2004: 6.99%, 2015: 10.53%). ARMM patients were older, white, on Medicare, and from the South. The most common treatment was surgery (48.77%), followed by surgery + radiation (11.75%), surgery + immunotherapy (8.68%), and surgery + chemotherapy (8.68%). 16.93% of patients received immunotherapy, with utilization increasing (7.24%: 2004, 21.27%: 2015, p < 0.001). Patients who received immunotherapy had a significantly better 2-year OS (42.47% vs. 49.21%, p < 0.001), and other therapies did not reveal a significant difference. Adjusted analysis showed no difference in 2- and 5-year OS based on therapy type.
Conclusion
The prevalence of ARMM has increased. The use of immunotherapy has increased substantially. Some survival benefit with the administration of immunotherapy may exist that has yet to be revealed. A more aggressive treatment paradigm is warranted.






Similar content being viewed by others
References
Chang AE, Karnell LH, Menck HR (1998) The National Cancer Data Base report on cutaneous and noncutaneous melanoma: a summary of 84,836 cases from the past decade. The American College of Surgeons Commission on Cancer and the American Cancer Society. Cancer 83:1664–1678
Bullard KM, Tuttle TM, Rothenberger DA, Madoff RD, Baxter NN, Finne CO, Spencer MP (2003) Surgical therapy for anorectal melanoma. J Am Coll Surg 196:206–211
Meguerditchian AN, Meterissian SH, Dunn KB (2011) Anorectal melanoma: diagnosis and treatment. Dis Colon Rectum 54:638–644
Moozar KL, Wong CS, Couture J (2003) Anorectal malignant melanoma: treatment with surgery or radiation therapy, or both. Can J Surg 46:345–349
Row D, Weiser MR (2009) Anorectal melanoma. Clin Colon Rectal Surg 22:120–126
Brady MS, Kavolius JP, Quan SH (1995) Anorectal melanoma. A 64-year experience at Memorial Sloan-Kettering Cancer Center. Dis Colon Rectum 38:146–151
Yeh JJ, Shia J, Hwu WJ, Busam KJ, Paty PB, Guillem JG, Coit DG, Wong WD, Weiser MR (2006) The role of abdominoperineal resection as surgical therapy for anorectal melanoma. Ann Surg 244:1012–1017
Yeh JJ, Weiser MR, Shia J, Hwu WJ (2005) Response of stage IV anal mucosal melanoma to chemotherapy. Lancet Oncol 6:438–439
Hillenbrand A, Barth TF, Henne-Bruns D, Formentini A (2008) Anorectal amelanotic melanoma. Colorectal Dis 10:612–615
McLaughlin CC, Wu XC, Jemal A, Martin HJ, Roche LM, Chen VW (2005) Incidence of noncutaneous melanomas in the US. Cancer 103:1000–1007
Carvajal RD, Antonescu CR, Wolchok JD, Chapman PB, Roman RA, Teitcher J, Panageas KS, Busam KJ, Chmielowski B, Lutzky J, Pavlick AC, Fusco A, Cane L, Takebe N, Vemula S, Bouvier N, Bastian BC, Schwartz GK (2011) KIT as a therapeutic target in metastatic melanoma. JAMA 305:2327–2334
Curtin JA, Fridlyand J, Kageshita T, Patel HN, Busam KJ, Kutzner H, Cho KH, Aiba S, Brocker EB, LeBoit PE, Pinkel D, Bastian BC (2005) Distinct sets of genetic alterations in melanoma. N Engl J Med 353:2135–2147
Carvajal RD, Spencer SA, Lydiatt W (2012) Mucosal melanoma: a clinically and biologically unique disease entity. J Natl Compr Cancer Netw 10:345–356
Callahan A, Anderson WF, Patel S, Barnholtz-Sloan JS, Bordeaux JS, Tucker MA, Gerstenblith MR (2016) Epidemiology of anorectal melanoma in the United States: 1992 to 2011. Dermatol Surg 42:94–99
Kiran RP, Rottoli M, Pokala N, Fazio VW (2010) Long-term outcomes after local excision and radical surgery for anal melanoma: data from a population database. Dis Colon Rectum 53:402–408
Pessaux P, Pocard M, Elias D, Duvillard P, Avril MF, Zimmerman P, Lasser P (2004) Surgical management of primary anorectal melanoma. Br J Surg 91:1183–1187
Ciarrocchi A, Pietroletti R, Carlei F, Amicucci G (2017) Extensive surgery and lymphadenectomy do not improve survival in primary melanoma of the anorectum: results from analysis of a large database (SEER). Colorectal Dis 19:158–164
Tchelebi L, Guirguis A, Ashamalla H (2016) Rectal melanoma: epidemiology, prognosis, and role of adjuvant radiation therapy. J Cancer Res Clin Oncol 142:2569–2575
Sadozai H, Gruber T, Hunger RE, Schenk M (2017) Recent successes and future directions in immunotherapy of cutaneous melanoma. Front Immunol 8:1617
(2017) National Cancer Database. https://www.facs.org/quality-programs/cancer/ncdb
(2017) Census regions and divisions of the United states. https://www2.census.gov/geo/pdfs/maps-data/maps/reference/us_regdiv.pdf
Hosmer DW Jr, Lemeshow S (2000) Applied logistic regression, 2nd edn. Wiley, New York
Weinstock MA (1993) Epidemiology and prognosis of anorectal melanoma. Gastroenterology 104:174–178
Cote TR, Sobin LH (2009) Primary melanomas of the esophagus and anorectum: epidemiologic comparison with melanoma of the skin. Melanoma Res 19:58–60
Micu E, Juzeniene A, Moan J (2011) Comparison of the time and latitude trends of melanoma incidence in anorectal region and perianal skin with those of cutaneous malignant melanoma in Norway. J Eur Acad Dermatol Venereol 25:1444–1449
Iddings DM, Fleisig AJ, Chen SL, Faries MB, Morton DL (2010) Practice patterns and outcomes for anorectal melanoma in the USA, reviewing three decades of treatment: is more extensive surgical resection beneficial in all patients? Ann Surg Oncol 17:40–44
Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, Gonzalez R, Robert C, Schadendorf D, Hassel JC, Akerley W, van den Eertwegh AJ, Lutzky J, Lorigan P, Vaubel JM, Linette GP, Hogg D, Ottensmeier CH, Lebbe C, Peschel C, Quirt I, Clark JI, Wolchok JD, Weber JS, Tian J, Yellin MJ, Nichol GM, Hoos A, Urba WJ (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363:711–723
Ku GY, Yuan J, Page DB, Schroeder SE, Panageas KS, Carvajal RD, Chapman PB, Schwartz GK, Allison JP, Wolchok JD (2010) Single-institution experience with ipilimumab in advanced melanoma patients in the compassionate use setting: lymphocyte count after 2 doses correlates with survival. Cancer 116:1767–1775
Gorka E, Fabo D, Gezsi A, Czirbesz K, Fedorcsak I, Liszkay G (2018) Dabrafenib therapy in 30 patients with melanoma metastatic to the brain: a single-centre controlled retrospective study in Hungary. Pathol Oncol Res 24:401–406
Kim KB, Eton O, Davis DW, Frazier ML, McConkey DJ, Diwan AH, Papadopoulos NE, Bedikian AY, Camacho LH, Ross MI, Cormier JN, Gershenwald JE, Lee JE, Mansfield PF, Billings LA, Ng CS, Charnsangavej C, Bar-Eli M, Johnson MM, Murgo AJ, Prieto VG (2008) Phase II trial of imatinib mesylate in patients with metastatic melanoma. Br J Cancer 99:734–740
Ugurel S, Hildenbrand R, Zimpfer A, La Rosee P, Paschka P, Sucker A, Keikavoussi P, Becker JC, Rittgen W, Hochhaus A, Schadendorf D (2005) Lack of clinical efficacy of imatinib in metastatic melanoma. Br J Cancer 92:1398–1405
Hodi FS, Friedlander P, Corless CL, Heinrich MC, Mac Rae S, Kruse A, Jagannathan J, Van den Abbeele AD, Velazquez EF, Demetri GD, Fisher DE (2008) Major response to imatinib mesylate in KIT-mutated melanoma. J Clin Oncol 26:2046–2051
Lutzky J, Bauer J, Bastian BC (2008) Dose-dependent, complete response to imatinib of a metastatic mucosal melanoma with a K642E KIT mutation. Pigment Cell Melanoma Res 21:492–493
Kluger HM, Dudek AZ, McCann C, Ritacco J, Southard N, Jilaveanu LB, Molinaro A, Sznol M (2011) A phase 2 trial of dasatinib in advanced melanoma. Cancer 117:2202–2208
Woodman SE, Trent JC, Stemke-Hale K, Lazar AJ, Pricl S, Pavan GM, Fermeglia M, Gopal YN, Yang D, Podoloff DA, Ivan D, Kim KB, Papadopoulos N, Hwu P, Mills GB, Davies MA (2009) Activity of dasatinib against L576P KIT mutant melanoma: molecular, cellular, and clinical correlates. Mol Cancer Ther 8:2079–2085
Zhao J, Zhu Y, Zhang C, Wang X, He H, Wang H, Wu Y, Zhou W, Shen Z (2013) Sorafenib or sunitinib as postoperative adjuvant therapy for Chinese patients with locally advanced clear cell renal cell carcinoma at high risk for disease recurrence. Urol Oncol 31:1800–1805
Quintas-Cardama A, Lazar AJ, Woodman SE, Kim K, Ross M, Hwu P (2008) Complete response of stage IV anal mucosal melanoma expressing KIT Val560Asp to the multikinase inhibitor sorafenib. Nat Clin Pract Oncol 5:737–740
Min L, Hodi FS (2014) Anti-PD1 following ipilimumab for mucosal melanoma: durable tumor response associated with severe hypothyroidism and rhabdomyolysis. Cancer Immunol Res 2:15–18
Tokuhara K, Nakatani K, Tanimura H, Yoshioka K, Kiyohara T, Kon M (2017) A first reported case of metastatic anorectal amelanotic melanoma with a marked response to anti-PD-1 antibody nivolumab: a case report. Int J Surg Case Rep 31:188–192
Faure M, Rochigneux P, Olive D, Taix S, Brenot-Rossi I, Gilabert M (2018) Hyperprogressive disease in anorectal melanoma treated by PD-1 inhibitors. Front Immunol 9:797
Malaguarnera G, Madeddu R, Catania VE, Bertino G, Morelli L, Perrotta RE, Drago F, Malaguarnera M, Latteri S (2018) Anorectal mucosal melanoma. Oncotarget 9:8785–8800
Kirchoff DD, Deutsch GB, Foshag LJ, Lee JH, Sim MS, Faries MB (2016) Evolving therapeutic strategies in mucosal melanoma have not improved survival over five decades. Am Surg 82:1–5
MacCallum C, Skandarajah A, Gibbs P, Hayes I (2018) The value of clinical colorectal cancer registries in colorectal cancer research: a systematic review. JAMA Surg 153:841–849
Funding
Mr. Edwin Lewis provided generous support to Dr. Efron’s Department of Surgery Research Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors of this paper have any conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Disclaimers: The National Cancer Data Base (NCDB) is a joint project of the Commission on Cancer (CoC) of the American College of Surgeons and the American Cancer Society. The CoC’s NCDB and the hospitals participating in the CoC NCDB are the source of the de-identified data used herein; they have not verified and are not responsible for the statistical validity of the data analysis or the conclusions derived by the authors.
Rights and permissions
About this article
Cite this article
Taylor, J.P., Stem, M., Yu, D. et al. Treatment Strategies and Survival Trends for Anorectal Melanoma: Is it Time for a Change?. World J Surg 43, 1809–1819 (2019). https://doi.org/10.1007/s00268-019-04960-w
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-019-04960-w