Abstract
Background
Identifying novel predictors of hematogenous metastasis is essential to select optimal therapeutic strategies for pancreatic cancer. The goal of this study was to clarify the clinical implications of p53 and platelet-derived growth factor receptor-β (PDGFR-β) in pancreatic cancer using immunohistochemistry (IHC).
Methods
Specimens obtained by surgical resection and endoscopic ultrasound-guided fine-needle aspiration (EUS–FNA) of the primary tumor were used for IHC. EUS–FNA was performed before any treatment. We analyzed the clinical implications of p53 and PDGFR-β IHC. We also performed a comparative analysis of the IHC findings between surgical and EUS–FNA specimens.
Results
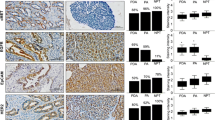
This study involved 149 patients (120 who did not receive and 29 who received preoperative therapy). EUS–FNA specimens were obtained from 20 patients without preoperative therapy and 20 patients with preoperative therapy. Abnormal p53 and high PDGFR-β expression was significantly correlated with a high incidence of hematogenous metastasis (p < 0.001) and poor postoperative survival (p = 0.006). In patients without preoperative therapy, the results for p53 and PDGFR-β staining were concordant between surgical and EUS–FNA specimens in 95 % of the cases. In patients with preoperative therapy, 80 and 70 % of the patients showed consistent p53 and PDGFR-β expression between surgical and EUS–FNA specimens, respectively.
Conclusions
Pancreatic cancer patients with simultaneous abnormal p53 and high PDGFR-β expression have a significantly higher risk of hematogenous metastasis and a poor prognosis after surgery. IHC evaluation using EUS–FNA specimens may be able to identify patients with highly metastatic pancreatic cancer who may benefit from aggressive therapeutic intervention.



Similar content being viewed by others
References
Vincent A, Herman J, Schulick R et al (2011) Pancreatic cancer. Lancet 378:607–620
Ying JE, Zhu LM, Liu BX (2012) Developments in metastatic pancreatic cancer: is gemcitabine still the standard? World J Gastroenterol 18:736–745
Cameron JL, Riall TS, Coleman J et al (2006) One thousand consecutive pancreaticoduodenectomies. Ann Surg 244:10–15
Yachida S, Iacobuzio-Donahue CA (2009) The pathology and genetics of metastatic pancreatic cancer. Arch Pathol Lab Med 133:413–422
Rose JB, Rocha FG, Alseidi A et al (2014) Extended neoadjuvant chemotherapy for borderline resectable pancreatic cancer demonstrates promising postoperative outcomes and survival. Ann Surg Oncol 21:1530–1537
Ferrone CR, Marchegiani G, Hong TS et al (2015) Radiological and surgical implications of neoadjuvant treatment with FOLFIRINOX for locally advanced and borderline resectable pancreatic cancer. Ann Surg 261:12–17
Jones S, Zhang X, Parsons DW et al (2008) Core signaling pathways in human pancreatic cancers revealed by global genomic analyses. Science 321:1801–1806
Yachida S, Iacobuzio-Donahue CA (2013) Evolution and dynamics of pancreatic cancer progression. Oncogene 32:5253–5260
Obata A, Eura M, Sasaki J et al (2000) Clinical significance of p53 functional loss in squamous cell carcinoma of the oropharynx. Int J Cancer 89:187–193
Freed-Pastor WA, Mizuno H, Zhao X et al (2012) Mutant p53 disrupts mammary tissue architecture via the mevalonate pathway. Cell 148:244–258
Soussi T, Beroud C (2001) Assessing TP53 status in human tumours to evaluate clinical outcome. Nat Rev Cancer 1:233–240
Oshima M, Okano K, Muraki S et al (2013) Immunohistochemically detected expression of 3 major genes (CDKN2A/p16, TP53, and SMAD4/DPC4) strongly predicts survival in patients with resectable pancreatic cancer. Ann Surg 258:336–346
Shin SH, Kim SC, Hong SM et al (2013) Genetic alterations of K-ras, p53, c-erbB-2, and DPC4 in pancreatic ductal adenocarcinoma and their correlation with patient survival. Pancreas 42:216–222
Kim H, Saka B, Knight S et al (2014) Having pancreatic cancer with tumoral loss of ATM and normal TP53 protein expression is associated with a poorer prognosis. Clin Cancer Res 20:1865–1872
Nenutil R, Smardova J, Pavlova S et al (2005) Discriminating functional and non-functional p53 in human tumours by p53 and MDM2 immunohistochemistry. J Pathol 207:251–259
Pietras K, Sjoblom T, Rubin K et al (2003) PDGF receptors as cancer drug targets. Cancer Cell 3:439–443
Cao R, Bjorndahl MA, Religa P et al (2004) PDGF-BB induces intratumoral lymphangiogenesis and promotes lymphatic metastasis. Cancer Cell 6:333–345
Kodama M, Kitadai Y, Sumida T et al (2010) Expression of platelet-derived growth factor (PDGF)-B and PDGF-receptor beta is associated with lymphatic metastasis in human gastric carcinoma. Cancer Sci 101:1984–1989
Ehnman M, Missiaglia E, Folestad E et al (2013) Distinct effects of ligand-induced PDGFRalpha and PDGFRbeta signaling in the human rhabdomyosarcoma tumor cell and stroma cell compartments. Cancer Res 73:2139–2149
Hwang RF, Yokoi K, Bucana CD et al (2003) Inhibition of platelet-derived growth factor receptor phosphorylation by STI571 (Gleevec) reduces growth and metastasis of human pancreatic carcinoma in an orthotopic nude mouse model. Clin Cancer Res 9:6534–6544
Singh PK, Wen Y, Swanson BJ et al (2007) Platelet-derived growth factor receptor beta-mediated phosphorylation of MUC1 enhances invasiveness in pancreatic adenocarcinoma cells. Cancer Res 67:5201–5210
Tsao AS, Wei W, Kuhn E et al (2011) Immunohistochemical overexpression of platelet-derived growth factor receptor-beta (PDGFR-beta) is associated with PDGFRB gene copy number gain in sarcomatoid non-small-cell lung cancer. Clin Lung Cancer 12:369–374
Weissmueller S, Manchado E, Saborowski M et al (2014) Mutant p53 drives pancreatic cancer metastasis through cell-autonomous PDGF receptor beta signaling. Cell 157:382–394
Sobin LH, Gospodarowicz MK, Wittekind C et al (2010) TNM classification of malignant tumours, 7th edn. Wiley-Blackwell, Hoboken
Juliachs M, Munoz C, Moutinho CA et al (2014) The PDGFRbeta-AKT pathway contributes to CDDP-acquired resistance in testicular germ cell tumors. Clin Cancer Res 20:658–667
Rhim AD, Mirek ET, Aiello NM et al (2012) EMT and dissemination precede pancreatic tumor formation. Cell 148:349–361
Chen H, Sun Y, Wu C et al (2012) Pathogenesis of prostatic small cell carcinoma involves the inactivation of the P53 pathway. Endocr Relat Cancer 19:321–331
Gao L, Antic T, Hyjek E et al (2013) Immunohistochemical analysis of E-cadherin and zeste homolog 2 expression in endoscopic ultrasound-guided fine-needle aspiration of pancreatic adenocarcinoma. Cancer Cytopathol 121:644–652
Boone BA, Sabbaghian S, Zenati M et al (2014) Loss of SMAD4 staining in pre-operative cell blocks is associated with distant metastases following pancreaticoduodenectomy with venous resection for pancreatic cancer. J Surg Oncol 110:171–175
Higashi M, Yokoyama S, Yamamoto T et al (2015) Mucin Expression in endoscopic ultrasound-guided fine-needle aspiration specimens is a useful prognostic factor in pancreatic ductal adenocarcinoma. Pancreas 44:728–734
Preis M, Gardner TB, Gordon SR et al (2011) MicroRNA-10b expression correlates with response to neoadjuvant therapy and survival in pancreatic ductal adenocarcinoma. Clin Cancer Res 17:5812–5821
Funding
This study was funded by Grand-in-Aid for Scientific Research (26462067) from the Japan Society for the Promotion of Science, Ministry of Health, Labour and Welfare, Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Rights and permissions
About this article
Cite this article
Kurahara, H., Maemura, K., Mataki, Y. et al. Impact of p53 and PDGFR-β Expression on Metastasis and Prognosis of Patients with Pancreatic Cancer. World J Surg 40, 1977–1984 (2016). https://doi.org/10.1007/s00268-016-3477-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-016-3477-2