Abstract
Background
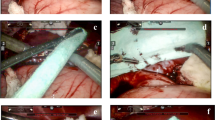
For patients undergoing liver resection that leaves an empty intraparenchymal cavity, traditional topical agents might be inadequate to achieve additional hemostasis. A new hemostatic expanding topical foam (BioFoam®) has been designed to provide a mechanical seal. The objective of this study was to report our preliminary results regarding the safety and the efficacy using this foam.
Methods
Between 2009 and 2011, BioFoam® was used to fill a three-dimensional defect following liver resection in 14 patients. The operative results and postoperative course of these patients were compared to those of 14 matched controls who underwent liver resection but did not receive BioFoam®.
Results
The two groups were similar in terms of demographics, indications for liver resection, type of surgical procedure, and type and duration of clamping. BioFoam® patients experienced significantly less operative blood loss (275 vs. 630 ml, p = 0.032) but similar operative transfusion rates (28.6 vs. 35.7 %, p = 0.686) compared to no-BioFoam® patients. The postoperative mortality was nil and no patient developed postoperative hemorrhage. While the two groups shared similar overall (64.3 vs. 57.1 %, p = 0.599) and major (28.6 vs. 14.3 %, p = 0.357) complications rates, BioFoam® patients experienced significantly higher major vascular thrombosis compared to no-BioFoam® patients (29 vs. 0 %, p = 0.04). In the BioFoam® group, major vascular thrombosis was associated with exposure of the vessel along the transection plane.
Conclusion
While the clinical benefit of BioFoam® in high-risk liver resections leaving a deep parenchymal defect remains to be proven, the associated risk of vascular thrombosis should preclude its use in contact with major veins.



Similar content being viewed by others
Abbreviations
- IVC:
-
Inferior vena cava
- MHV:
-
Middle hepatic vein
- CLM:
-
Colorectal liver metastases
- LCA:
-
Liver cell adenoma
References
Rahbari NN, Garden OJ, Padbury R et al (2011) Post-hepatectomy haemorrhage: a definition and grading by the International Study Group of Liver Surgery (ISGLS). HPB (Oxford) 13:528–535
Miura F, Asano T, Amano H et al (2009) Management of postoperative arterial hemorrhage after pancreato-biliary surgery according to the site of bleeding: re-laparotomy or interventional radiology. J Hepatobiliary Pancreat Surg 16:56–63
Lesurtel M, Selzner M, Petrowsky H et al (2005) How should transection of the liver be performed?: a prospective randomized study in 100 consecutive patients: comparing four different transection strategies. Ann Surg 242:814–822
Chavez-Tapia NC, Alfaro-Lara R, Tellez-Avila F et al (2011) Prophylactic activated recombinant factor VII in liver resection and liver transplantation: systematic review and meta-analysis. PLoS One 6:e22581
Schwartz M, Madariaga J, Hirose R et al (2004) Comparison of a new fibrin sealant with standard topical hemostatic agents. Arch Surg 139:1148–1154
Frilling A, Stavrou GA, Mischinger HJ et al (2005) Effectiveness of a new carrier-bound fibrin sealant versus argon beamer as haemostatic agent during liver resection: a randomised prospective trial. Langenbecks Arch Surg 390:114–120
Briceño J, Naranjo A, Ciria R et al (2010) A prospective study of the efficacy of clinical application of a new carrier-bound fibrin sealant after liver resection. Arch Surg 145:482–488
Fischer L, Seiler CM, Broelsch CE et al (2011) Hemostatic efficacy of TachoSil in liver resection compared with argon beam coagulator treatment: an open, randomized, prospective, multicenter, parallel-group trial. Surgery 149:48–55
Figueras J, Llado L, Miro M et al (2007) Application of fibrin glue sealant after hepatectomy does not seem justified: results of a randomized study in 300 patients. Ann Surg 245:536–542
Dokmak S, Ftériche FS, Borscheid R et al (2013) Liver resections in the 21st century: we are far from zero mortality. HPB (Oxford). doi:10.1111/hpb.12069
Belghiti J, Noun R, Malafosse R et al (1999) Continuous versus intermittent portal triad clamping for liver resection: a controlled study. Ann Surg 229:369–375
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6,336 patients and results of a survey. Ann Surg 240:205–213
Koch M, Garden OJ, Padbury R et al (2011) Bile leakage after hepatobiliary and pancreatic surgery: a definition and grading of severity by the International Study Group of Liver Surgery. Surgery 149:680–688
Balzan S, Belghiti J, Farges O et al (2005) The “50–50 criteria” on postoperative day 5: an accurate predictor of liver failure and death after hepatectomy. Ann Surg 242:824–828
Ishizawa T, Hasegawa K, Kokudo N et al (2009) Risk factors and management of ascites after liver resection to treat hepatocellular carcinoma. Arch Surg 144:46–51
Buc E, Dokmak S, Zappa M et al (2011) Hepatic veins as a site of clot formation following liver resection. World J Gastroenterol 17:403–406
Melloul E, Dondéro F, Vilgrain V et al (2012) Pulmonary embolism after elective liver resection: a prospective analysis of risk factors. J Hepatol 57:1268–1275
Jones RM, Moulton CE, Hardy KJ (1998) Central venous pressure and its effect on blood loss during liver resection. Br J Surg 85:1058–1060
Conflict of interest
None of the authors have any conflict of interest of any kind. None of the authors received any support from the BIOFOAM manufacturers. The BIOFOAM manufacturers did not support this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cauchy, F., Gaujoux, S., Ronot, M. et al. Local Venous Thrombotic Risk of an Expanding Haemostatic Agent Used During Liver Resection. World J Surg 38, 2363–2369 (2014). https://doi.org/10.1007/s00268-014-2552-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-014-2552-9