Abstract
Background
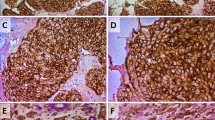
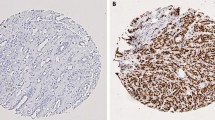
The purpose of present study was to examine the expression of cancer stem cell marker Bmi-1 in breast cancer tissue and to evaluate the clinical implication of Bmi-1 expression for these patients.
Methods
A total of 171 breast cancer patients who received surgical treatment in our hospital were enrolled in this study. Bmi-1 expression in breast cancer tissue was assayed by immunohistochemistry. Statistical analyses were applied to test the relationship between expression of Bmi-1 and clinicopathologic features and patient survival. The relationship between Bmi-1 and the basal-like phenotype of breast cancer also was analyzed in this study.
Results
Positive Bmi-1 expression was detected in 89 of 171 (52%) invasive breast cancers patients. The Bmi-1 status was significantly correlated to histological grade III (p = 0.001) and basal-like phenotype (p < 0.001). The 5 year overall survival of the patients with Bmi-1-positive and -negative cancers were 78 and 91.9%, respectively (p = 0.03). Histological grade (p = 0.046) and Bmi-1 status (p = 0.012) were detected as the independent prognostic factors in the Cox regression test.
Conclusions
Bmi-1 status is an independent prognostic factor, which also is associated with tumor histological grade and basal-like phenotype. The high proportions of positive Bmi-1 expression in basal-like breast cancer may be related to the high aggressiveness behavior of this subtype of breast cancer.



Similar content being viewed by others
References
Perou CM, Sorlie T, Eisen MB et al (2000) Molecular portraits of human breast tumours. Nature 406:747–752
Sorlie T, Perou CM, Tibshirani R et al (2001) Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 98:10869–10874
Sorlie T, Tibshirani R, Parker J et al (2003) Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc Natl Acad Sci USA 100:8418–8423
Sotiriou C, Neo SY, McShane LM et al (2003) Breast cancer classification and prognosis based on gene expression profiles from a population-based study. Proc Natl Acad Sci U S A 100:10393–10398
Shipitsin M, Campbell LL, Argani P et al (2007) Molecular definition of breast tumor heterogeneity. Cancer Cell 11:259–273
Rakha EA, Reis-Filho JS, Ellis IO (2008) Basal-like breast cancer: a critical review. J Clin Oncol 26:2568–2581
Badve S, Dabbs DJ, Schnitt SJ et al (2011) Basal-like and triple-negative breast cancers: a critical review with an emphasis on the implications for pathologists and oncologists. Mod Pathol 24:157–167
Haupt Y, Alexander WS, Barri G et al (1991) Novel zinc finger gene implicated as myc collaborator by retrovirally accelerated lymphomagenesis in E mu-myc transgenic mice. Cell 65:753–763
van Lohuizen M, Verbeek S, Scheijen B et al (1991) Identification of cooperating oncogenes in E mu-myc transgenic mice by provirus tagging. Cell 65:737–752
van der Lugt NM, Domen J, Linders K et al (1994) Posterior transformation, neurological abnormalities, and severe hematopoietic defects in mice with a targeted deletion of the bmi-1 proto-oncogene. Genes Dev 8:757–769
Pirrotta V (1998) Polycombing the genome: PcG, trxG, and chromatin silencing. Cell 93:333–336
Lessard J, Sauvageau G (2003) Bmi-1 determines the proliferative capacity of normal and leukaemic stem cells. Nature 423:255–260
Liu S, Dontu G, Mantle ID et al (2006) Hedgehog signaling and Bmi-1 regulate self-renewal of normal and malignant human mammary stem cells. Cancer Res 66:6063–6071
Jiang L, Li J, Song L (2009) Bmi-1, stem cells and cancer. Acta Biochim Biophys Sin (Shanghai) 41:527–534
Kim JH, Yoon SY, Jeong SH et al (2004) Overexpression of Bmi-1 oncoprotein correlates with axillary lymph node metastases in invasive ductal breast cancer. Breast 13:383–388
Feng Y, Song LB, Guo BH et al (2007) Expression and significance of Bmi-1 in breast cancer. Ai Zheng 26:154–157
Arnes JB, Collett K, Akslen LA (2008) Independent prognostic value of the basal-like phenotype of breast cancer and associations with EGFR and candidate stem cell marker BMI-1. Histopathology 52:370–380
Choi YJ, Choi YL, Cho EY et al (2009) Expression of Bmi-1 protein in tumor tissues is associated with favorable prognosis in breast cancer patients. Breast Cancer Res Treat 113:83–93
Honig A, Weidler C, Häusler S et al (2010) Overexpression of polycomb protein BMI-1 in human specimens of breast, ovarian, endometrial and cervical cancer. Anticancer Res 30:1559–1564
Riis ML, Lüders T, Nesbakken AJ et al (2010) Expression of BMI-1 and Mel-18 in breast tissue: a diagnostic marker in patients with breast cancer. BMC Cancer 10:686
Guo BH, Feng Y, Zhang R et al (2011) Bmi-1 promotes invasion and metastasis, and its elevated expression is correlated with an advanced stage of breast cancer. Mol Cancer 10:10
Joensuu K, Hagström J, Leidenius M et al (2011) Bmi-1, c-myc, and Snail expression in primary breast cancers and their metastases–elevated Bmi-1 expression in late breast cancer relapses. Virchows Arch 459:31–39
Vonlanthen S, Heighway J, Altermatt HJ et al (2001) The bmi-1 oncoprotein is differentially expressed in non-small cell lung cancer and correlates with INK4A-ARF locus expression. Br J Cancer 84:1372–1376
Kim JH, Yoon SY, Kim CN et al (2004) The Bmi-1 oncoprotein is overexpressed in human colorectal cancer and correlates with the reduced p16INK4a/p14ARF proteins. Cancer Lett 203:217–224
Song W, Tao K, Li H et al (2010) Bmi-1 is related to proliferation, survival and poor prognosis in pancreatic cancer. Cancer Sci 101:1754–1760
Zhang X, Wang CX, Zhu CB et al (2010) Overexpression of Bmi-1 in uterine cervical cancer: correlation with clinicopathology and prognosis. Int J Gynecol Cancer 20:1597–1603
Nielsen TO, Hsu FD, Jensen K et al (2004) Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin Cancer Res 10:5367–5374
Campbell LL, Polyak K (2007) Breast tumor heterogeneity: cancer stem cells or clonal evolution? Cell Cycle 6:2332–2338
Shackleton M, Quintana E, Fearon ER et al (2009) Heterogeneity in cancer: cancer stem cells versus clonal evolution. Cell 138:822–829
Polyak K, Hahn WC (2006) Roots and stems: stem cells in cancer. Nat Med 12:296–300
Visvader JE, Lindeman GJ (2008) Cancer stem cells in solid tumours: accumulating evidence and unresolved questions. Nat Rev Cancer 8:755–768
Rosen JM, Jordan CT (2009) The increasing complexity of the cancer stem cell paradigm. Science 324:1670–1673
Pece S, Tosoni D, Confalonieri S et al (2010) Biological and molecular heterogeneity of breast cancers correlates with their cancer stem cell content. Cell 140:62–73
Park SY, Lee HE, Li H et al (2010) Heterogeneity for stem cell-related markers according to tumor subtype and histologic stage in breast cancer. Clin Cancer Res 16:876–887
Kakarala M, Wicha MS et al (2008) Implications of the cancer stem-cell hypothesis for breast cancer prevention and therapy. J Clin Oncol 26:2813–2820
Acknowledgments
This work was supported by a grant of Ningxia Medical University (XT200916).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Y., Zhe, H., Ding, Z. et al. Cancer Stem Cell Marker Bmi-1 Expression is Associated with Basal-like Phenotype and Poor Survival in Breast Cancer. World J Surg 36, 1189–1194 (2012). https://doi.org/10.1007/s00268-012-1514-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-012-1514-3