Abstract
Background
Operative resection is the only curative treatment for pheochromocytomas. Inhibition of the phosphatidylinositol-3 kinase (PI3K)-Akt pathway has been shown to be an effective treatment of neuroendocrine (NE) tumors in vitro. We hypothesized that inhibition of the PI3K-Akt pathway would be a viable strategy to inhibit growth and hormonal secretion in pheochromocytoma cells.
Methods
Sixteen pheochromocytomas were analyzed for expression of phosphorylated Akt and the NE marker achaete scute complex-like 1 (ASCL1). Pheochromocytoma PC-12 cells were treated with up to 100 μM of the PI3K-specific inhibitor LY294002 for 48 h. Western blot analysis was used to measure phosphorylated Akt, total Akt, ASCL1, chromogranin A (CgA), and markers of apoptosis. Growth was assessed by a methylthiazolyldiphenyl-tetrazolium (MTT) bromide cellular proliferation assay for six days.
Results
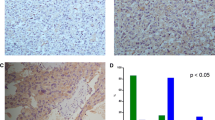
Human pheochromocytomas expressed significant amounts of phosphorylated Akt, and there was a significant correlation between malignant pheochromocytomas and the amount of expressed ASCL1. LY294002 significantly inhibited the PI3K-Akt pathway. Treatment led to a dose-dependent decrease in both ASCL1 and CgA, indicating an alteration in the NE phenotype and hormonal suppression. Treatment decreased cellular proliferation, and cleavage of the apoptotic markers caspase-3 and PARP was observed.
Conclusions
Human pheochromocytoma tumor samples express high levels of phosphorylated Akt. LY294002 effectively inhibits the PI3K-Akt pathway, suppresses NE tumor markers, and decreases cellular proliferation via apoptosis in vitro. Inhibition of the PI3K pathway may represent a new strategy in the treatment of pheochromocytomas.





Similar content being viewed by others
References
Adler JT, Meyer-Rochow GY, Chen H et al (2008) Pheochromocytoma: current approaches and future directions. Oncologist 13:779–793
Manger WM (2005) The vagaries of pheochromocytomas. Am J Hypertens 18:1266–1270
Manger WM, Gifford JRW (1996) Clinical and experimental pheochromocytoma. Blackwell Science, Cambridge
Stein PP, Black HR (1991) A simplified diagnostic approach to pheochromocytoma. A review of the literature and report of one institution’s experience. Medicine (Baltimore) 70:46–66
Adler JT, Mack E, Chen H (2007) Isolated adrenal mass in patients with a history of cancer: remember pheochromocytoma. Ann Surg Oncol 14:2358–2362
Zhang X, Jin B, Huang C (2007) The PI3K/Akt pathway and its downstream transcriptional factors as targets for chemoprevention. Curr Cancer Drug Targets 7:305–316
Bussink J, van der Kogel AJ, Kaanders JH (2008) Activation of the PI3-K/AKT pathway and implications for radioresistance mechanisms in head and neck cancer. Lancet Oncol 9:288–296
Dudek H, Datta SR, Franke TF et al (1997) Regulation of neuronal survival by the serine-threonine protein kinase Akt. Science 275:661–665
Yao R, Cooper GM (1995) Requirement for phosphatidylinositol-3 kinase in the prevention of apoptosis by nerve growth factor. Science 267:2003–2006
Alvarez-Tejado M, Naranjo-Suarez S, Jimenez C et al (2001) Hypoxia induces the activation of the phosphatidylinositol 3-kinase/Akt cell survival pathway in PC12 cells: protective role in apoptosis. J Biol Chem 276:22368–22374
Fassnacht M, Weismann D, Ebert S et al (2005) AKT is highly phosphorylated in pheochromocytomas but not in benign adrenocortical tumors. J Clin Endocrinol Metab 90:4366–4370
Sippel RS, Carpenter JE, Kunnimalaiyaan M et al (2003) Raf-1 activation suppresses neuroendocrine marker and hormone levels in human gastrointestinal carcinoid cells. Am J Physiol Gastrointest Liver Physiol 285:G245–G254
Kappes A, Vaccaro A, Kunnimalaiyaan M et al (2006) ZM336372, a Raf-1 activator, inhibits growth of pheochromocytoma cells. J Surg Res 133:42–45
Kappes A, Vaccaro A, Kunnimalaiyaan M et al (2007) Lithium ions: a novel treatment for pheochromocytomas and paragangliomas. Surgery 141:161–165
Adler JT, Hottinger DG, Kunnimalaiyaan M et al (2008) Histone deacetylase inhibitors upregulate Notch-1 and inhibit growth in pheochromocytoma cells. Surgery 144:956–961
Kunnimalaiyaan M, Ndiaye M, Chen H (2006) Apoptosis-mediated medullary thyroid cancer growth suppression by the PI3K inhibitor LY294002. Surgery 140:1009–1014
Sippel RS, Carpenter JE, Kunnimalaiyaan M et al (2003) The role of human achaete-scute homolog-1 in medullary thyroid cancer cells. Surgery 134:866–871
Dai Y, Wei Z, Sephton CF et al (2007) Haloperidol induces the nuclear translocation of phosphatidylinositol 3′-kinase to disrupt Akt phosphorylation in PC12 cells. J Psychiatry Neurosci JPN 32:323–330
Huber K, Bruhl B, Guillemot F et al (2002) Development of chromaffin cells depends on MASH1 function. Development 129:4729–4738
Garlich JR, De P, Dey N et al (2008) A vascular targeted pan phosphoinositide 3-kinase inhibitor prodrug, SF1126, with antitumor and antiangiogenic activity. Cancer Res 68:206–215
Acknowledgments
Joel T. Adler is a Howard Hughes Medical Institute Research Training Fellow and is supported by the University of Wisconsin General Clinical Research Center. Additional support was provided by National Institutes of Health grants R21-CA117117, R01-CA121115, and R01-CA109053; the George H. A. Clowes, Jr., Memorial Research Career Development Award of the American College of Surgeons; a Carcinoid Cancer Foundation research award (to H.C.), and a Carcinoid Cancer Foundation Research Award (to M.K.).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Adler, J.T., Hottinger, D.G., Kunnimalaiyaan, M. et al. Inhibition of the PI3K Pathway Suppresses Hormonal Secretion and Limits Growth in Pheochromocytoma Cells. World J Surg 33, 2452–2457 (2009). https://doi.org/10.1007/s00268-009-0175-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-009-0175-3