Abstract
Background
Tranexamic acid (TXA) is a versatile antifibrinolytic agent that is widely used in modern surgeries. This review assessed the safety and efficacy of intravenous (IV) TXA in plastic surgery versus controls.
Methods
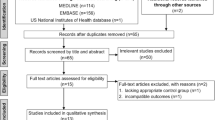
This review selected English-language Randomized controlled trials (RCTs) evaluating IV TXA effects in plastic surgery from four electronic databases, PubMed, Web of Science, Embase, and Cochrane Library up to April 9, 2023. Primary outcomes were blood loss volume (BLV) and transfusion occurrence, with operation time and surgical field assessment as secondary outcomes. IV TXA-related complications were also important indicators. Meta-analyses and qualitative analyses were conducted and the quality of the evidence was assessed.
Results
Thirty RCTs with 2150 patients were included. The total standard mean difference (SMD) of BLV and pooled relative risk of transfusion occurrence between the IV TXA and the control groups were − 1.11 (95% CI, − 1.42 to − 0.80) and 0.36 (95% CI, 0.23 to 0.55) respectively, indicating a significant blood loss reduction with IV TXA treatment, while an ambiguous outcome of operation time was observed, with an SMD of − 0.22 (95% CI, − 0.42 to − 0.02). The quality of evidence for BLV and transfusion occurrence was low and medium, respectively. A quantitative analysis of surgical field assessment was not performed because of the substantial heterogeneity in scoring methods. No IV TXA-related complications were observed.
Conclusions
In plastic surgery, IV TXA administration results in less blood loss, reduced need for transfusion and better surgical fields but probably does not increase the risk of adverse events.
Level of Evidence I
This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine Ratings, please refer to Table of Contents or online Instructions to Authors www.springer.com/00266.
Graphical Abstract







Similar content being viewed by others
Change history
02 October 2023
A Correction to this paper has been published: https://doi.org/10.1007/s00266-023-03694-4
References
Wang Y, Sun W, Wang X, Ren X, Gao A, Li M, Wang X (2022) Comparison of transfusion reactions in children and adults: a systematic review and meta-analysis. Pediatr Blood Cancer 69(9):e29842. https://doi.org/10.1002/pbc.29842
Jason MW, Banuelos J, Jacobson SR, Manrique OJ, Nguyen M-DT, Harless CA, Tran NV, Martinez-Jorge J (2020) Intravenous tranexamic acid in implant-based breast reconstruction safely reduces hematoma without thromboembolic events. Plast Reconstr Surg 146(2):238–245
Sharma H, Arora S, Bhatia N, Rattan V, Sethi S (2020) Tranexamic acid is associated with improved operative field in orthognathic surgery. J Oral Maxillofac Surg 78(9):1509–1517. https://doi.org/10.1016/j.joms.2020.04.037
Afzali SL, Panahi H, Ganji F, Ziaei S, Sedaghat N (2022) Re-evaluating the effect of preoperative tranexamic acid on blood loss and field quality during rhinoplasty: a randomized double-blinded controlled trial. Aesthetic Plast Surg 46(3):1314–1320. https://doi.org/10.1007/s00266-021-02594-9
Chen K, Sinelnikov MY, Nikolenko VN, Reshetov IV, Cao Y, Li Z, Kochurova EV, Nikolenko SN, Avila-Rodríguez M, Somasundaram SG, Kirkland CE, Aliev G (2019) The use of fibrin-based tissue adhesives for breast in reconstructive and plastic surgery. Curr Top Med Chem 19(32):2985–2990. https://doi.org/10.2174/1568026619666191112101448
Heyns M, Knight P, Steve AK, Yeung JK (2021) A single preoperative dose of tranexamic acid reduces perioperative blood loss: a meta-analysis. Ann Surg 273(1):75–81. https://doi.org/10.1097/sla.0000000000003793
Ockerman A, Vanassche T, Garip M, Vandenbriele C, Engelen MM, Martens J, Politis C, Jacobs R, Verhamme P (2021) Tranexamic acid for the prevention and treatment of bleeding in surgery, trauma and bleeding disorders: a narrative review. Thromb J. https://doi.org/10.1186/s12959-021-00303-9
Murao S, Nakata H, Roberts I, Yamakawa K (2021) Effect of tranexamic acid on thrombotic events and seizures in bleeding patients: a systematic review and meta-analysis. Crit Care 25(1):380. https://doi.org/10.1186/s13054-021-03799-9
Zheng C, Ma J, Xu J, Wu L, Wu Y, Liu Y, Shen B (2023) The optimal regimen, efficacy and safety of tranexamic acid and aminocaproic acid to reduce bleeding for patients after total hip arthroplasty: a systematic review and Bayesian network meta-analysis. Thromb Res 221:120–129. https://doi.org/10.1016/j.thromres.2022.11.010
Abboud NM, Kapila AK, Abboud S, Yaacoub E, Abboud MH (2021) The combined effect of intravenous and topical tranexamic acid in liposuction: a randomized double-blinded controlled trial. Aesthet Surg J Open Forum. https://doi.org/10.1093/asjof/ojab002
Apipan B, Rummasak D, Narainthonsaenee T (2018) The effect of different dosage regimens of tranexamic acid on blood loss in bimaxillary osteotomy: a randomized, double-blind, placebo-controlled study. Int J Oral Maxillofac Surg 47(5):608–612. https://doi.org/10.1016/j.ijom.2017.10.007
Morrison RJM, Tsang B, Fishley W, Harper I, Joseph JC, Reed MR (2017) Dose optimisation of intravenous tranexamic acid for elective hip and knee arthroplasty: the effectiveness of a single pre-operative dose. Bone Joint Res 6(8):499–505. https://doi.org/10.1302/2046-3758.68.BJR-2017-0005.R1
Justin CC, Glasgold RA, Alloju LM, Glasgold MJ (2021) Effects of intravenous tranexamic acid during rhytidectomy: a randomized, controlled, Double-Blind Pilot Study. Aesthet Surg J 41(2):155–160. https://doi.org/10.1093/asj/sjaa072
Nishat M, Iqbal S, Ahmed A, Younas S, Deokah LA, Rashid Z (2021) Clinical study of evaluation of the effect of tranexamic acid in primary cleft palate surgery in children. Pak J Med Health Sci. 15(10):2708–2709. https://doi.org/10.53350/pjmhs2115102708
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71. https://doi.org/10.1136/bmj.n71
Guyatt GH, Oxman AD, Vist G, Kunz R, Brozek J, Alonso-Coello P, Montori V, Akl EA, Djulbegovic B, Falck-Ytter Y, Norris SL, Williams JW Jr, Atkins D, Meerpohl J, Schünemann HJ (2011) GRADE guidelines: 4. Rating the quality of evidence–study limitations (risk of bias). J Clin Epidemiol 64(4):407–415. https://doi.org/10.1016/j.jclinepi.2010.07.017
Guyatt GH, Oxman AD, Montori V, Vist G, Kunz R, Brozek J, Alonso-Coello P, Djulbegovic B, Atkins D, Falck-Ytter Y, Williams JW Jr, Meerpohl J, Norris SL, Akl EA, Schünemann HJ (2011) GRADE guidelines: 5. Rating the quality of evidence–publication bias. J Clin Epidemiol 64(12):1277–1282. https://doi.org/10.1016/j.jclinepi.2011.01.011
Guyatt GH, Oxman AD, Kunz R, Brozek J, Alonso-Coello P, Rind D, Devereaux PJ, Montori VM, Freyschuss B, Vist G, Jaeschke R, Williams JW Jr, Murad MH, Sinclair D, Falck-Ytter Y, Meerpohl J, Whittington C, Thorlund K, Andrews J, Schünemann HJ (2011) GRADE guidelines 6. Rating the quality of evidence–imprecision. J Clin Epidemiol 64(12):1283–1293. https://doi.org/10.1016/j.jclinepi.2011.01.012
Guyatt GH, Oxman AD, Kunz R, Woodcock J, Brozek J, Helfand M, Alonso-Coello P, Glasziou P, Jaeschke R, Akl EA, Norris S, Vist G, Dahm P, Shukla VK, Higgins J, Falck-Ytter Y, Schünemann HJ (2011) GRADE guidelines: 7. Rating the quality of evidence–inconsistency. J Clin Epidemiol 64(12):1294–1302. https://doi.org/10.1016/j.jclinepi.2011.03.017
Guyatt GH, Oxman AD, Kunz R, Woodcock J, Brozek J, Helfand M, Alonso-Coello P, Falck-Ytter Y, Jaeschke R, Vist G, Akl EA, Post PN, Norris S, Meerpohl J, Shukla VK, Nasser M, Schünemann HJ (2011) GRADE guidelines: 8. Rating the quality of evidence–indirectness. J Clin Epidemiol 64(12):1303–1310. https://doi.org/10.1016/j.jclinepi.2011.04.014
Proctor MR, Meier P, Pereira L, McGowan F, Scharp L, Rogers G, Meara J, Prescilla R, Soriano S, Zurakowski D, Sethna N, Goobie S (2011) Efficacy of tranexamic acid on blood loss and transfusion rates in pediatric craniosynostosis surgery: a double blind placebo controlled study. J Neurosurg 115(2):A419–A420
Choi WS (2015) The value of tranexamic acid in orthognathic surgery. Int J Oral Max Surg 44:e3. https://doi.org/10.1016/j.ijom.2015.08.920
Hanna MG, Refaie A, Gouda N, Obaya G (2010) Reduction of peri-operative bleeding in craniofacial surgeries in paediatrics. Comparison between recombinant factor VII and tranexamic acid. Egypt J Anaesth 26(1):53–61
Khavidaki GD, Yaghoubi S, Naderi M, Malek F (2022) The effect of preoperative intravenous tranexamic acid on the amount of bleeding in patients undergoing cleft palate reconstruction surgery. Iran J Pediatrcs. https://doi.org/10.5812/ijp-118170
Durán de la Fuente P, García-Fernández J, Pérez-López C, Carceller F, Gilsanz Rodríguez F (2003) Usefulness of tranexamic acid in cranial remodeling surgery. Rev Esp Anestesiol Reanim 50(8):388–394
Mortazavi SH, Hamdzadeh H, Afsharinia S (2016) The effect of tranexamic acid on the blood loss volumes in the patients underwent orthognathic surgeries. J Kerman Univ Med Sci 23(1):137–144
Weissler JM, Banuelos J, Jacobson SR, Manrique OJ, Nguyen MDT, Harless CA, Tran NV, Martinez-Jorge J (2020) Intravenous tranexamic acid in implant-based breast reconstruction safely reduces hematoma without thromboembolic events. Plast Reconstr Surg 146(2):238–245. https://doi.org/10.1097/PRS.0000000000006967
Fenger-Eriksen C, Lindholm AD, Krogh L, Hell T, Berger M, Hermann M, Fries D, Juul N, Rasmussen M, Hvas AM (2020) Effect of tranexamic acid on coagulation and fibrin clot properties in children undergoing craniofacial surgery. Thromb Haemost 120(3):392–399. https://doi.org/10.1055/s-0039-3402762
Schaden E, Kimberger O, Kraincuk P, Baron DM, Metnitz PG, Kozek-Langenecker S (2012) Perioperative treatment algorithm for bleeding burn patients reduces allogeneic blood product requirements. Br J Anaesth 109(3):376–381. https://doi.org/10.1093/bja/aes186
Sidelmann JJ, Gram JB, Godtfredsen ACM, Thorn JJ, Ingerslev J, Pinholt EM (2020) Orthognathic surgery-induced fibrinolytic shutdown is amplified by tranexamic acid. J Oral Maxillofac Surg 78(7):1183–1189. https://doi.org/10.1016/j.joms.2020.02.026
Sakallioğlu Ö, Polat C, Soylu E, Düzer S, Orhan İ, Akyiğit A (2015) The efficacy of tranexamic acid and corticosteroid on edema and ecchymosis in septorhinoplasty. Ann Plas Surg 74(4):392–396. https://doi.org/10.1097/SAP.0b013e3182a1e527
Goobie SM, Staffa SJ, Meara JG, Proctor MR, Tumolo M, Cangemi G, Disma N (2020) High-dose versus low-dose tranexamic acid for paediatric craniosynostosis surgery: a double-blind randomised controlled non-inferiority trial. Br J Anaesth 125(3):336–345. https://doi.org/10.1016/j.bja.2020.05.054
Modir H, Moshiri E, Naseri N, Faraji F, Almasi-Hashiani A (2021) A randomized parallel design trial of the efficacy and safety of tranexamic acid, dexmedetomidine and nitroglycerin in controlling intraoperative bleeding and improving surgical field quality during septorhinoplasty under general anesthesia. Med Gas Res 11(4):131–137. https://doi.org/10.4103/2045-9912.318857
Jozefowicz E, Sabourdin N, Lambelin V, Lejeune V, Delassus R, Tavernier B (2022) The effect of tranexamic acid on blood loss in orthognathic surgery: a randomized, placebo-controlled, equivalence study. Int J Oral Maxillofac Surg 51(5):637–642. https://doi.org/10.1016/j.ijom.2021.08.018
Secher JJ, Sidelmann JJ, Ingerslev J, Thorn JJ, Pinholt EM (2018) The effect of tranexamic acid and gender on intraoperative bleeding in orthognathic surgery—a randomized controlled trial. J Oral Maxillofac Surg 76(6):1327–1333. https://doi.org/10.1016/j.joms.2017.11.015
Christabel A, Anantanarayanan P, Subash P, Soh CL, Ramanathan M, Muthusekhar MR, Narayanan V (2016) Comparison of pterygomaxillary dysjunction with tuberosity separation in isolated Le Fort I osteotomies: a prospective, multi-centre, triple-blind, randomized controlled trial. Int J Oral Maxillofac Surg 45(2):180–185. https://doi.org/10.1016/j.ijom.2015.07.021
Christabel A, Muthusekhar MR, Narayanan V, Ashok Y, Soh CL, Ilangovan M, Krishnan N (2014) Effectiveness of tranexamic acid on intraoperative blood loss in isolated Le Fort I osteotomies-a prospective, triple blinded randomized clinical trial. J Craniomaxillofac Surg 42(7):1221–1224. https://doi.org/10.1016/j.jcms.2014.03.003
Sankar D, Krishnan R, Veerabahu M, Vikraman B (2012) Evaluation of the efficacy of tranexamic acid on blood loss in orthognathic surgery. A prospective, randomized clinical study. Int J Oral Maxillofac Surg 41(6):713–717. https://doi.org/10.1016/j.ijom.2012.01.008
Karimi A, Mohammadi SS, Hasheminasab M (2012) Efficacy of tranexamic acid on blood loss during bimaxilary osteotomy: a randomized double blind clinical trial. Saudi J Anaesth 6(1):41–45. https://doi.org/10.4103/1658-354X.93057
Choi WS, Irwin MG, Samman N (2009) The effect of tranexamic acid on blood loss during orthognathic surgery: a randomized controlled trial. J Oral Maxillofac Surg 67(1):125–133. https://doi.org/10.1016/j.joms.2008.08.015
Alam MS, Barh A, Kundu D (2022) A randomized control trial on the role of tranexamic acid in preventing intraoperative bleeding during external dacryocystorhinostomy. Indian J Ophthalmol 70(10):3634–3637. https://doi.org/10.4103/ijo.IJO_925_22
Avci H (2021) The effect of different dose regimens of tranexamic acid in reducing blood loss in rhinoplasty: a prospective randomized controlled study. J Craniofac Surg 32(5):e442–e444. https://doi.org/10.1097/SCS.0000000000007247
Mehdizadeh M, Ghassemi A, Khakzad M, Mir M, Nekoohesh L, Moghadamnia A, Bijani A, Mehrbakhsh Z, Ghanepur H (2018) Comparison of the effect of dexamethasone and tranexamic acid, separately or in combination on post-rhinoplasty edema and ecchymosis. Aesthetic Plast Surg 42(1):246–252. https://doi.org/10.1007/s00266-017-0969-x
Ghavimi MA, Talesh KT, Ghoreishizadeh A, Chavoshzadeh MA, Zarandi A (2017) Efficacy of tranexamic acid on side effects of rhinoplasty: a randomized double-blind study. J Craniomaxillofac Surg 45(6):897–902. https://doi.org/10.1016/j.jcms.2017.03.001
Beikaei M, Ghazipour A, Derakhshande V, Saki N, Nikakhlagh S (2015) Evaluating the effect of intravenous tranexamic acid on intraoperative bleeding during elective rhinoplasty surgery. Biomed Pharmacol J. 8SE:753–759. https://doi.org/10.13005/bpj/779
Fenger-Eriksen C, D’Amore Lindholm A, Nørholt SE, von Oettingen G, Tarpgaard M, Krogh L, Juul N, Hvas AM, Rasmussen M (2019) Reduced perioperative blood loss in children undergoing craniosynostosis surgery using prolonged tranexamic acid infusion: a randomised trial. Br J Anaesth 122(6):760–766. https://doi.org/10.1016/j.bja.2019.02.017
Kim EJ, Kim YO, Shim KW, Ko BW, Lee JW, Koo BN (2018) Effects of tranexamic acid based on its population pharmacokinetics in pediatric patients undergoing distraction osteogenesis for craniosynostosis: rotational thromboelastometry (ROTEM(TM)) analysis. Int J Med Sci 15(8):788–795. https://doi.org/10.7150/ijms.25008
Dadure C, Sauter M, Bringuier S, Bigorre M, Raux O, Rochette A, Canaud N, Capdevila X (2011) Intraoperative tranexamic acid reduces blood transfusion in children undergoing craniosynostosis surgery: a randomized double-blind study. Anesthesiology 114(4):856–861. https://doi.org/10.1097/ALN.0b013e318210f9e3
Goobie SM, Meier PM, Pereira LM, McGowan FX, Prescilla RP, Scharp LA, Rogers GF, Proctor MR, Meara JG, Soriano SG, Zurakowski D, Sethna NF (2011) Efficacy of tranexamic acid in pediatric craniosynostosis surgery: a double-blind, placebo-controlled trial. Anesthesiology 114(4):862–871. https://doi.org/10.1097/ALN.0b013e318210fd8f
Cohen JC, Glasgold RA, Alloju LM, Glasgold MJ (2021) Effects of intravenous tranexamic acid during rhytidectomy: a randomized, controlled. Double-Blind Pilot Stud Aesthet Surg J 41(2):155–160. https://doi.org/10.1093/asj/sjaa072
El Minawi HM, Kadry HM, El-Essawy NM, El Saadany ZA, Nouh OM (2022) The effect of tranexamic acid on blood loss in liposuction: a randomized controlled study. Eur J Plast Surg. https://doi.org/10.1007/s00238-022-01995-6
Hoyos AE, Duran H, Cardenas-Camarena L, Bayter JE, Cala L, Perez M, Lopez A, Talleri G, Domínguez-Millan R, Mogollon IR (2022) Use of tranexamic acid in liposculpture: a double-blind, multicenter. Randomized Clin Trial Plast Reconstr Surg 150(3):569–577. https://doi.org/10.1097/prs.0000000000009434
Dakir A, Ramalingam B, Ebenezer V, Dhanavelu P (2014) Efficacy of Tranexamic acid in reducing blood loss during maxillofacial trauma surgery-A pilot study. J Clin Diagn Res 8(5):6–8. https://doi.org/10.7860/JCDR/2014/8680.4313
Zaman SU, Zakir I, Faraz Q, Akhtar S, Nawaz A, Adeel M (2019) Effect of single-dose intravenous tranexamic acid on postoperative nasal bleed in septoplasty. Eur Ann Otorhinolaryngol Head Neck Dis 136(6):435–438. https://doi.org/10.1016/j.anorl.2018.10.019
Durga P, Raavula P, Gurajala I, Gunnam P, Veerabathula P, Reddy M, Upputuri O, Ramachandran G (2015) Evaluation of the efficacy of tranexamic acid on the surgical field in primary cleft palate surgery on children-A prospective, randomized clinical study. Cleft Palate Craniofac J 52(5):e183–e187. https://doi.org/10.1597/14-263
Arantes GC, Pereira RMR, de Melo DB, Alonso N, Duarte MdCMB (2017) Effectiveness of tranexamic acid for reducing intraoperative bleeding in palatoplasties: a randomized clinical trial. J Craniomaxillofac Surg 45(5):642–648. https://doi.org/10.1016/j.jcms.2016.12.021
Shafa A, Shetabi H, Adineh-Mehr L, Bahrami K (2021) Selection of the optimal dosage of tranexamic acid to reduce blood loss during pediatric cleft palate surgery. Tzu Chi Med J 33(2):181–187. https://doi.org/10.4103/tcmj.tcmj_111_20
Bhatia N, Sen I, Kumari K, Kumar P, Bharti N (2017) Impact of single dose intravenous tranexamic acid on peri-operative blood transfusion requirements in burn patients: a prospective, randomized trial. Egypt J Anaesth 33(3):251–255. https://doi.org/10.1016/j.egja.2017.05.004
Ks A, Kumar P, Subair M, Sharma RK (2021) Effect of single dose intravenous tranexamic acid on blood loss in tangential excision of burn wounds—a double blind randomised controlled trial. Burns. https://doi.org/10.1016/j.burns.2021.08.021
Patel PA, Wyrobek JA, Butwick AJ, Pivalizza EG, Hare GMT, Mazer CD, Goobie SM (2022) Update on applications and limitations of perioperative tranexamic acid. Anesth Analg 135(3):460–473
Ausen K, Fossmark R, Spigset O, Pleym H (2015) Randomized clinical trial of topical tranexamic acid after reduction mammoplasty. Br J Surg 102(11):1348–1353. https://doi.org/10.1002/bjs.9878
Zapata-Copete JA, Gomez-Ospina JC, Garcia-Perdomo HA, Caycedo DJ (2022) Role of tranexamic acid in craniosynostosis surgery: Systematic review and meta-analysis. J Plast Reconstr Aesthet Surg 75(4):1389–1398. https://doi.org/10.1016/j.bjps.2021.11.064
Goldstein K, Jones C, Kay J, Shin J, de Sa D (2022) Tranexamic acid administration in arthroscopic surgery is a safe adjunct to decrease postoperative pain and swelling: a systematic review and meta-analysis. Arthroscopy 38(4):1366-1377.e1369. https://doi.org/10.1016/j.arthro.2021.10.001
Liechti R, van de Wall BJM, Hug U, Fritsche E, Franchi A (2022) Tranexamic acid use in breast surgery: a systematic review and meta-analysis. Plast Reconstr Surg. https://doi.org/10.1097/prs.0000000000010071
Taeuber I, Weibel S, Herrmann E, Neef V, Schlesinger T, Kranke P, Messroghli L, Zacharowski K, Choorapoikayil S, Meybohm P (2021) Association of intravenous tranexamic acid with thromboembolic events and mortality: a systematic review, meta-analysis, and meta-regression. JAMA Surg 156(6):e210884. https://doi.org/10.1001/jamasurg.2021.0884
Acknowledgement
The authors declare that they have no conflict of interest.
Funding
Funding agencies in the public, commercial, or non-profit sectors did not provide specific grants for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Human or Animal Rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
For this type of study, informed consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, S., Yang, J. & Lin, L. Intravenous Application of Tranexamic Acid in Patients Undergoing Plastic Surgery: A Systematic Review and Meta-analysis with GRADE Quality Assessment. Aesth Plast Surg 48, 543–558 (2024). https://doi.org/10.1007/s00266-023-03637-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-023-03637-z