Abstract
Background
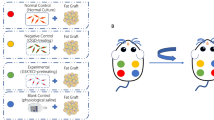
Fat transplantation supported by supplementation with ASCs has become a reliable procedure for treating soft tissue defects. However, the unpredictable survival rates for grafted fat remains a challenge with post-transplantation ischemia causing tissue loss. MiR126, which regulates VEGF signaling, is an endothelial cell-specific miRNA known to play an essential role in angiogenesis. We hypothesized that increased miR126 expression in grafted ASCs may promote fat survival within an autologous fat transfer model.
Methods
Rat adipose-derived stem cells were isolated, expanded ex vivo for three passages and then transduced with miR126. We used PCR to verify lentiviral transduction and ELISA to confirm VEGF expression. We then mixed autologous fat tissues from our rat model with transduced ASCs, augmented with a nonsense control or miR126 expression vector. These mixtures were used in the fat grafting procedure, completed via subcutaneous injection at three paravertebral points in each rat. Fat grafts were then harvested on days 4, 7, 14, and 28 post-transplant and evaluated for survival, neovascularization, and protein expression via western blot.
Results
VEGF expression levels in ASCs, Con-ASCs, and miR126-ASCs were not significantly different. However, miR126-ASCs experienced significantly improved survival on days 7, 14, and 28 when compared with the other groups. These ASCs also presented with the greatest capillary density on days 7, 14, and 28 post-transplantation as well as increased p-ERK and p-AKT expression when compared to the other groups.
Conclusion
This data suggests that miR126 augmentation of ASCs may help to enhance the survival and angiogenic capacity of transplanted fat tissues, and that this augmentation was not dependent on VEGF but rather the activation of the ERK/AKT pathway.
No Level Assigned
This journal requires that authors assign a level of evidence to each submission to which Evidence-Based Medicine rankings are applicable. This excludes Review Articles, Book Reviews, and manuscripts that concern Basic Science, Animal Studies, Cadaver Studies, and Experimental Studies. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.






Similar content being viewed by others
Abbreviations
- ASCs:
-
Adipose-derived stem cells
- VEGF:
-
Vascular endothelial growth factor
- IL-10:
-
Anti-inflammatory cytokine interleukin 10
- qRT-PCR:
-
Quantitative reverse transcriptional-Tim PCR
- ELISA:
-
Enzyme-linked immunosorbent assay
- PBS:
-
Phosphate buffered saline
References
Rubin JP, Marra KG (2011) Soft tissue reconstruction. Methods Mol Biol 702:395–400
Clark N, Sherman R (1993) Soft-tissue reconstruction of the foot and ankle. Orthop Clin North Am 24:489–503
Carpaneda CA, Ribeiro MT (1993) Study of the histologic alterations and viability of the adipose graft in humans. Aesthetic Plast Surg 17:43–47
Coleman SR (2006) Structural fat grafting: more than a permanent filler. Plast Reconstr Surg 118:108S-120S
Herold C, Ueberreiter K, Busche MN et al (2013) Autologous fat transplantation: volumetric tools for estimation of volume survival A systematic review. Aesthetic Plast Surg 37:380–387
Ross RJ, Shayan R, Mutimer KL et al (2014) Autologous fat grafting: current state of the art and critical review. Ann Plast Surg 73:352–357
Eto H, Kato H, Suga H et al (2012) The fate of adipocytes after nonvascularized fat grafting: evidence of early death and replacement of adipocytes. Plast Reconstr Surg 129:1081–1092
Gan F, Liu L, Zhou Q et al (2022) Effects of adipose-derived stromal cells and endothelial progenitor cells on adipose transplant survival and angiogenesis. PLoS ONE 17:e261498
Vanderstichele S, Vranckx JJ (2022) Anti-fibrotic effect of adipose-derived stem cells on fibrotic scars. World J Stem Cells 14:200–213
Inui M, Martello G, Piccolo S (2010) MicroRNA control of signal transduction. Nat Rev Mol Cell Biol 11:252–263
Endo-Takahashi Y, Negishi Y, Nakamura A et al (2014) Systemic delivery of miR-126 by miRNA-loaded Bubble liposomes for the treatment of hindlimb ischemia. Sci Rep 4:3883
Shoshani O, Livne E, Armoni M et al (2005) The effect of interleukin-8 on the viability of injected adipose tissue in nude mice. Plast Reconstr Surg 115:853–859
Chajchir A (1996) Fat injection: long-term follow-Up. Aesthetic Plast Surg 20:291–296
Zuk PA, Zhu M, Ashjian P et al (2002) Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell 13:4279–4295
Yoshimura K, Sato K, Aoi N et al (2008) Cell-assisted lipotransfer for cosmetic breast augmentation: supportive use of adipose-derived stem/stromal cells. Aesthetic Plast Surg 32(48–55):56–57
Gontijo-de-Amorim NF, Charles-de-Sa L, Rigotti G (2017) Mechanical supplementation with the stromal vascular fraction yields improved volume retention in facial lipotransfer: a 1-Year comparative study. Aesthet Surg J 37:975–985
Kolle ST, Duscher D, Taudorf M et al (2020) Ex vivo-expanded autologous adipose tissue-derived stromal cells ensure enhanced fat graft retention in breast augmentation: a randomized controlled clinical trial. Stem Cells Transl Med 9:1277–1286
Gentile P, Sterodimas A, Calabrese C et al (2020) Regenerative application of stromal vascular fraction cells enhanced fat graft maintenance: clinical assessment in face rejuvenation. Expert Opin Biol Ther 20:1503–1513
Naderi N, Combellack EJ, Griffin M et al (2017) The regenerative role of adipose-derived stem cells (ADSC) in plastic and reconstructive surgery. Int Wound J 14:112–124
Horie H, Hisatome I, Kurata Y et al (2022) alpha1-Adrenergic receptor mediates adipose-derived stem cell sheet-induced protection against chronic heart failure after myocardial infarction in rats. Hypertens Res 45:283–291
Bartynski J, Marion MS, Wang TD (1990) Histopathologic evaluation of adipose autografts in a rabbit ear model. Otolaryngol Head Neck Surg 102(4):314–321
Yamaguchi M, Matsumoto F, Bujo H et al (2005) Revascularization determines volume retention and gene expression by fat grafts in mice. Exp Biol Med (Maywood) 230:742–748
Lei M, Liu SQ, Peng H et al (2008) Effect of rhVEGF gene transfection on survival of grafts after autologous free granular fat transplantation in rats. Chin J Traumatol 11:49–53
Nishimura T, Hashimoto H, Nakanishi I et al (2000) Microvascular angiogenesis and apoptosis in the survival of free fat grafts. Laryngoscope 110:1333–1338
Lu F, Li J, Gao J et al (2009) Improvement of the survival of human autologous fat transplantation by using VEGF-transfected adipose-derived stem cells. Plast Reconstr Surg 124:1437–1446
Wang S, Aurora AB, Johnson BA et al (2008) The endothelial-specific microRNA miR-126 governs vascular integrity and angiogenesis. Dev Cell 15:261–271
van Solingen C, Seghers L, Bijkerk R et al (2009) Antagomir-mediated silencing of endothelial cell specific microRNA-126 impairs ischemia-induced angiogenesis. J Cell Mol Med 13:1577–1585
Tauber Z, Cizkova K, Janikova M et al (2019) Serum C-peptide level correlates with the course of muscle tissue healing in the rabbit model of critical limb ischemia. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 163:132–140
Qiu C, Xie Q, Zhang D et al (2014) GM-CSF induces cyclin D1 expression and proliferation of endothelial progenitor cells via PI3K and MAPK signaling. Cell Physiol Biochem 33:784–795
Acknowledgments
This work was supported by the East Hospital affiliated with Tongji University introduced a talent research startup fund (Grant Number DFRC2019008), and the featured clinical discipline project of Shanghai Pudong (Grant Number PWYts2021-07).
Funding
This work was supported by the East Hospital affiliated with Tongji University introduced a talent research startup fund (Grant Number DFRC2019008), and the featured clinical discipline project of Shanghai Pudong (Grant Number PWYts2021-07).
Author information
Authors and Affiliations
Contributions
XJ, HH, and BN performed the experiment. AL, LZ, and HJ designed the experimental technique. XJ and BN analyzed the data. XJ and HH wrote the paper.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest to disclose.
Human and Animals Rights
All applicable institutional and/or national guidelines for the care and use of animals were followed.
Informed Consent
For this type of study, informed consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jie, X., Hu, H., Nie, B. et al. Effects of miR126 Expressing Adipose-Derived Stem Cells on Fat Graft Survival and Angiogenesis. Aesth Plast Surg 47, 825–832 (2023). https://doi.org/10.1007/s00266-022-03077-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-022-03077-1