Abstract
Background
Rhinoplasty is one of the most challenging cosmetic surgical operations. The procedure has been known to precipitate higher levels of edema and ecchymosis in the periorbital and paranasal regions. The literature recommends the use of corticosteroids such as dexamethasone to alleviate these postoperative morbidities. In this review, we aim to provide a current state of evidence concerning the influence of dexamethasone together with rhinoplasty on intraoperative and postoperative morbidities.
Methods
A systematic identification of the literature was performed according to PRISMA guidelines on four academic databases: MEDLINE, Scopus, EMBASE and CENTRAL. A meta-analysis compared the influence of dexamethasone and normal saline administered during rhinoplasty on the amount of intraoperative blood loss, postoperative edema and ecchymosis.
Results
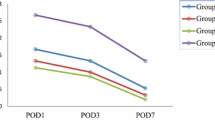
Out of 1045 records, ten articles including 374 participants (mean age: 25.8 ± 2.5 years) were included in this review. This systematic review presents a 1b level of evidence supporting the use of dexamethasone during rhinoplasty to reduce the amount of intraoperative blood loss, edema and ecchymosis as compared to normal saline. The meta-analysis reveals beneficial effects for dexamethasone interventions by demonstrating medium to large effect reduction of the amount of intraoperative blood loss (Hedge’s g: − 0.69), mean edema score (− 1.09) and mean ecchymosis score (− 1.03) as compared to placebo groups using normal saline.
Conclusion
The current systematic review and meta-analysis recommend the administration of dexamethasone with rhinoplasty. The review reports beneficial effects of dexamethasone’s administration as compared to normal saline for reducing the amount of intraoperative blood loss, postoperative edema and ecchymosis.
Level of Evidence III
This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.








Similar content being viewed by others
References
Angelos PC, Been MJ, Toriumi DM (2012) Contemporary review of rhinoplasty. Arch Fac Plast Surg 14:238–247. https://doi.org/10.1001/archfacial.2012.577
Lee MR, Unger JG, Rohrich RJ (2011) Management of the nasal dorsum in rhinoplasty: a systematic review of the literature regarding technique, outcomes, and complications. Plast Reconstr Surg 128:538e–550e. https://doi.org/10.1097/PRS.0b013e31822b6a82
Rhee JS, Arganbright JM, McMullin BT, Hannley M (2008) Evidence supporting functional rhinoplasty or nasal valve repair: a 25-year systematic review. Otolaryngol Head Neck Surg 139:10–20. https://doi.org/10.1016/j.otohns.2008.02.007
Burget GC (1985) Aesthetic restoration of the nose. Clin Plast Surg 12:463–480
Dayan S, Kanodia R (2009) Has the pendulum swung too far? Trends in the teaching of endonasal rhinoplasty. Arch Fac Plast Surg 11:414–416. https://doi.org/10.1001/archfacial.2009.70
Millard DR (1981) Aesthetic reconstructive rhinoplasty. Clin Plast Surg 8:169–175
Toriumi DM, Mueller RA, Grosch T et al (1996) Vascular anatomy of the nose and the external rhinoplasty approach. Arch Otolaryngol Head Neck Surg 122:24–34. https://doi.org/10.1001/archotol.1996.01890130020003
Cochran CS, Landecker A (2008) Prevention and management of rhinoplasty complications. Plast Reconstr Surg 122:60e–67e. https://doi.org/10.1097/PRS.0b013e31817d53de
Lawson W, Kessler S, Biller HF (1983) Unusual and fatal complications of rhinoplasty. Arch Otolaryngol 109:164–169. https://doi.org/10.1001/archotol.1983.00800170030008
Rettinger G (2007) Risks and complications in rhinoplasty. GMS Curr Top Otorhinolaryngol Head Neck Surg 6:Doc08
Eftekharian HR, Rajabzadeh Z (2016) The efficacy of preoperative oral tranexamic acid on intraoperative bleeding during rhinoplasty. J Craniofac Surg 27:97–100. https://doi.org/10.1097/SCS.0000000000002273
Kargi E, Hoşnuter M, Babucçu O et al (2003) Effect of steroids on edema, ecchymosis, and intraoperative bleeding in rhinoplasty. Ann Plast Surg 51:570–574. https://doi.org/10.1097/01.sap.0000095652.35806.c5
Gurlek A, Fariz A, Aydogan H et al (2006) Effects of different corticosteroids on edema and ecchymosis in open rhinoplasty. Aesthetic Plast Surg 30:150–154. https://doi.org/10.1007/s00266-005-0158-1
Hatef DA, Ellsworth WA, Allen JN et al (2011) Perioperative steroids for minimizing edema and ecchymosis after rhinoplasty: a meta-analysis. Aesthet Surg J 31:648–657. https://doi.org/10.1177/1090820X11416110
Coroneos CJ, Voineskos SH, Cook DJ et al (2016) Perioperative corticosteroids reduce short-term edema and ecchymosis in rhinoplasty: a meta-analysis. Aesthet Surg J 36:136–146. https://doi.org/10.1093/asj/sjv138
Abraham SM, Lawrence T, Kleiman A et al (2006) Antiinflammatory effects of dexamethasone are partly dependent on induction of dual specificity phosphatase 1. J Exp Med 203:1883–1889. https://doi.org/10.1084/jem.20060336
Tsurufuji S, Sugio K, Takemasa F (1979) The role of glucocorticoid receptor and gene expression in the anti-inflammatory action of dexamethasone. Nature 280:408–410. https://doi.org/10.1038/280408a0
Griffies WS, Kennedy K, Gasser C et al (1989) Steroids in rhinoplasty. Laryngoscope 99:1161–1164. https://doi.org/10.1288/00005537-198911000-00010
Alajmi MA, Al-Abdulhadi KA, Al-Noumas HS, Kavitha G (2009) Results of intravenous steroid injection on reduction of postoperative edema in rhinoplasty. Indian J Otolaryngol Head Neck Surg 61:266–269. https://doi.org/10.1007/s12070-009-0080-y
Mehdizadeh M, Ghassemi A, Khakzad M et al (2018) Comparison of the effect of dexamethasone and tranexamic acid, separately or in combination on post-rhinoplasty edema and ecchymosis. Aesthet Plast Surg 42:246–252. https://doi.org/10.1007/s00266-017-0969-x
Goodman LS, Gilman A, Brunton LL et al (2006) Goodman & Gilman’s the pharmacological basis of therapeutics, 11th edn. McGraw-Hill, New York
Hwang SH, Lee JH, Kim BG et al (2015) The efficacy of steroids for edema and ecchymosis after rhinoplasty: a meta-analysis. Laryngoscope 125:92–98. https://doi.org/10.1002/lary.24883
Youssef TA, Elibiary H, Amish KF (2013) Role of steroids in reducing postoperative edema in rhinoplasty: a meta-analytic study. Eur Arch Otorhinolaryngol 270:1189–1193. https://doi.org/10.1007/s00405-012-2144-y
Gutierrez S, Wuesthoff C (2014) Testing the effects of long-acting steroids in edema and ecchymosis after closed rhinoplasty. Plast Surg (Oakv) 22:83–87
Valente DS, Steffen N, Carvalho LA et al (2015) Preoperative use of dexamethasone in rhinoplasty: a randomized, double-blind, placebo-controlled clinical trial. JAMA Fac Plast Surg 17:169–173. https://doi.org/10.1001/jamafacial.2014.1574
Moher D, Liberati A, Tetzlaff J et al (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6:e1000097. https://doi.org/10.1371/journal.pmed.1000097
Jørgensen L, Paludan-Müller AS, Laursen DRT et al (2016) Evaluation of the Cochrane tool for assessing risk of bias in randomized clinical trials: overview of published comments and analysis of user practice in Cochrane and non-Cochrane reviews. Syst Rev. https://doi.org/10.1186/s13643-016-0259-8
Viswanathan M, Ansari MT, Berkman ND et al (2008) Assessing the risk of bias of individual studies in systematic reviews of health care interventions. In: Kronick R, Slutsky J, Chang S (eds) Methods guide for effectiveness and comparative effectiveness reviews. Agency for Healthcare Research and Quality (US), Rockville (MD)
Burns PB, Rohrich RJ, Chung KC (2011) The levels of evidence and their role in evidence-based medicine. Plast Reconstr Surg 128:305–310. https://doi.org/10.1097/PRS.0b013e318219c171
Bax L, Yu L-M, Ikeda N, Moons KG (2007) A systematic comparison of software dedicated to meta-analysis of causal studies. BMC Med Res Methodol 7:40. https://doi.org/10.1186/1471-2288-7-40
Higgins JPT, Thompson SG, Spiegelhalter DJ (2009) A re-evaluation of random-effects meta-analysis. J R Stat Soc Ser A Stat Soc 172:137–159. https://doi.org/10.1111/j.1467-985X.2008.00552.x
Rosenthal R (1994) Parametric measures of effect size. The handbook of research synthesis. Russell Sage Foundation, New York, pp 231–244
Higgins JPT, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558. https://doi.org/10.1002/sim.1186
Petitti DB (2001) Approaches to heterogeneity in meta-analysis. Stat Med 20:3625–3633. https://doi.org/10.1002/sim.1091
Duval S, Tweedie R (2000) Trim and fill: a simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 56:455–463. https://doi.org/10.1111/j.0006-341x.2000.00455.x
Hoffmann DF, Cook TA, Quatela VC et al (1991) Steroids and rhinoplasty. A double-blind study. Arch Otolaryngol Head Neck Surg 117:990–993. https://doi.org/10.1001/archotol.1991.01870210062009discussion 994
Kara CO, Gökalan I (1999) Effects of single-dose steroid usage on edema, ecchymosis, and intraoperative bleeding in rhinoplasty. Plast Reconstr Surg 104:2213–2218. https://doi.org/10.1097/00006534-199912000-00041
Ozdel O, Kara CO, Kara IG et al (2006) Does corticosteroid usage in rhinoplasty cause mood changes? Adv Ther 23:809–816. https://doi.org/10.1007/bf02850322
Tuncel U, Turan A, Bayraktar MA et al (2013) Efficacy of dexamethasone with controlled hypotension on intraoperative bleeding, postoperative oedema and ecchymosis in rhinoplasty. J Craniomaxillofac Surg 41:124–128. https://doi.org/10.1016/j.jcms.2012.06.003
Bagheri SC, Khan HA, Jahangirnia A et al (2012) An analysis of 101 primary cosmetic rhinoplasties. J Oral Maxillofac Surg 70:902–909. https://doi.org/10.1016/j.joms.2011.02.075
Foda HMT (2005) Rhinoplasty for the multiply revised nose. Am J Otolaryngol 26:28–34. https://doi.org/10.1016/j.amjoto.2004.06.014
Lam SM, Williams EF (2002) Anatomic considerations in aesthetic rhinoplasty. Fac Plast Surg 18:209–214. https://doi.org/10.1055/s-2002-36488
Bhattacherjee P, Williams RN, Eakins KE (1983) A comparison of the ocular anti-inflammatory activity of steroidal and nonsteroidal compounds in the rat. Invest Ophthalmol Vis Sci 24:1143–1146
Leung YF, Tam POS, Lee WS et al (2003) The dual role of dexamethasone on anti-inflammation and outflow resistance demonstrated in cultured human trabecular meshwork cells. Mol Vis 9:425–439
Rosenbaum RB (2014) Chapter 50: connective tissue diseases, vasculitis, and the nervous system. In: Aminoff MJ, Josephson SA (eds) Aminoff’s neurology and general medicine, 5th edn. Academic Press, Boston, pp 1003–1029
Cronin J, Kennedy U, McCoy S et al (2012) Single dose oral dexamethasone versus multi-dose prednisolone in the treatment of acute exacerbations of asthma in children who attend the emergency department: study protocol for a randomized controlled trial. Trials 13:141. https://doi.org/10.1186/1745-6215-13-141
Saukko P, Knight B, Knight B (2015) Knight’s forensic pathology. CRC Press, Boca Raton
PLoS Medicine Editors (2011) Best practice in systematic reviews: the importance of protocols and registration. PLoS Med 8:e1001009. https://doi.org/10.1371/journal.pmed.1001009
Harmon LJ, Losos JB (2005) The effect of intraspecific sample size on type I and type II error rates in comparative studies. Evolution 59:2705–2710. https://doi.org/10.1111/j.0014-3820.2005.tb00981.x
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest to disclose.
Human and Animal Rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
For this type of study, informed consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Hu Liu is co-first author.
Rights and permissions
About this article
Cite this article
Bian, X., Liu, H., Sun, J. et al. Efficacy of Dexamethasone for Reducing Edema and Ecchymosis After Rhinoplasty: A Systematic Review and Meta-analysis. Aesth Plast Surg 44, 1672–1684 (2020). https://doi.org/10.1007/s00266-020-01743-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-020-01743-w