Abstract
Background
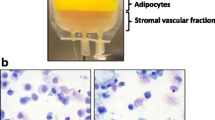
The ischemic environment of the receiving area compromises the outcome of autologous fat grafts. The aim of this study was to isolate and expand the stromal vascular fraction from patient lipoaspirates and investigate the gain in cell viability exerted by some protective agents against the blockage of mitochondrial respiration.
Methods
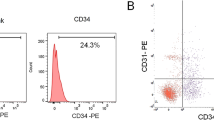
The aspirates were (1) washed, using the “Lull pgm system,” (2) centrifuged and (3) decanted. The corresponding stromal vascular fractions were isolated, and after cell adherence selection, the stromal/stem cell subpopulations were exposed to Antimycin A for 1 h. Then, the protection induced by cell pretreatment with deferoxamine, diazoxide and IGF-1 was evaluated.
Results
The residual cell viability of the “Lull pgm system”-washed samples was greater than that of the centrifuged samples (p < 0.05), and this advantage was maintained during the following 12 days of culture. The administration of 400 μM deferoxamine before Antimycin A treatment increased the number of viable cells from 56.5 to 80.8% (p < 0.05). On the contrary, the pretreatment with 250 μM diazoxide or 0.1 μg/ml IGF-1 did not exert any significant pro-survival action. Echinomycin abolished the positive effect of deferoxamine, suggesting that its protection involved HIF-1α.
Conclusions
Adipose-derived stromal–stem cells survive the inhibition of mitochondrial respiration better if the lipoaspirate is washed using the “Lull pgm system” rather than centrifuged. Moreover, a significant contribution to cell survival can be obtained by preconditioning stromal–stem cells with deferoxamine. In a clinical perspective, this drug could be safely administered before surgery to patients undergoing autologous fat transfer.
No Level Assigned
This journal requires that authors assign a level of evidence to each submission to which Evidence-Based Medicine rankings are applicable. This excludes Review Articles, Book Reviews, and manuscripts that concern Basic Science, Animal Studies, Cadaver Studies, and Experimental Studies. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.





Similar content being viewed by others
References
Khouri RK Jr, Khouri RK (2017) Current clinical applications of fat grafting. Plast Reconstr Surg. https://doi.org/10.1097/PRS.0000000000003648
Housman TS, Lawrence N, Mellen BG, George MN, Filippo JS, Cerveny KA, DeMarco M, Feldman SR, Fleischer AB (2002) The safety of liposuction: results of a national survey. Dermatol Surg 28:971–978
Chia CT, Neinstein RM, Theodorou SJ (2017) Evidence-based medicine: liposuction. Plast Reconstr Surg. https://doi.org/10.1097/PRS.0000000000002859
Strioga M, Viswanathan S, Darinskas A, Slaby O, Michalek J (2012) Same or not the same? Comparison of adipose tissue-derived versus bone marrow-derived mesenchymal stem and stromal cells. Stem Cells Dev 21:2724–2752
Rinker BD, Vyas KS (2016) Do stem cells have an effect when we fat graft? Ann Plast Surg 76(Suppl 4):S359–S363
Shim YH, Zhang RH (2017) Literature review to optimize the autologous fat transplantation procedure and recent technologies to improve graft viability and overall outcome: a systematic and retrospective analytic approach. Aesthetic Plast Surg 41:815–883
Coleman SR (1995) Long-term survival of fat transplants: controlled demonstrations. Aesthetic Plast Surg 19:421–425
Condé-Green A, Baptista LS, de Amorin NF, de Oliveira ED, da Silva KR, Pedrosa Cda S, Borojevic R, Pitanguy I (2010) Effects of centrifugation on cell composition and viability of aspirated adipose tissue processed for transplantation. Aesthet Surg J 30:249–255
Mecott GA, Gonzalez IZ, Montes de Oca R, Garza-Morales R, Gonzalez-Cantu I, Castro-Govea Y, Saucedo-Cárdenas O, García-Pérez MM (2019) Effect of decantation time on viability and apoptosis in adipocytes after liposuction. Aesthetic Plast Surg 43:228–232
Dos-Anjos Vilaboa S, Llull R, Mendel TA (2013) Returning fat grafts to physiologic conditions using washing. Plast Reconstr Surg. https://doi.org/10.1097/PRS.0b013e3182958be1
Morselli PG, Micai A, Giorgini FA (2016) “Lull pgm system” for autologous fat grafting: a simple closed system with minimal equipment and no extra cost. Plast Reconstr Surg Glob Open. https://doi.org/10.1097/GOX.0000000000000807
Morselli PG, Giorgini FA, Pazzini C, Muscari C (2017) Lull pgm system: a suitable technique to improve the regenerative potential of autologous fat grafting. Wound Repair Regen 25:722–729
Eto H, Kato H, Suga H, Aoi N, Doi K, Kuno S, Yoshimura K (2012) The fate of adipocytes after nonvascularized fat grafting: evidence of early death and replacement of adipocytes. Plast Reconstr Surg 129:1081–1092
Zuk PA, Zhu M, Ashjian P, De Ugarte DA, Huang JI, Mizuno H, Alfonso ZC, Fraser JK, Benhaim P, Hedrick MH (2002) Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell 13:4279–4295
Muscari C, Bonafè F, Fiumana E, Oranges CM, Pinto V, Caldarera CM, Guarnieri C, Morselli PG (2013) Comparison between stem cells harvested from wet and dry lipoaspirates. Connect Tissue Res 54:34–40
Kim YK, Lee SK, Ha MS, Woo JS, Jung JS (2002) Differential role of reactive oxygen species in chemical hypoxia-induced cell injury in opossum kidney cells and rabbit renal cortical slices. Exp Nephrol 10:275–284
Chandrika BB, Yang C, Ou Y, Feng X, Muhoza D, Holmes AF, Theus S, Deshmukh S, Haun RS, Kaushal GP (2015) Endoplasmic reticulum stress-induced autophagy provides cytoprotection from chemical hypoxia and oxidant injury and ameliorates renal ischemia-reperfusion Injury. PLoS ONE. https://doi.org/10.1371/journal.pone.0140025
Halliwell B (2014) Cell culture, oxidative stress, and antioxidants: avoiding pitfalls. Biomed J 37:99–105
Maumus M, Peyrafitte JA, D’Angelo R, Fournier-Wirth C, Bouloumié A, Casteilla L, Sengenès C, Bourin P (2011) Native human adipose stromal cells: localization, morphology and phenotype. Int J Obes (Lond) 35:1141–1153
Savi M, Bocchi L, Fiumana E, Karam JP, Frati C, Bonafé F, Cavalli S, Morselli PG, Guarnieri C, Caldarera CM, Muscari C, Montero-Menei CN, Stilli D, Quaini F, Musso E (2015) Enhanced engraftment and repairing ability of human adipose-derived stem cells, conveyed by pharmacologically active microcarriers continuously releasing HGF and IGF-1, in healing myocardial infarction in rats. J Biomed Mater Res A 103:3012–3025
Matsuno-Yagi A, Hatefi Y (2001) Ubiquinol:cytochrome c oxidoreductase (complex III). Effect of inhibitors on cytochrome b reduction in submitochondrial particles and the role of ubiquinone in complex III. J Biol Chem 276:19006–19011
Piskernik C, Haindl S, Behling T, Gerald Z, Kehrer I, Redl H, Kozlov AV (2008) Antimycin A and lipopolysaccharide cause the leakage of superoxide radicals from rat liver mitochondria. Biochim Biophys Acta 1782:280–285
Muscari C, Giordano E, Bonafè F, Govoni M, Pasini A, Guarnieri C (2013) Priming adult stem cells by hypoxic pretreatments for applications in regenerative medicine. J Biomed Sci. https://doi.org/10.1186/1423-0127-20-63
Tchanque-Fossuo CN, Dahle SE, Buchman SR, Isseroff RR (2017) Deferoxamine: potential novel topical therapeutic for chronic wounds. Br J Dermatol 176:1056–1059
Choudhry H, Harris AL (2018) Advances in hypoxia-inducible factor biology. Cell Metab 27:281–298
Gutteridge JM, Richmond R, Halliwell B (1979) Inhibition of the iron-catalysed formation of hydroxyl radicals from superoxide and of lipid peroxidation by desferrioxamine. Biochem J 184:469–472
Hernansanz-Agustín P, Izquierdo-Álvarez A, Sánchez-Gómez FJ, Ramos E, Villa-Piña T, Lamas S, Bogdanova A, Martínez-Ruiz A (2014) Acute hypoxia produces a superoxide burst in cells. Free Radic Biol Med 71:146–156
Kong D, Park EJ, Stephen AG, Calvani M, Cardellina JH, Monks A, Fisher RJ, Shoemaker RH, Melillo G (2005) Echinomycin, a small-molecule inhibitor of hypoxia-inducible factor-1 DNA-binding activity. Cancer Res 65:9047–9055
Tsuzuki T, Okada H, Cho H, Tsuji S, Nishigaki A, Yasuda K, Kanzaki H (2012) Hypoxic stress simultaneously stimulates vascular endothelial growth factor via hypoxia-inducible factor-1α and inhibits stromal cell-derived factor-1 in human endometrial stromal cells. Hum Reprod 27:523–530
Hou Z, Nie C, Si Z, Ma Y (2013) Deferoxamine enhances neovascularization and accelerates wound healing in diabetic rats via the accumulation of hypoxia-inducible factor-1α. Diabetes Res Clin Pract 101:62–71
Temiz G, Sirinoglu H, Yesiloglu N, Filinte D, Kaçmaz C (2016) Effects of deferoxamine on fat graft survival. Facial Plast Surg 32:438–443
Flacco J, Chung N, Blackshear CP, Irizarry D, Momeni A, Lee GK, Nguyen D, Gurtner GC, Longaker MT, Wan DC (2018) Deferoxamine preconditioning of irradiated tissue improves perfusion and fat graft retention. Plast Reconstr Surg 141:655–665
Okyay MF, Kömürcü H, Bağhaki S, Demiröz A, Aydın Ö, Arslan H (2019) Effects of insulin, metoprolol and deferoxamine on fat graft survival. Aesthetic Plast Surg 43:845–852
Arslan H (2019) Invited response on: “several opinions on effects of insulin, metoprolol and deferoxamine on fat graft”. Aesthetic Plast Surg. https://doi.org/10.1007/s00266-019-01430-5
Coetzee WA (2013) Multiplicity of effectors of the cardioprotective agent, diazoxide. Pharmacol Ther 140:167–175
Prendes MG, Hermann R, Torresin ME, Vélez D, Savino EA, Varela A (2014) Role of mitochondrial permeability transition pore and mitochondrial ATP-sensitive potassium channels in the protective effects of ischemic preconditioning in isolated hearts from fed and fasted rats. J Physiol Biochem 70:791–800
Dröse S, Hanley PJ, Brandt U (2009) Ambivalent effects of diazoxide on mitochondrial ROS production at respiratory chain complexes I and III. Biochim Biophys Acta 1790:558–565
Yang F, Chen WL, Zheng MZ, Yu GW, Xu HJ, Shen YL, Chen YY (2011) Heat shock protein 90 mediates anti-apoptotic effect of diazoxide by preventing the cleavage of Bid in hypothermic preservation rat hearts. J Heart Lung Transplant 30:928–934
Suleiman MS, Singh RJ, Stewart CE (2007) Apoptosis and the cardiac action of insulin-like growth factor I. Pharmacol Ther 114:278–294
Laviola L, Natalicchio A, Giorgino F (2007) The IGF-I signaling pathway. Curr Pharm Des 13:663–669
Kontoghiorghes GJ, Pattichi K, Hadjigavriel M, Kolnagou A (2000) Transfusional iron overload and chelation therapy with deferoxamine and deferiprone (L1). Transfus Sci 23:211–223
Acknowledgements
Funding for this research was obtained from RFO (Ricerca Fondamentale Orientata) of the Department of Biomedical and Neuromotor Sciences (DIBINEM), University of Bologna.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
Written informed consent was obtained from all subjects.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Morselli, P.G., Sorbi, G., Feliziani, C. et al. Deferoxamine Protects Stromal/Stem Cells of “Lull pgm System”-Processed Lipoaspirates Against Damages Induced by Mitochondrial Respiration Inhibition. Aesth Plast Surg 44, 168–176 (2020). https://doi.org/10.1007/s00266-019-01544-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-019-01544-w