Abstract
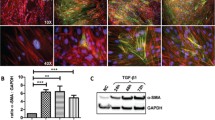
Myofibroblasts play a central role in matrix formation and wound contraction during wound healing and undergo apoptosis at the end of the healing. Hypertrophic scarring is a pathologic condition in which myofibroblasts persist in the tissue. It has been hypothesized that abnormalities in epidermal–dermal crosstalk underlie this pathology. Therefore, in this study, we investigated whether myofibroblasts are affected by keratinocytes. Transforming growth factor beta-induced myofibroblasts (Imyo) and myofibroblasts from hypertrophic scar tissue (Hmyo) were characterized using microarrays. Keratinocytes were co-cultured with myofibroblasts, and quantitative PCR analysis was performed. We found that numerous extracellular matrix- and smooth muscle cell-associated genes were upregulated in Imyo and Hmyo respectively, and these findings suggest that Hmyo are fully differentiated myofibroblasts and that Imyo are less differentiated than Hmyo. Decreased collagen type 1 gene expression was found in keratinocytes co-cultured with Imyo and Hmyo; further, α-smooth muscle actin expression in Imyo increased in the presence of keratinocytes. These observations indicate that keratinocytes play a role in the development of pathological fibrosis in hypertrophic scar tissue by regulating the behavior of dermal fibroblasts and myofibroblasts. We believe that this study provides the basis for understanding the pathophysiology of hypertrophic scarring and identifying new therapeutic approaches for this dysfunction.
No Level Assigned This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors -www.springer.com/00266.






Similar content being viewed by others
References
Van Loey NE, Bremer M, Faber AW et al (2008) Itching following burns: epidemiology and predictors. Br J Dermatol 158:95–100
Kischer CW, Wagner HN, Pindur J et al (1989) Increased fibronectin production by cell lines from hypertrophic scar and keloid. Connect Tissue Res 23:279–288
Gailit J, Marchese MJ, Kew RR et al (2001) The differentiation and function of myofibroblasts is regulated by mast cell mediators. J Invest Dermatol 117:1113–1119
Chen L, Liu S, Li SR et al (2006) Influence of substance P on the release of histamine in the human hypertrophic scar tissue. Zonghua Shao Shang Za Zhi 22:192–194
Niessen FB, Andreissen MP, Schalkwijk J et al (2001) Keratinocyte-derived growth factors play a role in the formation of hypertrophic scars. J Pathol 194:207–216
Ishida Y, Gao JL, Murphy PM (2008) Chemokine receptor CX3CR6 mediates skin wound healing by promoting macrophage and fibroblast accumulation and function. J Immunol 180:569–579
Wang R, Ghahary A, Shen Q et al (2000) Hypertrophic scar tissues and fibroblasts produce more transforming growth factor-beta1 mRNA and protein than normal skin and cells. Wound Repair Regen 8:128–137
Mori L, Bellini A, Stacey MA et al (2005) Fibrocytes contribute to the myofibroblast population in wounded skin ad originate from the bone marrow. Exp Cell Res 304:81–90
Yang L, Scott PG, Dodd C et al (2005) Identification of fibrocytes in postburn hypertrophic scar. Wound Repair Regen 13:398–404
Ueyama M, Maruyama I, Osame M et al (1992) Marked increase in plasma interleukin-6 in burn patients. J Lab Clin Med 120:693–698
Yeh FL, Lin WL, Shen HD et al (1999) Changes in circulating levels of interleukin 6 in burned patients. Burns 25:131–136
Ghahary A, Shen YJ, Nedelec B et al (1996) Collagenase production is lower in post-burn hypertrophic scar fibroblasts than in normal fibroblasts and is reduced by insulin-like growth factor-1. J Invest Dermatol 106:476–481
Moulin V, Larochells S, Langlois C et al (2004) Normal skin wound and hypertrophic scar myofibroblasts have differential responses to apoptotic inductors. J Cell Physiol 198:350–358
Gabbiani G (2003) The myofibroblast in wound healing and fibrocontractive disease. J Pathol 200:500–503
Rheinwald JG, Green H (1975) Serial cultivation of strains of human epidermal keratinocytes: the formation of keratinizing colonies from single cells. Cell 6:331–343
Rachel AE, Ya CT, Robert S et al (2003) TGF-β1-mediated fibroblast–myofibroblast terminal differentiation—the role of Smad proteins. Exp Cell Res. 282:90–100
Sime PJ, Xing Z, Graham FL et al (1997) Adenovector-mediated gene transfer of active transforming growth factor β1 induces prolonged severe fibrosis in rat lung. J Clin Invest 100:768–776
Verhaegen P, Zuijlen P, Pennings N et al (2009) Differentiation in collagen architecture between keloid, hypertrophic scar, normotrophic scar, and normal skin: an objective histopathological analysis. Wound Repair Regen 17:49–56
Yamamoto N, Nishioka S, Sasai Y (1995) Polarization microscopic investigation of collagen and acid glycosaminoglycans in the skin of progressive systemic sclerosis (PSS). Acta Histochem 97:195–202
De Vries HJ, Enomoto DN, van Marle J et al (2000) Dermal organization in scleroderma: the fast Fourier transform and the laser scatter method objectify fibrosis in nonlesional as well as lesional skin. Lab Invest 80:1281–1289
Holbrook KA, Byers PH, Pinnell SR (1982) The structure and function of dermal connective tissue in normal individuals and patients with inherited connective tissue disorders. Scan Electron Microsc (Pt 4):1731–1744
Ghahary A, Ghaffari A (2007) Role of keratinocyte-fibroblast cross-talk in development of hypertrophic scar. Wound Repair Regen 15:S46–S53
Machesney M, Tidman N, Waseem A et al (1998) Activated keratinocytes in the epidermis of hypertrophic scar. Am J Pathol 152:1133–1141
Hakvoort TE, Altun V, Ramrattan RS et al (1999) Epidermal participation in post-burn hypertrophic scar development. Virchows Arch 434:221–226
Butzelaar L, Ulrich MM, Mink van der Molen AB et al (2016) Currently known risk factors for hypertrophic skin scarring: a review. J Plast Reconstr Aesthet Surg 69(2):163–169
Martin P, Parkhurst SM (2004) Parallels between tissue repair and embryo morphogenesis. Development 131:3021–3034
Shepard P, Martin G, Smola-Hess S et al (2004) Myofibroblast differentiation in induced in keratinocyte-fibroblast co-cultures and is antagonistically regulated by endogenous transforming growth factor-β and interleukin-1. Am J Pathol 164:2055–2066
Werner S, Krieg T, Smolar H (2007) Keratinocyte-fibroblast interactions in wound healing. J Invest Dermatol 127:998–1008
Leask A, Abraham DJ (2004) TGF-beta signaling and the fibrotic response. FASEB J 18:816–827
Vaughan MB, Howard EW, Tomask JJ (2000) Transforming growth factor-beta1 promotes the morphological and functional differentiation of the myofibroblast. Exp Cell Res 257:180–189
Kane CJ, Hebda PA, Mansbridge JN et al (1991) Direct evidence for spatial and temporal regulation of transforming growth factor beta 1 expression during cutaneous wound healing. J Cell Physiol 148:157–173
Deitch EA, Wheelahan TM, Rose MP et al (1983) Hypertrophic burn scars: analysis of variables. J Trauma 23:895–898
Moulin V, Auger FA, Garrel D et al (2000) Role of wound healing myofibroblasts on re-epithelialization of human skin. Burns 26:3–12
Varkey M, Ding J, Tredget EE (2014) Fibrotic remodeling of tissue-engineered skin with deep dermal fibroblasts is reduced by keratinocytes. Tissue Eng A 20:716–727
Acknowledgements
This research was supported by National Research Funding granted by the Korean Government (NRF-2017R1A2B2007673).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest to disclose.
Statement of Animal Rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
For this type of study, informed consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lee, J.S., Kim, J.S., Lee, J.W. et al. Effect of Keratinocytes on Myofibroblasts in Hypertrophic Scars. Aesth Plast Surg 43, 1371–1380 (2019). https://doi.org/10.1007/s00266-019-01434-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-019-01434-1