Abstract
Introduction
The introduction of acellular dermal matrices (ADMs) contributed to the growing diffusion of direct-to-implant breast reconstruction (DTI-BR) following mastectomy for breast cancer. According to specific legislations, European specialists could not benefit from the use of human-derived ADMs, even though most evidence in the literature are available for this kind of device, showed optimal outcomes in breast reconstruction. The Skin Bank of the Bufalini Hospital (Cesena, Italy) obtained in 2009 the approval for the production and distribution of a new human cadaver-donor-derived ADM (named with the Italian acronym, MODA, for matrice omologa dermica acellulata) from the Italian National Transplant Center and National Health Institute. We report preliminary results of MODA application in direct-to-implant breast reconstruction following nipple–areola complex (NAC)-sparing mastectomy for breast cancer treatment.
Materials and Methods
We prospectively enrolled all women undergoing NAC-sparing mastectomy for breast cancer and DTI-BR in our breast surgical unit from June 2015 to January 2017. We enrolled a selected population without previous chest wall irradiation, not being heavy tobacco smokers or diabetic, with a BMI < 30 kg/m2 and requiring less than 550 cc silicone implants. We assessed short-term outcomes, defined as postoperative complications presenting in the first 30 postoperative days and long-term outcomes at 6 and 12 months.
Results
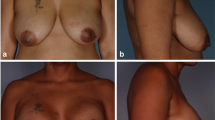
From June 2015 to January 2017, we treated 56 breasts. At a mean follow-up of 14 months, we observed only two minor complications described as limited wound dehiscences, conservatively managed with complete resolution without implant exposure or re-intervention.
Conclusions
Our preliminary results show very good performance of MODA in direct-to-implant breast reconstruction following NAC-sparing mastectomy for breast cancer treatment. This is particularly relevant for the European market, where no other human-derived devices are available for breast reconstruction due to regulatory restrictions.
Level of Evidence IV
This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.





Similar content being viewed by others
References
Baxter RA (2012) Current state of the art for acellular dermal matrices in breast surgery. Clin Plast Surg 39:9
Nahabedian MY (2012) Acellular dermal matrices in primary breast reconstruction: principles, concepts, and indications. Plast Reconstr Surg 130:44S–53S
Salzberg CA (2006) Nonexpansive immediate breast reconstruction using human acellular tissue matrix graft (AlloDerm). Ann Plast Surg 57:1–5
Spear SL, Parikh PM, Reisin E et al (2008) Acellular dermis-assisted breast reconstruction. Aesthet Plast Surg 32:418–425
Salzberg CA, Ashikari AY, Koch RM et al (2011) An 8-year experience of direct-to-implant immediate breast reconstruction using human acellular dermal matrix (AlloDerm). Plast Reconstr Surg 127(2):514–524
Topol BM, Dalton EF, Ponn T et al (2008) Immediate single-stage breast reconstruction using implants and human acellular dermal tissue matrix with adjustment of the lower pole of the breast to reduce unwanted lift. Ann Plast Surg 61(5):494499
Cabalag MS, Rostek M, Miller GS, Chae MP, Quinn T, Rozen WM, Hunter-Smith DJ (2016) Alloplastic adjuncts in breast reconstruction. Gland Surg 5(2):158–173
Salzberg CA (2012) Focus on technique: one-stage implant-based breast reconstruction. Plast Reconstr Surg 130:95S–103S
Jansen LA, Macadam SA (2011) The use of AlloDerm in postmastectomy alloplastic breast reconstruction: part I. A systematic review. Plast Reconstr Surg 127(6):2232–2244
Nahabedian MY (2016) Prosthetic breast reconstruction with acellular dermal matrices: achieving predictability and reproducibility. Plast Reconstr Surg Glob Open 4(5):e698
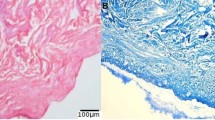
Bondioli E, Fini M, Veronesi F, Giavaresi G, Tschon M, Cenacchi G, Cerasoli S, Giardino R, Melandri D (2012) Development and evaluation of a decellularized membrane from human dermis. J Tissue Eng Regen Med 8:325–336
Fini M, Bondioli E, Castagna A, Torricelli P, Giavaresi G, Rotini R, Marinelli A, Guerra E, Orlandi C, Carboni A, Aiti A, Benedettini E, Giardino R, Melandri D (2012) Decellularized human dermis to treat massive rotator cuff tears: in vitro evaluations. Connect Tissue Res 53:298–306
Rotini R, Marinelli A, Guerra E, Bettelli G, Castagna A, Fini M, Bondioli E, Busacca M (2011) Human dermal matrix scaffold augmentation for large and massive rotator cuff repairs: preliminary clinical and MRI results at 1-year follow-up. Musculoskelet Surg 95(Suppl 1):S13–S23
Giavaresi G, Bondioli E, Melandri D, Giardino R, Tschon M, Torricelli P, Cenacchi G, Rotini R, Castagna A, Veronesi F, Pagani S, Fini M (2013) Response of human chondrocytes and mesenchymal stromal cells to a decellularized human dermis. BMC Musculoskelet Disord 14:12
Ghetti M, Bondioli E, Purpura V, Cenacchi G, Ruscelli P, Melandri D (2017) Decellularized human dermal matrix produced by a skin bank: a new treatment for abdominal wall defects. Ann Ital Chir 5:443–448
Gasperoni M, Neri R, Carboni A, Purpura V, Morselli PG, Melandri D (2016) The alexander surgical technique for the treatment of severe burns. Ann Burns Fire Disasters 29(4):281–285
Folli S, Curcio A, Buggi F, Mingozzi M, Lelli D, Barbieri C, Asioli S, Nava MB, Falcini F (2012) Improved sub-areolar breast tissue removal in nipple-sparing mastectomy using hydrodissection. Breast 21(2):190–193
Govshievich A, Somogyi RB, Brown MH (2015) Conservative mastectomies and immediate reconstruction with the use of ADMs. Gland Surg 4(6):453–462
Zenn M, Venturi M, Pittman T, Spear S, Gurtner G, Robb G, Mesbahi A, Dayan J (2017) Optimizing outcomes of postmastectomy breast reconstruction with acellular dermal matrix: a review of recent clinical data. Eplasty 17:156–171
Vu MM, Kim JY (2015) Current opinions on indications and algorithms for acellular dermal matrix use in primary prosthetic breast reconstruction. Gland Surg 4(3):195–203
Myckatyn TM, Cavallo JA, Sharma K et al (2015) The impact of chemotherapy and radiation therapy on the remodeling of acellular dermal matrices in staged, prosthetic breast reconstruction. Plast Reconstr Surg 135(1):43e–57e
Zenn MR, Salzberg CA (2016) A direct comparison of AlloDerm-ready to use (RTU) and DermACELL in immediate breast implant reconstruction. Eplasty 16:e23
Ricci JA, Treiser MD, Tao R et al (2016) Predictors of complications and comparison of outcomes using Surg-iMend fetal bovine and AlloDerm human cadaveric acellular dermal matrices in implant-based breast reconstruction. Plast Reconstr Surg 138(4):583e–591e
Selber JC, Wren JH, Garvey PB et al (2015) Critical evaluation of risk factors and early complications in 564 consecutive two-stage implant-based breast reconstructions using acellular dermal matrix at a single center. Plast Reconstr Surg 136(1):10–20
Lee KT, Mun GH (2016) Updated evidence of acellular dermal matrix use for implant-based breast reconstruction: a meta-analysis. Ann Surg Oncol 23(2):600–610
Pittman TA, Fan KL, Knapp A, Frantz S, Spear SL (2016) Comparison of different acellular dermal matrix (ADM) in breast reconstruction: the 50/50 study. Plast Reconstr Surg. https://doi.org/10.1097/PRS.0000000000003048
Palaia DA, Arthur KS, Cahan AC, Rosenberg MH (2015) Incidence of seromas and infections using fenestrated versus nonfenestrated acellular dermal matrix in breast reconstructions. Plast Reconstr Surg Glob Open 3(11):e569
Glasberg SB, Light D (2012) AlloDerm and Strattice in breast reconstruction: a comparison and techniques for optimizing outcomes. Plast Reconstr Surg 129(6):1223–1233
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest to disclose.
Rights and permissions
About this article
Cite this article
Folli, S., Curcio, A., Melandri, D. et al. A New Human-Derived Acellular Dermal Matrix for Breast Reconstruction Available for the European Market: Preliminary Results. Aesth Plast Surg 42, 434–441 (2018). https://doi.org/10.1007/s00266-017-1069-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-017-1069-7