Abstract
Background
A new commercial drug that contains autologous cultured fibroblasts has been developed and approved by the United States Food and Drug Administration for improving the appearance of nasolabial folds. However, the treatment requires three sessions every 3–6 weeks. It is known that the skin overlying the nasojugal groove is thinner, and the wrinkle is generally shallower than nasolabial folds. Therefore, we hypothesized that the nasojugal groove could be improved by just one treatment session. Therefore, the purpose of this study was to evaluate the efficacy and safety of autologous cultured fibroblast injection to correct nasojugal grooves.
Methods
Forty-six subjects with nasojugal grooves were enrolled in this study. They were injected with autologous cultured fibroblasts or placebo in one session. Blinded evaluators and subjects assessed the efficacy using a validated wrinkle assessment scale at 4, 12, and 24 weeks after the injection. Information of adverse events was collected at each visit.
Results
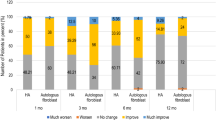
Based on the evaluators’ assessment at 24 weeks after the injection, 76% of subjects treated with autologous cultured fibroblasts showed improvement whereas 0% of subjects treated with placebo showed improvement (P < 0.0001). Based on self-assessment at 24 weeks after the injection, 72% of subjects treated with autologous cultured fibroblasts and 45% of subjects treated with placebo showed improvement (P = 0.0662). There were no serious adverse events related to autologous cultured fibroblast injection.
Conclusions
Autologous cultured fibroblast injection might be effective and safe to correct nasojugal grooves.
Level of Evidence I
This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.




Similar content being viewed by others
References
Jiang J, Wang X, Chen R, Xia X, Sun S, Hu K (2016) Tear trough deformity: different types of anatomy and treatment options. Postepy Dermatol Alergol 33(4):303–308
Knapp TR, Kaplan EN, Daniels JR (1977) Injectable collagen for soft tissue augmentation. Plast Reconstr Surg 60(3):398–405
Garfein ES, Orgill DP, Pribaz JJ (2003) Clinical applications of tissue engineered constructs. Clin Plast Surg 30(4):485–498
Duranti F, Salti G, Bovani B, Calandra M, Rosati ML (1998) Injectable hyaluronic acid gel for soft tissue augmentation. A clinical and histological study. Dermatol Surg 24(12):1317–1325
Alster TS, West TB (2000) Human-derived and new synthetic injectable materials for soft-tissue augmentation: current status and role in cosmetic surgery. Plast Reconstr Surg 105(7):2515–2525 (discussion 2526-8)
Smith SR, Jones D, Thomas JA, Murphy DK, Beddingfield FC 3rd (2010) Duration of wrinkle correction following repeat treatment with Juvederm hyaluronic acid fillers. Arch Dermatol Res 302(10):757–762
Lowe NJ, Lowe PL, St Clair Roberts J (2010) A phase IIa open-label dose-escalation pilot study using allogeneic human dermal fibroblasts for nasolabial folds. Dermatol Surg 36(10):1578–1585
Watson D, Keller GS, Lacombe V, Fodor PB, Rawnsley J, Lask GP (1999) Autologous fibroblasts for treatment of facial rhytids and dermal depressions. A pilot study. Arch Facial Plast Surg 1(3):165–170
Weiss RA, Weiss MA, Beasley KL, Munavalli G (2007) Autologous cultured fibroblast injection for facial contour deformities: a prospective, placebo-controlled, Phase III clinical trial. Dermatol Surg 33(3):263–268
Boss WK Jr, Usal H, Chernoff G, Keller GS, Lask GP, Fodor PB (2000) Autologous cultured fibroblasts as cellular therapy in plastic surgery. Clin Plast Surg 27(4):613–626
Martin I, Baldomero H, Bocelli-Tyndall C, Slaper-Cortenbach I, Passweg J, Tyndall A (2011) The survey on cellular and engineered tissue therapies in Europe in 2009. Tissue Eng Part A 17(17–18):2221–2230
Schmidt C (2011) FDA approves first cell therapy for wrinkle-free visage. Nat Biotechnol 29(8):674–675
Smith SR, Munavalli G, Weiss R, Maslowski JM, Hennegan KP, Novak JM (2012) A multicenter, double-blind, placebo-controlled trial of autologous fibroblast therapy for the treatment of nasolabial fold wrinkles. Dermatol Surg 38(7 Pt 2):1234–1243
Breslin L, Prosser SL, Cuffe S, Morrison CG (2014) Ciliary abnormalities in senescent human fibroblasts impair proliferative capacity. Cell Cycle 13(17):2773–2779
Kudlow JE, Khosravi MJ, Kobrin MS, Mak WW (1984) Inability of anti-epidermal growth factor receptor monoclonal antibody to block “autocrine” growth stimulation in transforming growth factor-secreting melanoma cells. J Biol Chem 259(19):11895–11900
Mazlyzam AL, Aminuddin BS, Saim L, Ruszymah BH (2008) Human serum is an advantageous supplement for human dermal fibroblast expansion: clinical implications for tissue engineering of skin. Arch Med Res 39(8):743–752
Benny P, Badowski C, Lane EB, Raghunath M (2015) Making more matrix: enhancing the deposition of dermal-epidermal junction components in vitro and accelerating organotypic skin culture development, using macromolecular crowding. Tissue Eng Part A 21(1–2):183–192
Narins RS, Brandt F, Leyden J, Lorenc ZP, Rubin M, Smith S (2003) A randomized, double-blind, multicenter comparison of the efficacy and tolerability of Restylane versus Zyplast for the correction of nasolabial folds. Dermatol Surg 29(6):588–595
Flowers RS (1993) Tear trough implants for correction of tear trough deformity. Clin Plast Surg 20(2):403–415
Loeb R (1993) Naso-jugal groove leveling with fat tissue. Clin Plast Surg 20(2):393–400 (discussion 401)
Goldberg RA, McCann JD, Fiaschetti D, Ben Simon GJ (2005) What causes eyelid bags? Analysis of 114 consecutive patients. Plast Reconstr Surg 115(5):1395–1402 (discussion 1403-4)
Loeb R (1981) Fat pad sliding and fat grafting for leveling lid depressions. Clin Plast Surg 8(4):757–776
Stutman RL, Codner MA (2012) Tear trough deformity: review of anatomy and treatment options. Aesthet Surg J 32(4):426–440
Atiyeh BS, Hayek SN (2004) Combined arcus marginalis release, preseptal orbicularis muscle sling, and SOOF plication for midfacial rejuvenation. Aesthetic Plast Surg 28(4):197–202
Barton FE Jr, Ha R, Awada M (2004) Fat extrusion and septal reset in patients with the tear trough triad: a critical appraisal. Plast Reconstr Surg 113(7):2115–2121 (discussion 2122-3)
Seiff SR (2002) The fat pearl graft in ophthalmic plastic surgery: everyone wants to be a donor! Orbit 21(2):105–109
Goldberg RA, Edelstein C, Shorr N (1999) Fat repositioning in lower blepharoplasty to maintain infraorbital rim contour. Facial Plast Surg 15(3):225–229
Hanke CW, Higley HR, Jolivette DM, Swanson NA, Stegman SJ (1991) Abscess formation and local necrosis after treatment with Zyderm or Zyplast collagen implant. J Am Acad Dermatol 25(2 Pt 1):319–326
Swanson NA, Stoner JG, Siegle RJ, Solomon AR (1983) Treatment site reactions to Zyderm Collagen Implantation. J Dermatol Surg Oncol 9(5):377–380
Sklar JA, White SM (2004) Radiance FN: a new soft tissue filler. Dermatol Surg 30(5):764–768 (discussion 768)
Glavas IP (2005) Filling agents. Ophthalmol Clin North Am 18(2):249–57, v–vi
Bosniak S, Cantisano-Zilkha M, Glavas IP (2004) Nonanimal stabilized hyaluronic acid for lip augmentation and facial rhytid ablation. Arch Facial Plast Surg 6(6):379–383
Bosniak SL, Cantisano-Zilkha M (2005) Minimally invasive techniques of oculofacial rejuvenation. Thieme xvi, New York, p 183
Kane MA (2005) Treatment of tear trough deformity and lower lid bowing with injectable hyaluronic acid. Aesthetic Plast Surg 29(5):363–367
Wong T, McGrath JA, Navsaria H (2007) The role of fibroblasts in tissue engineering and regeneration. Br J Dermatol 156(6):1149–1155
Cohen JL (2008) Understanding, avoiding, and managing dermal filler complications. Dermatol Surg 34(Suppl 1):S92–S99
Hirsch RJ, Stier M (2008) Complications of soft tissue augmentation. J Drugs Dermatol 7(9):841–845
Smith KC (2008) Reversible vs. nonreversible fillers in facial aesthetics: concerns and considerations. Dermatol Online J 14(8):3
Requena L, Requena C, Christensen L, Zimmermann US, Kutzner H, Cerroni L (2011) Adverse reactions to injectable soft tissue fillers. J Am Acad Dermatol 64(1):1–34 (quiz 35-6)
Van Dorp AG, Verhoeven MC, Koerten HK, Der Nat-Van Van, Der Meij TH, Van Blitterswijk CA, Ponec M (1998) Dermal regeneration in full-thickness wounds in Yucatan miniature pigs using a biodegradable copolymer. Wound Repair Regen 6(6):556–568
Lamme EN, Van Leeuwen RT, Brandsma K, Van Marle J, Middelkoop E (2000) Higher numbers of autologous fibroblasts in an artificial dermal substitute improve tissue regeneration and modulate scar tissue formation. J Pathol 190(5):595–603
Krejci NC, Cuono CB, Langdon RC, McGuire J (1991) In vitro reconstitution of skin: fibroblasts facilitate keratinocyte growth and differentiation on acellular reticular dermis. J Invest Dermatol 97(5):843–848
Spiekstra SW, Breetveld M, Rustemeyer T, Scheper RJ, Gibbs S (2007) Wound-healing factors secreted by epidermal keratinocytes and dermal fibroblasts in skin substitutes. Wound Repair Regen 15(5):708–717
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Moon, KC., Lee, HS., Han, SK. et al. Correcting Nasojugal Groove with Autologous Cultured Fibroblast Injection: A Pilot Study. Aesth Plast Surg 42, 815–824 (2018). https://doi.org/10.1007/s00266-017-1044-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-017-1044-3