Abstract
Background
Capsular contracture remains the most frequent long-term complication after augmentation mammoplasty with silicone implants. Thereby, the main part of the fibrotic capsule is collagen. The collagenase of the bacterium Clostridium histolyticum is approved for the treatment of fibrotic diseases and has been demonstrated to be effective for capsular fibrosis treatment in the short term. However, long-term effectiveness is currently unknown but mandatory for clinical utilization.
Materials and Methods
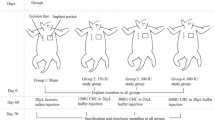
Forty-eight rats received miniature silicone implants and an injection with either collagenase (treatment group) or plain solvent solution (control group) 120 days post insertion. Ten and 60 days after the injections, the rats underwent 7-Tesla magnetic resonance imaging (MRI) and high-resolution ultrasound (HR-US). Capsule tissue was harvested, and capsule thickness and collagen density were evaluated through histology. Furthermore, the expression levels of inflammatory (CD68, IL4, IL10, IL12, IL13), pro-, and anti-fibrotic (TGFb1, TGFb3, Smad3, Col1-4) genes were analyzed using qRT-PCR.
Results
On days 10 and 60 after injection of collagenase, histology showed that capsule thickness was significantly reduced in the treatment group when compared with the control (p < 0.05). Thickness measurements were verified by MRI and HR-US analysis. Skin perforation occurred in two cases after collagenase injection. The initial up-regulation of pro-fibrotic and inflammatory genes 10 days after collagenase injection did not persist in the long term. Contrarily, on day 60, a slight trend towards lower expression levels with a significant down-regulation of TGFb3 was detected in the treatment group.
Conclusion
The collagenase of the bacterium C. histolyticum effectively degrades capsular fibrosis around silicone implants with stable outcomes throughout 60 days post injection. Skin perforation and adequate and uniform drug distribution within the implant pocket are issues that need to be addressed. Further studies are warranted to clarify whether collagenase injections have the potential to become a viable treatment option for capsular contracture.
No Level Assigned
This journal requires that authors 46 assign a level of evidence to each article. For a full 47 description of these Evidence-Based Medicine ratings, 48 please refer to the Table of Contents or the online 49 Instructions to Authors. www.springer.com/00266.








Similar content being viewed by others
References
American Society of Plastic Surgeons (2013) Complete plastic surgery statistics report
Handel N, Cordray T, Gutierrez J, Jensen JA (2006) A long-term study of outcomes, complications, and patient satisfaction with breast implants. Plast Reconstr Surg 117:757–767 discussion 768–772
Araco A, Caruso R, Araco F, Overton J, Gravante G (2009) Capsular contractures: a systematic review. Plast Reconstr Surg 124:1808–1819
Rosato RM, Dowden RV (1994) Radiation therapy as a cause of capsular contracture. Ann Plast Surg 32:342–345
Anderson JM, Rodriguez A, Chang DT (2008) Foreign body reaction to biomaterials. Semin Immunol 20:86–100
Wilflingseder P, Hoinkes G, Mikuz G (1983) Tissue reactions from silicone implant in augmentation mammaplasties. Minerva Chir 38:877–880
Tamboto H, Vickery K, Deva AK (2010) Subclinical (biofilm) infection causes capsular contracture in a porcine model following augmentation mammaplasty. Plast Reconstr Surg 126:835–842
Wick G, Backovic A, Rabensteiner E, Plank N, Schwentner C, Sgonc R (2010) The immunology of fibrosis: innate and adaptive responses. Trends Immunol 31:110–119
Gurunluoglu R, Sacak B, Arton J (2013) Outcomes analysis of patients undergoing autoaugmentation after breast implant removal. Plast Reconstr Surg 132:304–315
Young VL (1998) Guidelines and indications for breast implant capsulectomy. Plast Reconstr Surg 102:884–891 discussion 892–884
Collis N, Sharpe DT (2000) Recurrence of subglandular breast implant capsular contracture: anterior versus total capsulectomy. Plast Reconstr Surg 106:792–797
Graf R, Ascenco AS, Freitas Rda S, Balbinot P, Peressutti C, Costa DF, Dos Santos Fde H, Ratti MA, Kulchetscki RM (2015) Prevention of capsular contracture using leukotriene antagonists. Plast Reconstr Surg 136:592e–596e
Gilpin D, Coleman S, Hall S, Houston A, Karrasch J, Jones N (2010) Injectable collagenase Clostridium histolyticum: a new nonsurgical treatment for Dupuytren’s disease. J Hand Surg Am 35(2027–2038):e2021
Hammoudeh ZS (2014) Collagenase Clostridium histolyticum injection for plantar fibromatosis (Ledderhose disease). Plast Reconstr Surg 134:497e–498e
Hay DC, Louie DL, Earp BE, Kaplan FT, Akelman E, Blazar PE (2014) Surgical findings in the treatment of Dupuytren’s disease after initial treatment with clostridial collagenase (Xiaflex). J Hand Surg Eur 39:463–465
Freshwater MF (2012) What were the adverse events for Dupuytren’s patients treated with Xiaflex who had contractures less than 20 degrees? Hand 7:348–349
Levine LA, Cuzin B, Mark S, Gelbard MK, Jones NA, Liu G, Kaufman GJ, Tursi JP, Ralph DJ (2015) Clinical safety and effectiveness of collagenase Clostridium histolyticum injection in patients with Peyronie’s disease: a phase 3 open-label study. J Sex Med 12:248–258
Holzer LA, Holzer G (2009) Injectable collagenase Clostridium histolyticum for Dupuytren’s contracture. N Engl J Med 361:2579–2580
Fischer S, Hirsch T, Diehm Y, Kiefer J, Bueno EM, Kueckelhaus M, Kremer T, Hirche C, Kneser U, Pomahac B (2015) The collagenase of the bacterium Clostridium histolyticum for the treatment of capsular fibrosis after silicone implants. Plast Reconstr Surg 136:981–989
Fischer S, Hirche C, Reichenberger MA, Kiefer J, Diehm Y, Mukundan S, Alhefzi M, Bueno EM, Kneser U, Pomahac B (2015) Silicone implants with smooth surfaces induce thinner but denser fibrotic capsules compared to those with textured surfaces in a rodent model. PLoS ONE 10:e0132131
Syed F, Thomas AN, Singh S, Kolluru V, Emeigh Hart SG, Bayat A (2012) In vitro study of novel collagenase (XIAFLEX®) on Dupuytren’s disease fibroblasts displays unique drug related properties. PLoS ONE 7:e31430
Brazzelli M, Cruickshank M, Tassie E, McNamee P, Robertson C, Elders A, Fraser C, Hernandez R, Lawrie D, Ramsay C (2015) Collagenase Clostridium histolyticum for the treatment of Dupuytren’s contracture: systematic review and economic evaluation. Health Technol Assess 19:1–202
Sangkum P, Yafi FA, Kim H, Bouljihad M, Ranjan M, Datta A, Mandava SH, Sikka SC, Abdel-Mageed AB, Moparty K, Hellstrom WJG (2015) Collagenase Clostridium histolyticum (Xiaflex) for the treatment of urethral Stricture disease in a rat model of urethral fibrosis. Urology 86(647):e641
Gelbard M, Lipshultz LI, Tursi J, Smith T, Kaufman G, Levine LA (2012) Phase 2b study of the clinical efficacy and safety of collagenase Clostridium histolyticum in patients with Peyronie disease. J Urol 187:2268–2274
Gelbard M, Goldstein I, Hellstrom WJG, McMahon CG, Smith T, Tursi J, Jones N, Kaufman GJ, Carson CC (2013) Clinical efficacy, safety and tolerability of collagenase Clostridium histolyticum for the treatment of peyronie disease in 2 large double-blind, randomized, placebo controlled phase 3 studies. J Urol 190:199–207
Levine LA, Larsen SM (2015) Surgical correction of persistent Peyronie’s disease following collagenase Clostridium histolyticum treatment. J Sex Med 12:259–264
Thomas A, Bayat A (2010) The emerging role of Clostridium histolyticum collagenase in the treatment of Dupuytren disease. Ther Clin Risk Manag 6:557–572
Badalamente MA, Hurst LC, Hentz VR (2002) Collagen as a clinical target: nonoperative treatment of Dupuytren’s disease. J Hand Surg Am 27:788–798
Hurst LC, Badalamente MA, Hentz VR, Hotchkiss RN, Kaplan FTD, Meals RA, Smith TM, Rodzvilla J, Group CIS (2009) Injectable collagenase Clostridium histolyticum for Dupuytren’s contracture. N Engl J Med 361:968–979
Badalamente MA, Hurst LC (2007) Efficacy and safety of injectable mixed collagenase subtypes in the treatment of Dupuytren’s contracture. J Hand Surg Am 32:767–774
Sengupta P (2013) The laboratory rat: relating its age with human’s. Int J Prev Med 4:624–630
Leask A, Abraham DJ (2004) TGF-beta signaling and the fibrotic response. FASEB J 18:816–827
Murata H, Zhou L, Ochoa S, Hasan A, Badiavas E, Falanga V (1997) TGF-beta3 stimulates and regulates collagen synthesis through TGF-beta1-dependent and independent mechanisms. J Invest Dermatol 108:258–262
Peimer CA, Wilbrand S, Gerber RA, Chapman D, Szczypa PP (2015) Safety and tolerability of collagenase Clostridium histolyticum and fasciectomy for Dupuytren’s contracture. J Hand Surg Eur 40:141–149
Gelbard MK, Walsh R, Kaufman JJ (1982) Collagenase for Peyronie’s disease experimental studies. Urol Res 10:135–140
Badalamente MA, Hurst LC (1996) Enzyme injection as a nonoperative treatment for Dupuytren’s disease. Drug Delivery 3:35–40
Peak TC, Mitchell GC, Yafi FA, Hellstrom WJ (2015) Role of collagenase Clostridium histolyticum in Peyronie’s disease. Biologics 9:107–116
Acknowledgments
SF received financial support from the German Research Foundation for this study (GZ: FI 2003/1-1). BP and SF received an Educational Research Grant from Auxilium Pharmaceuticals, Inc. (BWH-381) to conduct the study. All other authors have no financial interest in any of the products, devices, or drugs mentioned in this article. We thank Polytech (Dieburg, Germany) for the silicone implants.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fischer, S., Diehm, Y., Henzler, T. et al. Long-Term Effects of the Collagenase of the Bacterium Clostridium histolyticum for the Treatment of Capsular Fibrosis After Silicone Implants. Aesth Plast Surg 41, 211–220 (2017). https://doi.org/10.1007/s00266-016-0724-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-016-0724-8