Abstract
Background
The late-stage clinical course, histopathologic analysis, and surgical salvage procedures after eventful cosmetic injection of hydroxyethylmethacrylate particles suspended in hyaluronic acid (DermaLive, marketed through Dermatech, Paris, France and Novamedical GmbH, Langenfeld, Germany) have been studied. The study was designed to present modified treatment guidelines, report results, and draw conclusions regarding the future management of patients with late-stage DermaLive complications.
Methods
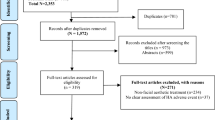
The study consulted and reviewed 21 patients with late-stage complications after facial implantation of DermaLive. Histology samples from excisional biopsies followed by radical surgical excision procedures were obtained and analyzed for nine patients.
Results
The treated zones included nasolabial folds, forehead and glabella, lips, and the perioral and zygoma regions. The late-onset period between injection and first symptoms averaged 17.5 months. Lesions were mostly symptomatic, with discomfort, pain, swelling, and edema. Clinical aspects included bulging, palpable nodules, erythema, and scar formation. A positive correlation between patient age and the delay period was found. All surgically treated patients recovered well. The initial conservative measures achieved only partial success, whereas all nine surgically treated patients (43 %) demonstrated complete relief of their clinical symptoms.
Conclusions
Soft tissue augmentation with DermaLive led to a series of severe delayed adverse reactions. Sclerosing DermaLive granulomas demonstrated low success rates with conservative measures, whereas early radical surgical excision achieved good results.
Level of Evidence IV
This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.








Similar content being viewed by others
References
Allen O (1992) Response to subdermal implantation of textured microimplants in humans. Aesthet Plast Surg 16:227–230
Angus JE, Affleck AG, Leach IH, Millard LG (2006) Two cases of delayed granulomatous reactions to the cosmetic filler Dermalive®, a hyaluronic acid and acrylic hydrogel. Br J Dermatol 155:1077–1078
Bergeret-Galley C, Latouche X, Ilouz Y-G (2001) The value of a new filler material in corrective and cosmetic surgery: DermaLive and DermaDeep. Aesthet Plast Surg 25:249–255
Bigata X, Ribera M, Bielsa I, Ferrándiz C (2001) Adverse granulomatous reaction after cosmetic dermal silicone injection. Dermatol Surg 27:198–200
Breiting V, Aasted A, Jørgensen A, Opitz P, Rosetzsky A (2004) A study on patients treated with polyacrylamid hydrogel injection for facial corrections. Aesthet Plast Surg 28:45–53
Carroll LA, Hanasono MM, Mikulec AA, Kita M, Koch RJ (2002) Triamcinolone stimulates bFGF production and inhibits TGF-b1 production by human dermal fibroblasts. Dermatol Surg 28:704–709
Christensen LC, Breiting VB, Aasted A, Jørgensen A, Kebuladze I (2003) Long-term effects of polyacrylamid hydrogel (PAAG, Interfall/Contura SA) in human breast tissue. Plast Reconstr Surg 111:1883–1890
Christensen L, Breiting V, Janssen M, Vuust J, Hogdall E (2005) Adverse reactions to injectable soft tissue permanent fillers. Aesthet Plast Surg 29:34–48
Cramer JE (1964) Intralesional injection of triamcinolone. Australas J Dermatol 13:140–147
Darouiche RO (2004) Treatment of infections associated with surgical implants. N Engl J Med 350:1422–1429
De Bree R, Middelweerd MJ, van der Waal I (2004) Severe granulomatous inflammatory response induced by injection of polyacrylamide gel into facial tissue. Arch Facial Plast Surg 6:204–206
Duffy DM (2005) Tissue injectable liquid silicone: new perspectives. In: Klein AW (ed) Tissue augmentation in clinical practice: procedures and techniques, 2nd edn. Taylor & Francis, New York
Duranti F, Salti G, Bovani B, Calandra M, Rosati ML (1998) Injectable hyaluronic acid for soft tissue augmentation: a clinical and histological study. Dermatol Surg 24:1317–1325
Ellis DA, Makdessian AS, Brown DJ (2001) Facial survey of future injectables. Plast Surg Clin N Am 9:405–411
Gelfer A, Carruthers A, Carruthers J, Jang F, Bernstein SC (2007) The natural history of polymethylmethacrylate microspheres granulomas. Dermatol Surg 33:614–620
Hoenig JF, Brink U, Korabiowska M (2003) Severe granulomatous allergic tissue reaction after hyaluronic acid injection in the treatment of facial lines and its surgical correction. J Craniofac Surg 14:197–200
Jones DH, Carruthers A, Orentreich D, Brody HJ, Lai MY, Azen S, Van Dyke GS (2004) Highly purified 1,000-cSt silicone oil for treatment of human immunodeficiency virus-associated facial lipoatrophy: an open pilot trial. Dermatol Surg 30:1279–1286
Lemperle G, Gauthier-Hazan N (2009) Foreign-body granulomas after all injectable dermal fillers: part 2. Treatment options. Plast Reconstr Surg 123:1864–1873
Lemperle G, Gauthier-Hazan N, Wolters M, Eisenmann-Klein M, Zimmermann U, Duffy DM (2009) Foreign-body granulomas after all injectable dermal fillers: part 1. Possible causes. Plast Reconstr Surg 123:1842–1863
Lemperle G, Hazan-Gauthier N, Lemperle M (1998) PMMA microspheres (Artecoll) for long-lasting correction of wrinkles: part III. Refinements and statistics. Aesthet Plast Surg 22:356–365
Lemperle G, Hazan-Gauthier N, Lemperle M (2003) PMMA microspheres (Artecoll) for skin and soft tissue augmentation: part II. Clinical investigations. Plast Reconstr Surg 96:627–634
Lemperle G, Morhenn VB, Charrier U (2003) Human histology and persistence of various injectable filler substances for soft tissue augmentation. Aesthet Plast Surg 27:354–366
Lemperle G, Romano JJ, Busso M (2003) Soft tissue augmentation with Artecoll: 10-year history, indications, techniques, and complications. Dermatol Surg 29:573–587
Lemperle G, Rullan PP, Gauthier-Hazan N (2006) Avoiding and treating dermal filler complications. Plast Reconstr Surg 118(Suppl):92S–106S
Lombardi T, Samson J, Plantier F, Husson C, Küffer R (2004) Orofacial granulomas after injection of cosmetic fillers: histopathologic and clinical study of 11 cases. J Oral Pathol Med 33:115–120
Lupton JR, Alster TS (2000) Cutaneous hypersensitivity reaction to injectable hyaluronic acid. Dermatol Surg 26:135–137
Maas CS, Papel ID, Greene D, Stoker DA (1997) Complications of injectable synthetic polymers in facial augmentation. Dermatol Surg 23:871–877
Moscona RR, Bergman R, Friedman-Birnbaum R (1993) An unusual late reaction to Zyderm I injections: a challenge for treatment. Plast Reconstr Surg 92:331–334
Myers E, Shea CR, Krausz T, Petronic-Rosic V (2007) Eosinophilic granuloma of the skin: response to intralesional corticosteroid injection. J Drugs Dermatol 6:329–332
Orentreich DS (2000) Liquid injectable silicone: techniques for soft tissue augmentation. Clin Plast Surg 27:595–612
Pearl RM, Laub DR, Kaplan DN (1978) Complications following silicone injections for augmentation of the contour of the face. Plast Reconstr Surg 71:251–253
Rapaport MJ, Vinnik C, Zarem H (1996) Injectable silicone: cause of facial nodules, cellulites, ulceration, and migration. Aesthet Plast Surg 20:267–276
Requena C, Izquierdo MJ, Navarro M, Martínez A, Vilata JJ, Botella R, Amorrortu J, Sabater V, Aliaga A, Requena L (2001) Adverse reactions to injectable aesthetic microimplants. Am J Dermatopathol 23:197–202
Rudolph CM, Soyer HP, Schuller-Petrovic S, Kerl H (1999) Foreign-body granuloma due to injectable aesthetic microimplants. Am J Surg Pathol 23:113–117
Shapiro PE (1997) Noninfectious granulomas. In: Elder D, Elanitsas R, Jaworsky C, Johnson B (eds) Lever’s histopathology of the skin, 8th edn. Lippincott Raven, Philadelphia
Tang L, Eaton JW (1995) Inflammatory responses to biomaterials. Am J Clin Pathol 103:466–471
Vargas-Machuca I, Gonzalez-Guerra E, Angulo J, del Carmen Farina M, Martin L, Requena L (2006) Facial granulomas secondary to DermaLive microimplants: report of a case with histopathologic differential diagnosis among the granulomas secondary to different injectable permanent filler materials. Am J Dermatopathol 28:173–177
Webster RC, Gaunt JM, Hamdan US, Fuleihan NS, Smith RC (1986) Injectable silicone for facial soft-tissue augmentation. Arch Otolaryngol Head Neck Surg 112:290–296
Wilkie TF (1977) Late development of granuloma after liquid silicone injections. Plast Reconstr Surg 60:179–188
Williams GT, Williams WJ (1983) Granulomatous inflammation: a review. J Clin Pathol 36:723–733
Zeller JM (1983) Surgical implants: physiological response. AORN J 37:1284–1291
Zielke H, Wölber L, Wiest L, Rzany B (2008) Risk profiles of different injectable fillers: results from the Injectable Filler Safety Study (IFS Study). Dermatol Surg 34:326–335
Zimmermann US, Clerici TJ (2004) The histological aspects of filler complications. Semin Cutan Med Surg 23:241–250
Acknowledgments
The authors (E. Demir, A. Perez-Bouza, N. Pallua) are not employees, consultants, agents or representatives of Dermatech which is the manufacturer of DermaLive.
Conflict of interest
The authors declare that they have no conflict of interest. None of the authors have a financial interest in any of the products, devices or drugs mentioned in this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Demir, E., Perez-Bouza, A. & Pallua, N. Adverse Late Reactions After Cosmetic Implantation of Hydroxyethylmethacrylate Particles Suspended in Hyaluronic Acid: Clinics and Complication Management. Aesth Plast Surg 37, 576–586 (2013). https://doi.org/10.1007/s00266-013-0107-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-013-0107-3