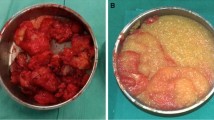
Since 1997, the hydrophilic polyacrylamide gel (PAAG), an injectable alloplastic biomaterial, imported from the Ukraine has been used for augmentation mammaplasty in China. There were twelve patients with various complications visiting our hospitals after breast augmentation with injected hydrophilic polyacrylamide gel by other clinics, even though such procedures are not performed in our clinical practices. The complications included four cases of PAAG-induced multiple induration and lumps, and one with lactation, three cases of hematoma, two cases of inflammation (infection), and persistent mastodymia, unsatisfactory contour results and abnormal skin sensations in each case. All patients have been treated by removing the injected gel and giving antibiotics. Other procedures included replacement by silicon breast prostheses, mammotomy, and segmental mastectomy. We suggest that the injectable hydrophilic polyacrylamide gel be contraindications for breast augmentation in any young female who is not married, or without a child, with a tendency to hemorrhage, and with little mammary tissue. Some considerations to prevent and manage these complications are discussed.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cheng, Nx., Wang, Yl., Wang, Jh. et al. Complications of Breast Augmentation with Injected Hydrophilic Polyacrylamide Gel . Aesth. Plast. Surg. 26, 375–382 (2002). https://doi.org/10.1007/s00266-002-2052-4
Issue Date:
DOI: https://doi.org/10.1007/s00266-002-2052-4