Abstract
In many social hymenopterans, workers of different ages engage in different tasks; younger workers remain inside the nest as intranidal workers, while older workers go outside the nest as extranidal workers (i.e., age polyethism). Previous studies have shown that ovarian activity is diminished in old, extranidal workers, but it remains unclear whether workers’ reproductive ability persists for life or whether they exhibit post-reproductive lifespans. In this study, we investigated the age-dependence of worker reproductive ability in a monomorphic ponerine ant Diacamma cf. indicum. In Diacamma ants, all females in a colony have reproductive ability, but effective reproduction is limited to one or a few dominant females, and the remaining females act as sterile helpers. Using long-term laboratory rearing, we investigated whether worker reproductive ability lasts throughout a worker’s lifetime. The ages of workers were accurately tracked, and the reproductive ability of young and old workers was examined by creating several gamergate-less sub-colonies. Results showed that at least one individual in each sub-colony developed ovaries, even in the sub-colonies that solely consisted of very old workers (> 252 days old). Interestingly, in the presence of younger workers, old workers rarely showed ovarian development. Besides age, we found a positive correlation between the amount of fat (i.e., nutritional condition) and ovarian development in old workers. Our data suggest that reproductive activity of old workers is low but maintained throughout their life in Diacamma.
Significance statement
Females of social animals, such as cetaceans, are known to stop reproducing before the end of their lifespan. It is suggested that the reproductive ability of ant queens does not decline and is maintained throughout their lives; however, it is unclear whether this is also true in ant workers. We maintained ant colonies for more than 500 days and then tested whether the reproductive ability of ant workers is maintained throughout their lifespan. Even in small groups composed of only very old workers (> 252 days old), at least one individual in each group always reproduced actively. Interestingly, the presence of young workers seemed to suppress the reproduction of old workers. In addition, fat content was positively associated with the individual’s reproductive potential.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Age-related division of labor is common in the social hymenopteran species. Young workers mostly perform intranidal tasks such as brood care, whereas old workers perform extranidal tasks such as foraging and defense (Wilson 1971; Oster and Wilson 1978; Seeley 1982; Gordon 1996). Therefore, old workers tend to engage in riskier tasks (Robinson 1992). The age-related change is also observed in physiological characters. In workers of bees and ants, fat content and/or fat bodies (an indicator of the nutritional status) decrease with age (Blanchard et al. 2000; Toth and Robinson 2005; Bernadou et al. 2015). Meta-analyses in ants have shown that old workers generally have regressed ovaries compared to young workers (Pamminger and Hughes 2017), implying that old workers may retain less reproductive potential. The low nutritional condition and degenerated ovaries characterize the ecological and physiological features of old workers.
In the case of ant queens, it is suggested that the reproductive ability is maintained throughout their lives. The queen of Cardiocondyla obscurior continues to produce broods until just before the end of her life (Heinze and Schrempf 2012; Jaimes-Nino et al. 2022). So far, however, it remains largely unknown whether the very old workers still retain reproductive ability in social hymenopterans. One prediction is that old females have lost their reproductive ability, imitating the prolonged lifespan after menopause in social animals such as cetaceans, humans and some aphids, in which post-menopause old females play roles in nursing, social hunting, and defense (Uematsu et al. 2010; Ellis et al. 2018). Alternatively, old workers might still retain reproductive ability so that they can produce unfertilized eggs in cases when the queen and young workers are lost from the colony. Under the condition in which post-reproductive altruism is more beneficial and/or continuation of the reproductive effort is more costly, the extension of the post-reproductive lifespan can be favored by selection (Bourke 2007; Cant and Johnstone 2008).
Previous studies of ants suggested that old workers have lost their reproductive ability. In the clonal ant Pristomyrmex punctatus, young workers perform intranidal tasks and egg-laying, while old workers become helpers performing extranidal tasks. When only external workers are experimentally isolated, they do not reproduce, even under well-nourished conditions (Tsuji 1988; 1990). In some queenless ponerine ants, young workers tend to be dominant egg-layers whereas old workers are usually subordinate and do not lay eggs (Ito 1993; Higashi et al. 1994; Walsum et al. 1998; Monnin and Peeters 1999). On the other hand, in the clonal queenless ponerine ant Platythyrea punctata, external workers began to lay eggs in the absence of internal workers (Bernadou et al. 2015). Unfortunately, however, these previous studies were only able to approximate the ages of workers based on cuticular color (Bernadou et al. 2015) or task roles (i.e., intranidal or extranidal) (Tsuji 1988; 1990). Cuticular color or chemical concentration can be used as a rough indicator of age, but not an accurate age marker (Hartmann et al. 2019). Additionally, the social compositions of workers’ ages have not been considered. It is still unclear whether aging itself reduces worker reproductive ability or if the presence of younger individuals (i.e., age structure) socially suppresses the reproduction of old workers (Ito 1993; Higashi et al. 1994; Walsum et al. 1998). To fill the above knowledge gaps, it is essential to accurately track worker ages and control the age structures. Thus, we conducted the study using a queenless ponerine ant Diacamma cf. indicum.
Diacamma cf. indicum is a monomorphic ant that has morphologically identical functional queens (i.e., gamergates) and workers. In this species, whether the worker becomes a queen or worker is determined by a dominance hierarchy. All Diacamma females have a pair of wing-like appendages that determine their caste, called ‘‘gemmae’’ (Fukumoto et al. 1989; Peeters and Higashi 1989). In each colony, only one female retaining the gemma becomes a gamergate (i.e., mated worker). The gamergate cuts off the gemmae of newly eclosed females, thus turning them into workers. Gemma-mutilated females lose their ability to mate, but they still maintain the ability to produce unfertilized eggs that develop as males (Allard et al. 2005). Worker reproduction is mostly suppressed in the presence of a gamergate but occurs when a gamergate is absent (Fukumoto et al. 1989; Peeters and Tsuji 1993). The mean lifespan of workers of this species is 208 ± 15 days (Tsuji 1996). As typical in other social hymenopterans, young workers (< a few months old) are intranidal, and old workers are extranidal (Nakata 1995; Fujioka et al. 2019). In addition, worker fat content decreases with age, supporting the low nutritional state of old workers (Fujioka et al. in prep; this study). The histological observation did not detect oocytes in the ovaries of old workers (> 6 months old), implying that old workers have lost reproductive ability (Okada et al. 2010). Considering these studies, we hypothesize that old workers have lower reproductive ability and may be in a post-reproductive state.
In this study, variable ages were separated from the queen-right colony to make experimental orphan colonies wherein workers typically start egg-laying in about 11 days (Fukumoto et al. 1989; Peeters and Tsuji 1993; Okada et al. 2010). To evaluate the effect of age structure on ovarian development, we created three conditions: young workers only (pure-young treatment), old workers only (pure-old treatment), and a mixture of young and old workers (mixed treatment). The mixed treatment is the most natural condition since young and old workers co-exist like in normal colonies. Under these colony compositions and age-controlled conditions, we investigated the age-dependence of worker reproductive ability, fat content, and aggressive dominance behavior.
Materials and methods
Insect
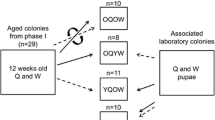
A total of six colonies of Diacamma cf. indicum were excavated in Sueyoshi Park (Naha), Kenmin-no mori (Onna), and Tama-gusuku (Nanjyo) in Okinawa, Japan, from 2018 to 2021. Colonies containing one gamergate (i.e., functional queen) and 73–164 workers were reared in plastic cases (24 × 17.5 × 10 cm) with artificial nests made of moistened plaster and Petri dishes (diameter 9 cm). The nests were kept at 25 ± 3 °C under 12L:12D light/dark conditions (light, 8:00–20:00), fed frozen crickets, and a commercial feed Pro-Jelly (KB Farm, Japan) three times a week. Water was supplied ad libitum. In this study, we painted all newly emerged workers’ thorax and abdomen every 2 weeks using an enamel marker (TAMIYA, Japan) over 1 year so that the exact ages of all workers could be followed by thoracic and abdominal color. The mean lifespan of workers of this species is 208 ± 15 days (Tsuji 1996), and therefore, rearing for 1 year or more is sufficient to follow the lifetime physiological transition.
Individual identification of workers
Individual identification number tags were attached for all workers on the back of the thorax at least 2 days before observation experiments. The tags were made of waterproofed paper of 2 mm2, and they were glued to the dorsal side of the alitrunk by instant glue (3M Japan, UPC: 04519001907820). Based on the notion that oocytes and nurse cells of workers of this species are regressed at 6 months or older (Okada et al. 2010), workers younger than 180 days were defined as young workers. In order to test the very old workers’ reproductive ability, workers of intermediate age (days 181 to 250) were not used in our experiment. Workers older than 251 days were used as old workers. The mean age of young workers was 89.6 days (range, 0–166 days; n = 192), and that of old workers was 353.3 days (range, 259–604 days; n = 208).
From six original colonies, workers were allocated to three types of treatments with different age structures; Mixed groups included two categories, consisting of 8 young workers (Mixed young treatment, MY, n = 10 colonies), 8 old workers (Mixed old treatment, MO, n = 10 colonies). Pure-old treatment consists of 16 old workers (PO treatment, n = 8 colonies), and pure-young treatment consists of 16 young workers (PY treatment, n = 7 colonies). Each experimental sub-colony originated from the nestmates of the same mother colony. A detailed allocation of sub-colonies is summarized in supplementary table S1. Sub-colonies were reared in the same way as described above but smaller artificial nests were used (diameter 6cm). The nests were covered with red film to imitate dark conditions.
Observation of dominance interaction
We observed dominance interactions among workers (i.e., bite and jerk behavior except for antennal boxing, Peeters and Tsuji 1993) in each sub-colony for 15 days, since a previous study showed that Diacamma workers develop ovaries within about 2 weeks after isolation from the gamergate (Peeters and Tsuji 1993). Observations were conducted for 30 min per day. We recorded the attacker and attacked individuals and attacking frequencies based on the tag number. All observations were conducted between 11:00–17:00.
If more than half the individuals died in either of the pure sub-colonies (PO and PY treatments), or more than four young workers or old workers died in the mixed treatments, the observation was stopped and such colonies were omitted from the analysis (one sub-colony, m7) was omitted from the analysis). Individuals dead during the 15-day experimental period were likewise excluded from the analysis (number of dead individuals: MO;10/80, PO;14/128, MY;6/80, PY;4/112; Table S1).
During the observation of dominance interaction, all workers were given random numbers, and sub-colonies were also given random alphabets, so the observations were blinded entirely with regard to group compositions.
Ovary dissection and evaluation of reproductive state
The previous study showed that some workers isolated from the mother colony began oviposition in 7–15 days, and then after 4–6 weeks, only one worker kept developing her ovaries in an isolated sub-colony (Peeters and Tsuji 1993). Thus, in order to quantify reproductive ability, we focused on the physiological states of workers 15 days after isolation. All individuals were frozen (− 20 °C), and the ovarian state was subsequently checked by dissection. Dissection was performed in 1 × PBS, and individual ovaries were photographed using a stereo microscope (S9i, Leica, Germany). Individual ovarian development was ranked into five scores based on previous studies (Heinze et al. 1992; Schilder et al. 1999); Score 1: completely undeveloped ovaries with no oocytes; Score 2: ovaries with at least one or more immature oocytes; Score 3: ovaries with at least one or more mature oocytes; Score 4: ovaries with terminal oocyte larger than the adjacent nurse cell; Score 5: ovaries containing well-developed oocytes and one or more eggs.
Fat content measurement
After gaster dissection, the remaining body parts (i.e., head, alitrunk, and legs) were subjected to fat content analysis based on the methanol-chloroform method (Barnes and Blackstock 1973; Idogawa et al. 2017). Since whole body fat contents can be finely estimated from fat contents in the thorax (Bernadou et al. 2020), we used thorax and head fat content to characterize overall nutritional status. Worker bodies except for the gaster were individually put into glass vials, then dried at 65 °C for 2 days, and the dry mass weighed to the nearest 0.01 mg with an MS105 ultra-microbalance (Mettler-Toledo, Switzerland). Subsequently, fat was extracted using solvent (2:1 mixture of chloroform and methanol, Wako, Japan) by soaking the dried samples for 4 days in 1 ml of solvent at room temperature. After fat removal, the ants were dried at 65 °C for 2 days and weighed again to measure their lean mass. The body fat content was characterized as a body-fat percentage as follows: (dry mass − lean mass) × 100/dry mass.
Statistical analyses
Considering the non-Gaussian data distributions, whether ovarian development, fat content, and dominance behavior frequency differed among worker groups (MO, PO, MY, and MO) were tested using the Steel–Dwass multiple comparison tests using the function NSM3 implemented in R (package: NSM3; Schneider et al. 2022). The effect of treatment was additionally confirmed by generalized linear mixed models (GLMM) that account for the potential effect of sub-colonies as a random factor. Ovarian development scores were ordinal dependent variables, so we applied an ordered logistic error distribution in the clmm function of the “ordinal” package (Christensen 2023). For dominance behavior (i.e., count data), we used a Poisson error distribution in the glmer function of the “lme4” package (Bates et al. 2015). Fat content data did not comply with the assumptions of normality, so we used gamma error distribution in the glmer function of the “lme4” package (Bates et al. 2015). By using a Tukey HSD pairwise multiple comparison method (lsmeans function of the “lsmeans” package; Lenth 2016) the differences among worker groups (MO, PO, MY, and MO) were tested in the GLMM analyses. In all the models, “social_category” (MO, PO, MY, and PY) was given as a fixed effect factor, and “subcolony” was considered as a random factor (Supplemental Information 1). Spearman’s rank correlation test was used to determine whether age, fat content, and dominance behavior frequency correlated with ovarian development for each treatment.
We considered p < 0.05 as the statistically significant threshold. All analyses were performed with R statistical software version 4.2.3 (R Core Team 2023).
Result
Ovarian development
The degree of ovarian development in relation to age is summarized in Fig. 1. The percentage of individuals with well-developed ovaries (scores of 4 or 5) was 5.7% (4/70) for mixed treatment old workers (MO), 70.2% (52/74) for mixed treatment young workers (MY), 23.7% (27/114) for pure old workers (PO), and 60.2% (65/108) for pure young workers (PY). Notably, in the PO treatment (Fig. 1b), at least one, and often several workers had well-developed ovaries (Fig. 1b; Table S1). Ovarian development of old workers was consistently lower than that of young workers, regardless of age structure. While no significant difference in ovarian development was detected between the PY and MY treatments, MO workers had significantly lower ovarian development than PO workers, suggesting that old workers’ ovarian development was suppressed under age-mixed conditions (Fig. 2). This result was also confirmed by GLMM (Supplemental Information 1).
Comparison of ovarian development in different social and age conditions. The 4 bar charts represent results for MO, MY, PO, and PY, respectively. The vertical axis shows the ovarian development score 15 days after the creation of the sub-colonies. Different alphabets on the charts indicate statistically significant differences between groups by multiple comparisons (Steel–Dwass test: MO, n = 70; PO, n = 114; MY, n = 74, PY, n = 108, df = 3; MO vs. PO, p = 0.031; MO vs. MY, MO vs. PY, PO vs. MY, PO vs. PY, p < 0.001; MY vs. PY, p = 0.315)
In our experiment, the oldest worker that had well-developed ovaries was 464 days old, far exceeding the mean worker longevity of 209 days reported for this species (Tsuji 1996). There was a significant negative correlation between age and ovarian development when young and old workers were pooled together in the mixed treatment. However, within each age category, no correlation between age and ovarian development was detected, implying that age variation within the same age categories has no effect on the variation of ovarian development (Fig. S1a).
Dominance interactions and foraging task
We compared the frequency of dominance interactions in different social conditions and age categories (MO, MY, PO, and PY). The 15 days of observation showed that old workers had significantly lower attack frequency than young workers both in pure- and mixed treatments. No difference in attack frequency was observed between the MY and PY workers. In contrast, MO workers showed significantly lower aggression compared to PO workers (Fig. 3a). This result was also supported by GLMM (Supplemental Information 1). Correlation analysis confirmed the significant positive relationship between attack frequency and ovarian development in all four categories (i.e., MY, MO, PY, PO, Fig. S1b). The frequency of being attacked by other workers was consistently higher in young workers than old workers (Fig. 3b; Supplemental Information 1).
A comparison of the frequency of dominance interactions among different social conditions. The four histograms represent the results of MO, MY, PO, and PY, respectively. Different alphabets on the graph indicate statistically significant differences between the groups due to multiple comparisons. a Total number of attacks times by individual ants during 15 days, 450 min of observation (Steel–Dwass test, MO, n = 70; PO, n = 114; MY, n = 74; PY, n = 108, df = 3; MO vs. MY, p < 0.001; MO vs. PO, p = 0.0481; MO vs. MY, MO vs. PY, PO vs. MY, PO vs. PY, p < 0.001; MY vs. PY, p = 0.9649). b Total number of attacks received by individual ants during 15 days, 450 min of observation (Steel–Dwass test: MO, n = 70; MY, n = 74; PO, n = 114; PY, n = 108, df = 3; MO vs. PO, p = 0.2134; MO vs. MY, MO vs. PY, PO vs. MY, PO vs. PY, p < 0.001; MY vs. PY, p = 1.00)
Fat contents
The fat percentages (fat weight/total dry weight) of old workers were significantly lower than those of young workers. No statistical difference was detected in the fat percentages of MY and PY workers. Steel–Dwass test showed that MO workers had less fat than PO workers (Fig. 4), but this difference was not supported by GLMM (Supplemental Information 1), suggesting that the difference between MO and PO was subtle. Correlation analysis showed positive correlations between ovarian development and fat percentages, except for MY workers (Fig. S1c).
Comparison of body fat percentage of workers in different social and age conditions. The horizontal axis represents the fat content of workers, which is calculated as a percentage of dry mass, and the four histograms represent the results of MO, MY, PO, and PY, respectively. Different alphabets on the graph indicate statistically significant differences between the groups due to multiple comparisons (Steel–Dwass test, MO, n = 69; PO, n = 111; MY, n = 72; PY, n = 86, df = 3; MO vs. PO, p < 0.01; MO vs. MY, MO vs. PY, PO vs. MY, PO vs. PY, p < 0.001; MY vs. PY, p = 0.20)
Discussion
In this study, we show that Diacamma workers retain reproductive ability for unexpectedly long periods. Ovarian dissections of 366 workers from 25 sub-colonies showed that even the sub-colonies that only consisted of very old workers produced some egg-layers (Fig. 1). The previously reported worker longevity is 209 days and that of gamergates is 579 days on average (Tsuji 1996). Therefore, workers much older than their average lifespan retained reproductive ability. At maximum, a 464-day-old worker was found to develop ovaries. The fact that sub-colonies solely consisting of very old workers (> 180 days old) can always produce reproductive workers suggests that Diacamma old workers do not lose reproductive ability. Contrary to the initial prediction, our data suggest that Diacamma workers retain reproductive ability for most of their lifetime, and there are no signs of “menopause” in this species.
The maintenance of reproductive potential until late in life may not be specific to Diacamma workers. Studies of Cardiocondyla obscurior have shown that the reproductive ability of ant queens is maintained until just before death (Heinze and Schrempf 2012; Jaimes-Nino et al. 2022). Given the superorganismality of social insects, such a pattern is broadly expected in social insect queens (Jaimes-Nino et al. 2022). Our current study adds an insight that not only queens but also workers can maintain reproductive ability late in life, especially in morphologically casteless species and/or species in which worker reproduction is frequent and functional (e.g., queenless ants).
The reason why Diacamma workers maintain their reproductive potential for such a long period may be related to the reproductive ecology and nesting habitat of this species. Diacamma colonies consist of a small number of individuals, ranging from 30 to 200 (Fukumoto et al. 1989), and colonies frequently proliferate by fission. Although the actual age structure of dividing colonies is unknown, divided colonies may sometimes have biased age structures, such as containing only old workers, and this may lead the old workers to have a chance of reproduction. Additionally, Diacamma cf indicum makes shallow, less elaborate nests in unstable places such as small cavities under/between stones, and frequently relocates nests. Their shoddily-built nests are vulnerable to natural disturbances such as typhoons and floods that are common in their habitats (Bhattacharyya et al. 2021; Kaur and Sumana 2014; Kolay and Annagiri 2015) Consequently, the catastrophic loss of nest center, that is, the loss of the gamergate and intranidal workers may occasionally occur in nature. In such a situation, the returning foragers should take over the colony by themselves and even if workers cannot mate and can only produce males, they may benefit from producing males by themselves. The presence of many potential egg-layers in a colony is considered to reduce the risk of catastrophic colony damage (Tsuji 1988; 1990; Ravary and Jaisson 2004). The high reproductive plasticity of old workers may be an adaptation to a presumably variable colonial age structure and/or compensation for catastrophic loss of colony and/or gamergate.
Within mixed groups, young workers largely monopolized egg production (Fig. 1). This is consistent with previous studies using other ponerine ants (Ito 1993; Higashi et al. 1994; Walsum et al. 1998; Monnin and Peeters 1999). In our study, age structure was controlled, and this social manipulation showed that PO workers had higher ovarian development than MO workers. This means that the lowered ovarian activity in old workers is not only due to physiological deterioration but also due to social factors, (i.e., the presence of young workers). Since dominance behavior is costly (Gobin and Ito 2003; Bocher et al. 2008), and ovarian development takes time (11–14 days, Fukumoto et al. 1989; Okada et al. 2010), a turnover of reproductive females should impose a colonial cost. When an old worker becomes a reproductive, the lifetime expectancy of this new reproductive is short, and thus the costly turnover of the reproductive may occur in a short period. The relatively low reproductive activity of old workers should enhance the efficient reproductive division of labor that consequently reduces the colony-level cost of reproductive turnovers.
The result also showed that the amount of fat content is highly associated with reproductive activity. The body fat percentage of old workers was clearly lower than that of young workers (Fig. 4). This is consistent with previous studies in other social hymenopterans (Bernadou et al. 2015; 2020). While Diacamma workers maintained their reproductive potential throughout their lifetime, there was a steady deterioration of their nutritional status, which is likely strongly age-dependent and inevitable. Correlation analysis showed no strong relationship between ovarian development and fat percentage in young workers. Interestingly, however, a significant correlation was observed in old workers, especially in PO workers. This suggests that in young workers, where all individuals are well nourished, differences in fat content among workers have little influence on reproductive ability. Instead, the consequence of dominance interaction is more fundamental in the determination of reproduction in young workers (Fig. 3). In contrast, old workers’ nutritional status was generally low due to aging (Fig. 4), but among them, the nutritional variability plays a central role in the determination of reproductive potential. This pattern is in concordance with an idea of the colony-level minimization of the cost associated with reproductive turnover.
In this study, we clarified that the reproductive potential of old workers lasts for a very long period, likely for the entire lifetime of workers in a monomorphic ponerine ant. Ants have diverse reproductive and caste systems and among them, queenless ponerine ants have high worker reproductive potential. Worker ovarian activity may be lower in queen-worker dimorphic species with ovary-possessing workers. For example, Khila and Abouheif (2008) showed that workers can lay eggs, but some of these eggs cannot develop due to errors in embryogenesis. Such inability of worker reproduction may represent an adaptive proximate mechanism to maintain social harmony (Khila and Abouheif 2010). In this study, it is not clear whether the eggs produced by old workers of Diacamma are viable or not. The viability of the eggs laid by the very old workers should be validated in future studies. Investigation of age-dependent deterioration of worker reproductive ability may help understand the diversity and mechanism of social evolution in future works.
Data availability
The raw data for this study is available in Table S1.
References
Allard D, Ito F, Gobin B et al (2005) Differentiation of the reproductive tract between dominant and subordinate workers in the Japanese queenless ant Diacamma sp. Acta Zool 86:159–166. https://doi.org/10.1111/j.1463-6395.2005.00197.x
Barnes H, Blackstock J (1973) Estimation of lipids in marine animals and tissues: detailed investigation of the sulphophosphovanilun method for “total” lipids. J Exp Mar Bio Ecol 12:103–118. https://doi.org/10.1016/0022-0981(73)90040-3
Bates D, Mächler M, Bolker B, Walker (2015) Fitting linear mixed-effects models using lme4. Journal of Statistical Software 67(1):1–48. https://doi.org/10.18637/jss.v067.i01
Bernadou A, Busch J, Heinze J (2015) Diversity in identity: behavioral flexibility, dominance, and age polyethism in a clonal ant. Behav Ecol Sociobiol 69:1365–1375. https://doi.org/10.1007/s00265-015-1950-9
Bernadou A, Hoffacker E, Pable J, Heinze J (2020) Lipid content influences division of labour in a clonal ant. J Exp Biol 223. https://doi.org/10.1242/jeb.219238
Bhattacharyya K, Kolay S, Annagiri S (2021) The structure and importance of nest mounds in a tropical ant Diacamma indicum. Ecol Entomol 46:1324–1332. https://doi.org/10.1111/een.13079
Blanchard GB, Orledge GM, Reynolds SE, Franks NR (2000) Division of labour and seasonality in the ant Leptothorax albipennis: worker corpulence and its influence on behaviour. Anim Behav 59:723–738. https://doi.org/10.1006/anbe.1999.1374
Bocher A, Doums C, Millot L, Tirard C (2008) Reproductive conflicts affect labor and immune defense in the queenless ant Diacamma SP. “nilgiri.” Evolution 62:123–134. https://doi.org/10.1111/j.1558-5646.2007.00273.x
Bourke AFG (2007) Kin selection and the evolutionary theory of aging. Annu Rev Ecol Evol Syst 38:103–128. https://doi.org/10.1146/annurev.ecolsys.38.091206.095528
Cant MA, Johnstone RA (2008) Reproductive conflict and the separation of reproductive generations in humans. Proc Natl Acad Sci U S A 105:5332–5336. https://doi.org/10.1073/pnas.0711911105
Christensen R (2023) Ordinal-regression models for ordinal data. R package version 2023.12–4, <https://CRAN.R-project.org/package=ordinal>.
Ellis S, Franks DW, Nattrass S et al (2018) Analyses of ovarian activity reveal repeated evolution of post-reproductive lifespans in toothed whales. Sci Rep 8:12833. https://doi.org/10.1038/s41598-018-31047-8
Fujioka H, Abe MS, Okada Y (2019) Ant activity-rest rhythms vary with age and interaction frequencies of workers. Behav Ecol Sociobiol 73:30. https://doi.org/10.1007/s00265-019-2641-8
Fukumoto Y, Abe T, Taki A (1989) A novel form of colony organization in the “queenless” ant Diacamma rugosum. Physiol Ecol Japan 26:55–61
Gobin B, Ito F (2003) Sumo wrestling in ants: major workers fight over male production in Acanthomyrmex ferox. Naturwissenschaften 90:318–321. https://doi.org/10.1007/s00114-003-0430-7
Gordon DM (1996) The organization of work in social insect colonies. Nature 380:121–124. https://doi.org/10.1038/380121a0
Hartmann C, Heinze J, Bernadou A (2019) Age-dependent changes in cuticular color and pteridine levels in a clonal ant. J Insect Physiol 118:103943. https://doi.org/10.1016/j.jinsphys.2019.103943
Heinze J, Schrempf A (2012) Terminal investment: individual reproduction of ant queens increases with age. PLoS ONE 7:e35201. https://doi.org/10.1371/journal.pone.0035201
Heinze J, Lipski N, Hölldobler B (1992) Reproductive competition in colonies of the ant Leptothorax gredleri. Ethology 90:265–278. https://doi.org/10.1111/j.1439-0310.1992.tb00838.x
Higashi S, Ito F, Sugiura N, Ohkawara K (1994) Worker’s age regulates the linear dominance hierarchy in the queenless ponerine ant, Pachycondyla sublaevis (Hymenoptera: Formicidae). Anim Behav 47:179–184. https://doi.org/10.1006/anbe.1994.1020
Idogawa N, Watanabe M, Yokoi T (2017) Nutrient allocation for somatic maintenance and worker production by the queen of the Japanese black carpenter ant, Camponotus japonicus (Hymenoptera: Formicidae). Appl Entomol Zool 52:527–530. https://doi.org/10.1007/s13355-017-0505-0
Ito F (1993) Functional monogyny and dominance hierarchy in the queenless ponerine ant Pachycondyla (=Bothroponera) sp. in West Java, Indonesia (Hymenoptera, Formicidae, Ponerinae). Ethology 95:126–140. https://doi.org/10.1111/j.1439-0310.1993.tb00463.x
Jaimes-Nino LM, Heinze J, Oettler J (2022) Late-life fitness gains and reproductive death in Cardiocondyla obscurior ants. Elife 11. https://doi.org/10.7554/eLife.74695
Kaur R, Sumana A (2014) Coupled adult-brood transport augments relocation in the Indian queenless ant Diacamma indicum. Insectes Soc 61:141–143. https://doi.org/10.1007/s00040-014-0338-6
Khila A, Abouheif E (2008) Reproductive constraint is a developmental mechanism that maintains social harmony in advanced ant societies. Proc Natl Acad Sci U S A 105:17884–17889. https://doi.org/10.1073/pnas.0807351105
Khila A, Abouheif E (2010) Evaluating the role of reproductive constraints in ant social evolution. Philos Trans R Soc Lond B Biol Sci 365:617–630. https://doi.org/10.1098/rstb.2009.0257
Kolay S, Annagiri S (2015) Dual response to nest flooding during monsoon in an Indian ant. Sci Rep 5:13716. https://doi.org/10.1038/srep13716
Lenth R (2016) Least-squares means: the R package lsmeans. Journal of Statistical Software 69(1):1–33. https://doi.org/10.18637/jss.v069.i01
Monnin T, Peeters C (1999) Dominance hierarchy and reproductive conflicts among subordinates in a monogynous queenless ant. Behav Ecol 10:323–332. https://doi.org/10.1093/beheco/10.3.323
Nakata K (1995) Age polyethism, idiosyncrasy and behavioural flexibility in the queenless ponerine ant, Diacamma sp. J Ethol 13:113–123. https://doi.org/10.1007/BF02352570
Okada Y, Miyazaki S, Miyakawa H et al (2010) Ovarian development and insulin-signaling pathways during reproductive differentiation in the queenless ponerine ant Diacamma sp. J Insect Physiol 56:288–295. https://doi.org/10.1016/j.jinsphys.2009.10.013
Oster GF, Wilson EO (1978) Caste and ecology in the social insects. Monogr Popul Biol 12:1–352. https://doi.org/10.1007/BF00046355
Pamminger T, Hughes WOH (2017) Testing the reproductive groundplan hypothesis in ants (Hymenoptera: Formicidae). Evolution 71:153–159. https://doi.org/10.1111/evo.13105
Peeters C, Higashi S (1989) Reproductive dominance controlled by mutilation in the queenless ant Diacamma australe. Sci Nat 76:177–180
Peeters C, Tsuji K (1993) Reproductive conflict among ant workers in Diacamma sp. from Japan: dominance and oviposition in the absence of the gamergate. Insectes Soc 40:119–136. https://doi.org/10.1007/BF01240701
R Core Team (2023) R: A language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria. http://www.R-project.org/
Ravary F, Jaisson P (2004) Absence of individual sterility in thelytokous colonies of the ant Cerapachys biroi Forel (Formicidae, Cerapachyinae). Insectes Soc 51:67–73. https://doi.org/10.1007/s00040-003-0724-y
Robinson GE (1992) Regulation of division of labor in insect societies. Annu Rev Entomol 37(1):637–665. https://doi.org/10.1146/annurev.en.37.010192.003225
Schilder K, Heinze J, Hölldobler B (1999) Colony structure and reproduction in the thelytokous parthenogenetic ant Platythyrea punctata (F. Smith) (Hymenoptera, Formicidae). Insectes Soc 46:150–151
Schneider G, Chicken E, Becvarik R, Schneider MG (2022) NSM3: functions and datasets to accompany Hollander, Wolfe, and chicken - nonparametric statistical methods, Third Edition. R package version 1.17. https://CRAN.R-project.org/package=NSM3>.
Seeley TD (1982) Adaptive significance of the age polyethism schedule in honeybee colonies. Behav Ecol Sociobiol 11:287–293. https://doi.org/10.1007/BF00299306
Toth AL, Robinson GE (2005) Worker nutrition and division of labour in honeybees. Anim Behav 69:427–435. https://doi.org/10.1016/j.anbehav.2004.03.017
Tsuji K (1988) Obligate parthenogenesis and reproductive division of labor in the Japanese queenless ant Pristomyrmex pungens. Behav Ecol Sociobiol 23:247–255. https://doi.org/10.1007/BF00302947
Tsuji K (1990) Reproductive division of labour related to age in the Japanese queenless ant, Pristomyrmex pungens. Anim Behav 39:843–849. https://doi.org/10.1016/S0003-3472(05)80948-0
Tsuji K (1996) Lifespan and reproduction in a queenless ant. Naturwissenschaften. https://doi.org/10.1007/BF01141985
Uematsu K, Kutsukake M, Fukatsu T et al (2010) Altruistic colony defense by menopausal female insects. Curr Biol 20:1182–1186. https://doi.org/10.1016/j.cub.2010.04.057
Walsum E, Gobin B, Ito F, Billen J (1998) Worker reproduction in the ponerine ant Odontomachus simillimus. Sociobiology 32:427–440
Wilson EO (1971) The insect societies. Belknap Press of Harvard University Press, Cambridge
Acknowledgements
We thank Dr. Adam L. Cronin (TMU), Dr. Jürgen Heinze, Dr. Eva Schultner, and an anonymous reviewer for kindly editing the early version of this manuscript. We also thank Dr. Yuya Tachiki for the statistical advice. The member of the Animal Ecology Laboratory, TMU supported this study.
Funding
Open access funding provided by Tokyo Metropolitan University. This work is supported by JSPS KAKENHI 17K19381, 19H04913, and Grantship for Young Scientists in Tokyo Metropolitan University to YO.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflicts of interest associated with this manuscript.
Additional information
Communicated by J. Heinze
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kishino, K., Sakiyama, K., Fujioka, H. et al. No sign of reproductive cessation in the old workers of a queenless ponerine ant. Behav Ecol Sociobiol 78, 32 (2024). https://doi.org/10.1007/s00265-024-03445-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-024-03445-8