Abstract
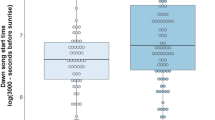
Vocal communication is used across taxa to convey a range of information. One of the most well-studied vocal behaviors is the song of temperate passerine birds. Among individuals, male song differs across numerous acoustic parameters, many of which are used by females to assess male quality. Males in better condition often produce higher song output and start singing earlier in the day compared to other males. However, the energetic cost of singing varies throughout the day and may be affected by the singer’s foraging abilities. Along an elevation gradient, harsher environmental conditions at higher elevations shape foraging abilities in mountain chickadees (Poecile gambeli), which are non-migratory food hoarders. High-elevation mountain chickadees exhibit superior spatial cognitive abilities and a higher propensity to cache food, which enables them to forage more efficiently, compared to lower elevation birds. High-elevation males may therefore be able to produce higher song output, especially earlier in the day, than low-elevation males. We compared the daily singing routines of male mountain chickadees inhabiting high and low elevations. We found no difference in total daily song output between elevations; however, high-elevation males sang significantly more than low-elevation males at dawn, when singing is both crucial for reproductive opportunities and most costly due to overnight fasting. Song output at dawn may therefore act as an indicator of condition in male mountain chickadees.
Significance statement
Animals communicate across modalities to exchange information about foraging, predators, and movements. Researchers have studied the vocalizations of songbirds to understand how individual variation affects the behavior of receives, particularly regarding male song and female mating preferences. The song of many songbirds is learned locally and contains information about the location of origin and respective selective pressures. We tested whether natural access to food via food cache recovery was associated with daily singing routines in food-caching birds. Singing at dawn is thought to be costly as it follows overnight fasting. We found that birds from harsher environments—where selection for enhanced spatial cognitive abilities required for cache retrieval is much stronger—sing more at dawn compared to males from milder environments, where selection on spatial cognition is relaxed. This work shows that environmental selection pressures affect signal production in male songbirds, which may provide information to females during mate choice.


Similar content being viewed by others
Data availability
Raw data for all analyses is available as supplementary material.
References
Ballentine B, Hyman J, Nowicki S (2004) Vocal performance influences female response to male bird song: an experimental test. Behav Ecol 15:163–168. https://doi.org/10.1093/beheco/arg090
Barbour MG, Minnich RA (2000) Californian upland forests and woodlands. In: Barbour MG, Billings WD (eds) North American Terrestrial Vegetation. Cambridge University Press, Cambridge, pp 161–202
Beani L, Dessì-Fulgheri F (1995) Mate choice in the grey partridge, Perdix perdix: role of physical and behavioural male traits. Anim Behav 49:347–356. https://doi.org/10.1006/anbe.1995.0047
Bednekoff PA, Houston AI (1994) Avian daily foraging patterns: effects of digestive constraints and variability. Evol Ecol 8:36–52. https://doi.org/10.1007/BF01237664
Benedict LM, Pitera AM, Branch CL, Sonnenberg BR, Pravosudov VV (2020) Elevation-related differences in annual survival of adult food-caching mountain chickadees are consistent with natural selection on spatial cognition. Behav Ecol Sociobiol 74:40. https://doi.org/10.1007/s00265-020-2817-2
Berg ML, Beintema NH, Welbergen JA, Komdeur J (2005) Singing as a handicap: the effects of food availability and weather on song output in the Australian reed warbler Acrocephalus australis. J Avian Biol 36:102–109. https://doi.org/10.1111/j.0908-8857.2005.03285.x
Branch CL, Kozlovsky DY, Pravosudov VV (2015) Elevation-related differences in female mate preference in mountain chickadees: are smart chickadees choosier? Anim Behav 99:89–94. https://doi.org/10.1016/j.anbehav.2014.10.021
Branch CL, Pitera AM, Kozlovsky DY, Sonnenberg B, Benedict L, Pravosudov VV (2019) Elevation-related differences in the age structure of breeding birds suggest stronger selection at harsher elevations. Behav Ecol Sociobiol 73:143. https://doi.org/10.1007/s00265-019-2750-4
Branch CL, Pitera AM, Kozlovsky DY, Bridge ES, Pravosudov VV (2019) Smart is the new sexy: female mountain chickadees increase reproductive investment when mated to males with better spatial cognition. Ecol Lett 22:897–903. https://doi.org/10.1111/ele.13249
Branch CL, Pravosudov VV (2015) Mountain chickadees from different elevations sing different songs: acoustic adaptation, temporal drift or signal of local adaptation? R Soc Open Sci 2:150019. https://doi.org/10.1098/rsos.150019
Branch CL, Pravosudov VV (2020) Variation in song structure along an elevation gradient in a resident songbird. Behav Ecol Sociobiol 74:9. https://doi.org/10.1007/s00265-019-2786-5
Byers BE (2007) Extrapair paternity in chestnut-sided warblers is correlated with consistent vocal performance. Behav Ecol 18:130–136. https://doi.org/10.1093/beheco/arl058
Collins SA, Hubbard C, Houtman AM (1994) Female mate choice in the zebra finch — the effect of male beak colour and male song. Behav Ecol Sociobiol 35:21–25. https://doi.org/10.1007/BF00167055
Croston R, Branch CL, Pitera AM, Kozlovsky DY, Bridge ES, Parchman TL, Pravosudov VV (2017) Predictably harsh environment is associated with reduced cognitive flexibility in wild food-caching mountain chickadees. Anim Behav 123:139–149. https://doi.org/10.1016/j.anbehav.2016.10.004
Croston R, Kozlovsky DY, Branch CL, Parchman TL, Bridge ES, Pravosudov VV (2016) Individual variation in spatial memory performance in wild mountain chickadees from different elevations. Anim Behav 111:225–234. https://doi.org/10.1016/j.anbehav.2015.10.015
Drăgănoiu TI, Nagle L, Kreutzer M (2002) Directional female preference for an exaggerated male trait in canary (Serinus canaria) song. Proc R Soc Lond B 269:2525–2531. https://doi.org/10.1098/rspb.2002.2192
Endler JA (1986) Natural Selection in the Wild. Princeton University Press, Princeton
Fox J, Weisberg S (2019) An R Companion to Applied Regression, 3rd edn. Sage Publications, Thousand Oaks, CA
Freas CA, LaDage LD, Roth TC, Pravosudov VV (2012) Elevation-related differences in memory and the hippocampus in mountain chickadees, Poecile gambeli. Anim Behav 84:121–127. https://doi.org/10.1016/j.anbehav.2012.04.018
Futuyma DJ (2005) The genetical theory of natural selection. Evolution. Sinauer Associates Inc., Sunderland, MA, pp 269–296
Gil D, Gahr M (2002) The honesty of bird song: multiple constraints for multiple traits. Trends Ecol Evol 17:133–141. https://doi.org/10.1016/S0169-5347(02)02410-2
Gil D, Llusia D (2020) The bird dawn chorus revisited. In: Aubin T, Mathevon N (eds) Coding Strategies in Vertebrate Acoustic Communication. Springer International Publishing, Cham, pp 45–90
Grava T, Grava A, Otter KA (2009) Supplemental feeding and dawn singing in black-capped chickadees. Condor 111:560–564. https://doi.org/10.1525/cond.2009.080061
Grava T, Otter KA, Grava A (2012) Vocal performance varies with habitat quality in black-capped chickadees (Poecile atricapillus). Behaviour 149:35–50. https://doi.org/10.1163/156853912X625854
Hasan NM, Badri M (2016) Effect of ambient temperature on dawn chorus of house sparrows. Environ Ecol Res 4:161–168. https://doi.org/10.13189/eer.2016.040308
Hill GE (1994) Geographic variation in male ornamentation and female mate preference in the house finch: a comparative test of models of sexual selection. Behav Ecol 5:64–73. https://doi.org/10.1093/beheco/5.1.64
Hutchinson JMC, McNamara JM, Cuthill IC (1993) Song, sexual selection, starvation and strategic handicaps. Anim Behav 45:1153–1177. https://doi.org/10.1006/anbe.1993.1139
Kingsolver JG, Hoekstra HE, Hoekstra JM, Berrigan D, Vignieri SN, Hill CE, Hoang A, Gibert P, Beerli P (2001) The strength of phenotypic selection in natural populations. Am Nat 157:245–261. https://doi.org/10.1086/319193
Kozlovsky D, Branch C, Freas CA, Pravosudov VV (2014) Elevation-related differences in novel environment exploration and social dominance in food-caching mountain chickadees. Behav Ecol Sociobiol 68:1871–1881. https://doi.org/10.1007/s00265-014-1796-6
Kozlovsky DY, Branch CL, Pitera AM, Pravosudov VV (2018) Fluctuations in annual climatic extremes are associated with reproductive variation in resident mountain chickadees. R Soc Open Sci 5:171604. https://doi.org/10.1098/rsos.171604
Krebs J, Ashcroft R, Webber M (1978) Song repertoires and territory defence in the great tit. Nature 271:539–542. https://doi.org/10.1038/271539a0
Lenth RV (2016) Least-squares means: the R package lsmeans. J Stat Softw 69:1–33. https://doi.org/10.18637/jss.v069.i01
Lucas JR, Schraeder A, Jackson C (1999) Carolina chickadee (Aves, Paridae, Poecile carolinensis) vocalization rates: effects of body mass and food availability under aviary conditions. Ethology 105:503–520. https://doi.org/10.1046/j.1439-0310.1999.00445.x
McCallum DA, Grundel R, Dahlsten DL (2020) Mountain chickadee (Poecile gambeli). In: Poole AF, Gill FB (eds) Birds of the World. Cornell Lab of Ornithology, Ithaca, NY, USA. https://doi.org/10.2173/bow.mouchi.01
McNamara JM, Barta Z, Houston AI, Race P (2005) A theoretical investigation of the effect of predators on foraging behaviour and energy reserves. Proc R Soc Lond B 272:929–934. https://doi.org/10.1098/rspb.2004.3037
McNamara JM, Houston AI, Lima SL (1994) Foraging routines of small birds in winter: a theoretical investigation. J Avian Biol 25:287–302. https://doi.org/10.2307/3677276
Meigs JB, Smith DC, Van Buskirk J (1983) Age determination of black-capped chickadees. J Field Ornithol 54:283–286
Mennill DJ, Otter KA (2007) Status signaling and communication networks in chickadees: Complex communication with a simple song. In: Otter KA (ed) Ecology and Behavior of Chickadees and Titmice: an Integrated Approach. Oxford University Press, Oxford, pp 215–233
Møller AP (1988) Female choice selects for male sexual tail ornaments in the monogamous swallow. Nature 332:640–642. https://doi.org/10.1038/332640a0
Møller AP (1991) Why mated songbirds sing so much: mate guarding and male announcement of mate fertility status. Am Nat 138:994–1014
Møller AP, de Lope F (1994) Differential costs of a secondary sexual character: an experimental test of the handicap principle. Evolution 48:1676–1683. https://doi.org/10.1111/j.1558-5646.1994.tb02204.x
Mountjoy DJ, Lemon RE (1996) Female choice for complex song in the European starling: a field experiment. Behav Ecol Sociobiol 38:65–71. https://doi.org/10.1007/s002650050218
Ota N, Soma M (2014) Age-dependent song changes in a closed-ended vocal learner: elevation of song performance after song crystallization. J Avian Biol 45:566–573. https://doi.org/10.1111/jav.00383
Otter K, Chruszcz B, Ratcliffe L (1997) Honest advertisement and song output during the dawn chorus of black-capped chickadees. Behav Ecol 8:167–173. https://doi.org/10.1093/beheco/8.2.167
Pitera AM, Branch CL, Bridge ES, Pravosudov VV (2018) Daily foraging routines in food-caching mountain chickadees are associated with variation in environmental harshness. Anim Behav 143:93–104. https://doi.org/10.1016/j.anbehav.2018.07.011
Poesel A, Pedersen SB, Dabelsteen T (2004) Dawn song of male blue tits as a predictor of competitiveness in midmorning singing interactions. Acta Ethol 6:65–71. https://doi.org/10.1007/s10211-004-0086-0
Pravosudov VV, Roth TC II (2013) Cognitive ecology of food hoarding: the evolution of spatial memory and the hippocampus. Annu Rev Ecol Evol S 44:173–193. https://doi.org/10.1146/annurev-ecolsys-110512-135904
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, http://www.R-project.org
Ratcliffe L, Mennill DJ, Schubert KA (2007) Social dominance and fitness in black-capped chickadees. In: Otter KA (ed) Ecology and Behavior of Chickadees and Titmice: an integrated approach. Oxford University Press, Oxford, pp 131–146
Reid ML (1987) Costliness and reliability in the singing vigour of Ipswich sparrows. Anim Behav 35:1735–1743. https://doi.org/10.1016/S0003-3472(87)80066-0
Safran RJ, McGraw KJ (2004) Plumage coloration, not length or symmetry of tail-streamers, is a sexually selected trait in North American barn swallows. Behav Ecol 15:455–461. https://doi.org/10.1093/beheco/arh035
Searcy WA (1984) Song repertoire size and female preferences in song sparrows. Behav Ecol Sociobiol 14:281–286. https://doi.org/10.1007/BF00299499
Sherry DF (1989) Food storing in the Paridae. Wilson Bull 101:289–304
Sonnenberg BR, Branch CL, Pitera AM, Bridge ES, Pravosudov VV (2019) Natural selection and spatial cognition in wild food-caching mountain chickadees. Curr Biol 29:670–676. https://doi.org/10.1016/j.cub.2019.01.006 (e1-e3)
Stehelin TE, Ross Lein M (2014) Social stimulation of dawn singing in Dusky Flycatchers: a serendipitous experiment. J Field Ornithol 85:63–74. https://doi.org/10.1111/jofo.12050
Strain JG, Mumme RL (1988) Effects of food supplementation, song playback, and temperature on vocal territorial behavior of Carolina wrens. Auk 105:11–16
Tello-Ramos MC, Branch CL, Kozlovsky DY, Pitera AM, Pravosudov VV (2019) Spatial memory and cognitive flexibility trade-offs: to be or not to be flexible, that is the question. Anim Behav 147:129–136. https://doi.org/10.1016/j.anbehav.2018.02.019
Tello-Ramos MC, Branch CL, Pitera AM, Kozlovsky DY, Bridge ES, Pravosudov VV (2018) Spatial memory in wild mountain chickadees from different elevations: comparing first-year birds with older survivors. Anim Behav 137:149–160. https://doi.org/10.1016/j.anbehav.2017.12.019
Thomas RJ (1999) Two tests of a stochastic dynamic programming model of daily singing routines in birds. Anim Behav 57:277–284. https://doi.org/10.1006/anbe.1998.0956
Vander Wall SB (1990) Food hoarding in animals. University of Chicago Press, Chicago
Wickham H (2016) ggplot2: Elegant graphics for data analysis, https://github.com/hadley/ggplot2
Yasukawa K (1981) Male quality and female choice of mate in the red-winged blackbird (Agelaius phoeniceus). Ecology 62:922–929. https://doi.org/10.2307/1936990
Zahavi A (1975) Mate selection — a selection for a handicap. J Theor Biol 53:205–214. https://doi.org/10.1016/0022-5193(75)90111-3
Zhang VY, Celis-Murillo A, Ward MP (2016) Conveying information with one song type: changes in dawn song performance correspond to different female breeding stages. Bioacoustics 25:19–28. https://doi.org/10.1080/09524622.2015.1076348
Acknowledgements
VVP and CLB were supported by the National Science Foundation grant (IOS2119824 and IOS1856181) to VVP. CLB was also supported by the NSF Doctoral Dissertation Improvement Grant (IOS1600845) and Edward W. Rose Postdoctoral Fellowship from the Cornell Lab of Ornithology. We would like to thank Dr. Jeff Podos and 2 anonymous reviewers for providing feedback that greatly improved the manuscript. We would also like to thank Jeff Brown and Dan Sayer of Sagehen Creek Field Station (University of California Berkeley) for their invaluable assistance at our field site.
Author information
Authors and Affiliations
Contributions
CLB designed the experiment and collected automated song recordings. SYH, DSL, and VH analyzed song data. SYH conducted statistical analyses and wrote the first draft of the manuscript. CLB and VVP advised on statistical analyses and writing of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
To the best of our knowledge, no birds were harmed by the collection of this data. All applicable national and institutional guidelines for the use of animals were followed. All procedures were approved by the UNR IACUC ethics committee in accordance with the UNR IACUC protocol (00046), under California Department of Fish and Wildlife Permit SC-5210 (DocID: D-0019571790–9).
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by J. Podos.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Huang, S.Y., Schaening-Lopez, D., Halterman, V. et al. Differences in daily singing routines reflect male condition along a montane gradient. Behav Ecol Sociobiol 76, 138 (2022). https://doi.org/10.1007/s00265-022-03246-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-022-03246-x