Abstract
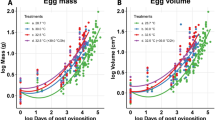
Parental thermoregulation can provide an efficient way to control embryonic temperature and optimize developmental durations and timing. To date, most studies on parental thermal effects have focused on maternal influences, because of the predominant role of females for parental care in most systems. Yet, paternal thermal effects are equally expected to occur in species that display paternal care to the progeny. We studied the midwife toad, a small amphibian with exclusive male parental care and terrestrial eggs transport. We examined the variations of body temperature of egg-carrying males and non-reproductive individuals in the field, and determined their thermal preferences in a laboratory thermal gradient. We also experimentally examined the influence of temperature on embryonic development durations across five treatments (16, 18, 20, 22, 24 °C). In support of our predictions, we detected a shift in thermal preferences with reproduction, with carrying males preferring higher temperatures than non-carrying ones. Field monitoring indicated that carrying males used diurnal shelters early in the season when thermal conditions were more constraining. Meanwhile, they minimized overheating risks by avoiding shelters with high substrate temperature. Finally, we detected a marked thermal dependence of embryonic development, suggesting potential phenological benefits of paternal thermoregulation in this species.
Significance statement
Paternal thermal care is well known among birds during incubation but male thermal care and thermal effects remain virtually unstudied among terrestrial ectotherms. In ectotherms, body temperature directly depends on the thermal environment and behavioral thermoregulation can influence developmental durations and offspring traits. We studied the midwife toad, a species that display exclusive male parental care with terrestrial egg transport. Our results demonstrate that carrying males selected preferred higher temperatures than non-reproductive individuals in a thermal gradient but also avoided thermal extreme in the field. The duration of development was considerably shortened at the temperature selected by reproductive male. Our study provides evidences of active paternal thermoregulation for an amphibian species with exclusive male parental care.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available as supplementary material.
Code availability
Not applicable.
References
Abbey-Lee RN, Dingemanse NJ (2019) Adaptive individual variation in phenological responses to perceived predation levels. Nat Commun 10:1601. https://doi.org/10.1038/s41467-019-09138-5
Alonso-Alvarez C, Velando A (2012) Benefits and costs of parental care. In: Royle NJ, Smiseth PT, Kölliker M (eds) The evolution of parental care. Oxford University Press, Oxford, pp 40–61
Álvarez D, Nicieza AG (2002) Effects of temperature and food quality on anuran larval growth and metamorphosis. Funct Ecol 16:640–648. https://doi.org/10.1046/j.1365-2435.2002.00658.x
Anduaga S, Huerta C (2001) Effect of parental care on the duration of larval development and offspring survival in Nicrophorus mexicanus Matthews (Coleoptera: Silphidae). Coleopt Bull 55:264–270. https://doi.org/10.1649/0010-065x(2001)055[0264:eopcot]2.0.co;2
Angilletta MJ Jr (2009) Thermal adaptation: a theoretical and empirical synthesis. Oxford University Press, Oxford
Angilletta MJ, Sears MW (2003) Is parental care the key to understanding endothermy in birds and mammals? Am Nat 162:821–825
Angilletta MJ Jr, Cooper BS, Schuler MS, Boyles JG (2010) The evolution of thermal physiology in endotherms. Front Biosci 26:861–881
Blouin-Demers G, Weatherhead PJ, McCracken HA (2003) A test of the thermal coadaptation hypothesis with black rat snakes (Elaphe obsoleta) and northern water snakes (Nerodia sipedon). J Therm Biol 28:331–340. https://doi.org/10.1016/S0306-4565(03)00009-3
Bosch J, Márquez R (1996) Discriminant functions for sex identification in two midwife toads (Alytes obstetricans and A. cisternasii). Herpetol J 6:105–110
Chen Y-H, Yu H-T, Kam Y-C (2007) The ecology of male egg attendance in an arboreal breeding frog, Chirixalus eiffingeri (Anura: Rhacophoridae), from Taiwan. Zool Sci 24:434–440. https://doi.org/10.2108/zsj.24.434
Chuang M-F, Lee W-H, Sun J-S, You C-H, Kam Y-C, Poo S (2017) Predation risk and breeding site value determine male behavior and indirectly affect survivorship of their offspring. Behav Ecol Sociobiol 71:122. https://doi.org/10.1007/s00265-017-2349-6
Consolmagno RC, Requena GS, Machado G, Brasileiro CA (2016) Costs and benefits of temporary egg desertion in a rocky shore frog with male-only care. Behav Ecol Sociobiol 70:785–795. https://doi.org/10.1007/s00265-016-2102-6
Cook CL, Ferguson JWH, Telford SR (2001) Adaptive male parental care in the giant bullfrog, Pyxicephalus adspersus. J Herpetol 35:310–315. https://doi.org/10.2307/1566122
da Silva FR, Almeida-Neto M, do Prado VHM, Haddad CFB, de CerqueiraRossa-Feres D (2012) Humidity levels drive reproductive modes and phylogenetic diversity of amphibians in the Brazilian Atlantic Forest. J Biogeogr 39:1720–1732. https://doi.org/10.1111/j.1365-2699.2012.02726.x
Delia JRJ, Ramirez-Bautista A, Summers K (2013) Parents adjust care in response to weather conditions and egg dehydration in a Neotropical glassfrog. Behav Ecol Sociobiol 67:557–569. https://doi.org/10.1007/s00265-013-1475-z
DeNardo DF, Lourdais O, Stahlschmidt ZR (2012) Are females maternal manipulators, selfish mothers or both? Insight from pythons. Herpetologica 68:299–307. https://doi.org/10.1655/herpetologica-d-12-00023.1
Dickerson BR, Brinck KW, Willson MF, Bentzen P, Quinn TP (2005) Relative importance of salmon body size and arrival time at breeding grounds to reproductive success. Ecology 86:347–352. https://doi.org/10.1890/03-625
Du WG, Lu YW, Shen JY (2005) The influence of maternal thermal environments on reproductive traits and hatchling traits in a Lacertid lizard, Takydromus septentrionalis. J Therm Biol 30:153–161. https://doi.org/10.1016/j.jtherbio.2004.09.005
Duarte H, Tejedo M, Katzenberger M, Marangoni F, Baldo D, Beltrán JF, Martí DA, Richter-Boix A, Gonzalez-Voyer A (2012) Can amphibians take the heat? Vulnerability to climate warming in subtropical and temperate larval amphibian communities. Glob Chang Biol 18:412–421. https://doi.org/10.1111/j.1365-2486.2011.02518.x
Dvořák J, Gvoždík L (2009) Oviposition preferences in newts: does temperature matter? Ethology 115:533–539. https://doi.org/10.1111/j.1439-0310.2009.01633.x
Eens M, Pinxten R (2000) Sex-role reversal in vertebrates: behavioural and endocrinological accounts. Behav Process 51:135–147. https://doi.org/10.1016/S0376-6357(00)00124-8
Farmer CG (2000) Parental care: the key to understanding endothermy and other convergent features in birds and mammals. Am Nat 155:326–334
Foucart T, Heulin B, Lourdais O (2018) Small changes, big benefits: testing the significance of maternal thermoregulation in a lizard with extended egg retention. Biol J Linn Soc 125:280–291. https://doi.org/10.1093/biolinnean/bly105
Gillooly JF, Charnov EL, West GB, Savage VM, Brown JH (2002) Effects of size and temperature on developmental time. Nature 417:70–73. https://doi.org/10.1038/417070a
Gururaja KV, Dinesh KP, Priti H, Ravikanth G (2014) Mud-packing frog: a novel breeding behaviour and parental care in a stream dwelling new species of Nyctibatrachus (Amphibia, Anura, Nyctibatrachidae). Zootaxa 3796:33–61. https://doi.org/10.11646/zootaxa.3796.1.2
Gvozdik L (2005) Does reproduction influence temperature preferences in newts? Can J Zool 83:1038–1044
Hanley D (2013) Eggshell conspicuousness is related to paternal brood patch vascularisation in the American thrashers. Avian Biol Res 6:163–177. https://doi.org/10.3184/175815513X665926
Hayward A, Gillooly JF (2011) The cost of sex: Quantifying energetic investment in gamete production by males and females. PLoS ONE 6:e16557. https://doi.org/10.1371/journal.pone.0016557
Heath JE (1964) Reptilian thermoregulation: evaluation of field studies. Science 146:784–785
Huey RB, Berrigan D, Gilchrist GW, Herron JC (1999) Testing the adaptive significance of acclimation: a strong inference approach. Am Zool 39:323–336. https://doi.org/10.1093/icb/39.2.323
Kingsolver J, Huey RB (2008) Size, temperature, and fitness: three rules. Evol Ecol Res 10:251–268
Kurdikova V, Smolinsky R, Gvozdik L (2011) Mothers matter too: benefits of temperature oviposition preferences in newts. PLoS ONE 6:e23842. https://doi.org/10.1371/journal.pone.0023842
Lange L, Brischoux F, Lourdais O (2020) Midwife toads (Alytes obstetricans) select their diurnal refuges based on hydric and thermal properties. Amphibia-Reptilia 41:275–280. https://doi.org/10.1163/15685381-20201246
Lange L, Bégué L, Brischoux F, Lourdais O (2021) The costs of being a good dad: egg-carrying and clutch size impair locomotor performance in male midwife toads (Alytes obstetricans). Biol J Linn Soc 132:270–282. https://doi.org/10.1093/biolinnean/blaa185
Le Henanff M, Meylan S, Lourdais O (2013) The sooner the better: reproductive phenology drives ontogenetic trajectories in a temperate squamate (Podarcis muralis). Biol J Linn Soc 108:384–395. https://doi.org/10.1111/j.1095-8312.2012.02005.x
Lehtinen RM, Green SE, Pringle JL (2014) Impacts of paternal care and seasonal change on offspring survival: a multiseason experimental study of a Caribbean frog. Ethology 120:400–409. https://doi.org/10.1111/eth.12215
Li MA, Zhihua LIN, Jianfang GAO, Hong LI, Xiang JI,Hongliang LU (2018) Maternal thermal effects on female reproduction and hatchling phenotype in the chinese skink (Plestiodon chinensis). Asian Herpetol Res 9:250–257. https://doi.org/10.16373/j.cnki.ahr.180056
Lissåker M, Kvarnemo C (2006) Ventilation or nest defense—parental care trade-offs in a fish with male care. Behav Ecol Sociobiol 60:864–873. https://doi.org/10.1007/s00265-006-0230-0
Lorioux S, DeNardo DF, Gorelick R, Lourdais O (2012) Maternal influences on early development: preferred temperature prior to oviposition hastens embryogenesis and enhances offspring traits in the Children’s python, Antaresia childreni. J Exp Biol 215:1346–1353. https://doi.org/10.1242/jeb.059113
Lorioux S, Vaugoyeau M, Denardo DF, Clobert J, Guillon M, Lourdais O (2013) Stage dependence of phenotypical and phenological maternal effects: insight into squamate reptile reproductive strategies. Am Nat 182:223–233. https://doi.org/10.1086/670809
Márquez R (1992) Terrestrial paternal care and short breeding seasons: reproductive phenology of the midwife toads Alytes obstetricans and A. cisternasii. Ecography 15:279–288. https://doi.org/10.1111/j.1600-0587.1992.tb00036.x
Monteiro NM, Almada VC, Vieira MN (2005) Implications of different brood pouch structures in syngnathid reproduction. J Mar Biol Assoc UK 85:1235–1241. https://doi.org/10.1017/S0025315405012373
Pearman PB (1995) Effects of pond size and consequent predator density on two species of tadpoles. Oecologia 102:1–8. https://doi.org/10.1007/BF00333303
Pellet J, Schmidt BR (2005) Monitoring distributions using call surveys: estimating site occupancy, detection probabilities and inferring absence. Biol Conserv 123:27–35. https://doi.org/10.1016/j.biocon.2004.10.005
Penick CA, Tschinkel WR (2008) Thermoregulatory brood transport in the fire ant, Solenopsis invicta. Insect Soc 55:176–182. https://doi.org/10.1007/s00040-008-0987-4
Poo S, Bickford DP (2013) The adaptive significance of egg attendance in a south-east Asian tree frog. Ethology 119:671–679. https://doi.org/10.1111/eth.12108
Reed TE, Warzybok P, Wilson AJ, Bradley RW, Wanless S, Sydeman WJ (2009) Timing is everything: flexible phenology and shifting selection in a colonial seabird. J Anim Ecol 78:376–387. https://doi.org/10.1111/j.1365-2656.2008.01503.x
Rodríguez-Díaz T, González F, Ji X, Braña F (2010) Effects of incubation temperature on hatchling phenotypes in an oviparous lizard with prolonged egg retention: are the two main hypotheses on the evolution of viviparity compatible? Zoology 113:33–38. https://doi.org/10.1016/j.zool.2009.05.001
Royle NJ, Smiseth PT, Kölliker M (eds) (2012) The evolution of parental care. Oxford University Press, Oxford
Rozen-Rechels D, Dupoué A, Lourdais O, Chamaillé-Jammes S, Meylan S, Clobert J, Le Galliard JF (2019) When water interacts with temperature: ecological and evolutionary implications of thermo-hydroregulation in terrestrial ectotherms. Ecol Evol 9:10029–10043. https://doi.org/10.1002/ece3.5440
Rubalcaba JG, Gouveia SF, Olalla-Tárraga MA (2019) Upscaling microclimatic conditions into body temperature distributions of ectotherms. Am Nat 193:677–687. https://doi.org/10.1086/702717
Ruthsatz K, Peck MA, Dausmann KH, Sabatino NM, Glos J (2018) Patterns of temperature induced developmental plasticity in anuran larvae. J Therm Biol 74:123–132. https://doi.org/10.1016/j.jtherbio.2018.03.005
Schwarzkopf L, Andrews RM (2012) Are moms manipulative or just selfish? Evaluating the “maternal manipulation hypothesis” and implications for life-history studies of reptiles. Herpetologica 68:147–159. https://doi.org/10.1655/herpetologica-d-11-00009.1
Shine R (1995) A new hypothesis for the evolution of viviparity in reptiles. Am Nat 145:809–823. https://doi.org/10.1086/285769
Shine R (2012) Manipulative mothers and selective forces: the effects of reproduction on thermoregulation in reptiles. Herpetologica 68:289–298
Singh SK, Das D, Rhen T (2020) Embryonic temperature programs phenotype in reptiles. Front Physiol 11:35
Smith GD, Hopkins GR, Mohammadi S, Skinner MH, Hansen T, Brodie ED, French SS (2015) Effects of temperature on embryonic and early larval growth and development in the rough-skinned newt (Taricha granulosa). J Therm Biol 51:89–95. https://doi.org/10.1016/J.JTHERBIO.2015.03.010
Stiver KA, Alonzo SH (2009) Parental and mating effort: is there necessarily a trade-off? Ethology 115:1101–1126. https://doi.org/10.1111/j.1439-0310.2009.01707.x
Taigen TL, Pough FH, Stewart MM (1984) Water balance of terrestrial anuran (Eleutherodactylus coqui) eggs: importance of parental care. Ecology 65:248–255. https://doi.org/10.2307/1939477
Telemeco RS, Radder RS, Baird TA, Shine R (2010) Thermal effects on reptile reproduction: adaptation and phenotypic plasticity in a montane lizard. Biol J Linn Soc 100:642–655. https://doi.org/10.1111/j.1095-8312.2010.01439.x
Toufarova E, Gvozdik L (2016) Do female newts modify thermoregulatory behavior to manipulate egg size? J Therm Biol 57:72–77. https://doi.org/10.1016/j.jtherbio.2016.02.001
Townsend DS, Stewart MM, Pough FH (1984) Male parental care and its adaptive significance in a neotropical frog. Anim Behav 32:421–431. https://doi.org/10.1016/S0003-3472(84)80278-X
Treasure AM, Chown SL (2019) Phenotypic plasticity in locomotor performance of a monophyletic group of weevils accords with the ‘warmer is better’ hypothesis. J Exp Biol 222: jeb195255. https://doi.org/10.1242/jeb.195255
Vargas-Salinas F, Quintero-Ángel A, Osorio-Domínguez D, Rojas-Morales JA, Escobar-Lasso S, Gutiérrez-Cárdenas PDA, Rivera-Correa M, Amézquita A (2014) Breeding and parental behaviour in the glass frog Centrolene savagei (Anura: Centrolenidae). J Nat Hist 48:1689–1705. https://doi.org/10.1080/00222933.2013.840942
Wapstra E, Uller T, While GM, Olsson M, Shine R (2010) Giving offspring a head start in life: field and experimental evidence for selection on maternal basking behaviour in lizards. J Evol Biol 23:651–657
Warner DA, Shine R (2007) Fitness of juvenile lizards depends on seasonal timing of hatching, not offspring body size. Oecologia 154:65–73. https://doi.org/10.1007/s00442-007-0809-9
Wells E, Garcia-Alonso D, Rosa GM, Tapley B (2015) Husbandry guidelines for Alytes. European Association of Zoos and Aquaria (EAZA), Amsterdam, https://www.eaza.net/assets/Uploads/CCC/2015-Midwife-toads-EAZA-Best-Practice-Guidelines-Approved.doc.pdf
Zamorano J, Bozinovic F, Veloso C (2017) Colder is better: The differential effects of thermal acclimation on life history parameters in a parasitoid fly. J Therm Biol 68:1–4. https://doi.org/10.1016/j.jtherbio.2017.03.011
Zuo W, Moses ME, West GB, Hou C, Brown JH (2012) A general model for effects of temperature on ectotherm ontogenetic growth and development. Proc R Soc Lond B 279:1840–1846. https://doi.org/10.1098/rspb.2011.2000
Acknowledgements
The authors thank Lauriane Bégué, Juliette Scheuer, and Lisa Poncet for participation in data collection. We warmly thank Pascal Marteau and the sawmill company “Marteau-Bois” for allowing to access to their site. We also thank Jean-Pierre Vacher and the two reviewers for their very constructive comments that have considerably improved the manuscript.
Funding
This study received the financial and technical support from the Centre National de la Recherche Scientifique (CNRS) and was funded by La Rochelle Université (PhD grant to LL) and by the Region Nouvelle-Aquitaine under the “Aquastress” project (2018-1R20214 to OL).
Author information
Authors and Affiliations
Contributions
LL carried out the field work and lab experiments, participated in the statistical analyses, and drafted the manuscript; OL and FB conceived and coordinated the study, participated in data analysis, and critically revised the manuscript. All authors gave final approval for publication and agree to be held accountable for the work performed therein.
Corresponding author
Ethics declarations
Ethics approval
All applicable national, and/or institutional guidelines for the use of animals were followed. This study was performed in accordance with French laws relative to capture, transport, and experiments on Alytes obstetricans (DREAL permit #04022016) and all procedures were approved by an independent ethical committee (Apafis #14193–2018032114365130 v3).
Consent for publication
All authors gave final approval for publication and agree to be held accountable for the work performed therein.
Competing interests
The authors declare no competing interests.
Additional information
Communicated by C. R Gabor.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lange, L., Brischoux, F. & Lourdais, O. Benefits of paternal thermoregulation: male midwife toads select warmer temperature to shorten embryonic development. Behav Ecol Sociobiol 76, 48 (2022). https://doi.org/10.1007/s00265-022-03155-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-022-03155-z