Abstract
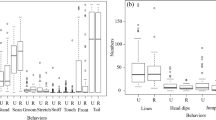
Predator odors are natural stressors that induce anti-predator defensive responses, which have developed under evolutionary pressures to maximize the fitness of an individual to avoid dangerous confrontations. In this study, we analyzed the defensive responses of a subterranean rodent species, the plateau zokor (Eospalax baileyi), to predation threat, and examined the trade-offs between defensive responses and foraging. Plateau zokors were exposed to 1, 3, and 6 g of eagle owl (Bubo bubo) feces, and the anti-predator behavioral, hormonal, and hypothalamic corticotropin-releasing factor and c-fos mRNA expression responses were analyzed and compared with the control (distilled water). Exposure of the animals to owl feces led to significant reductions in both duration of feeding and amount of food consumed, while some defensive behavioral responses (e.g., freezing and rearing) were more significant in the exposed animals than in the control. Serum levels of adrenocorticotropic hormone and corticosterone in plateau zokors exposed to owl feces were significantly higher compared with the control. The hypothalamic corticotropin-releasing factor and c-fos mRNA expression levels of plateau zokors exposed to 1, 3, and 6 g owl feces increased significantly, concomitant with the amount of eagle owl feces presented. Owl feces induced various behavioral, hormonal, and hypothalamic gene expression changes in plateau zokors exposed to them. Some behavioral and gene expression (but not hormonal) responses appeared to be threshold dependent.
Significance statement
Predator odors supersede other signals and can provide comprehensive information to prey during life-threatening events. The plateau zokor is a typical subterranean rodent living in dark and enclosed subterranean habitats. Owing to this special habitat, zokors may be able to recognize ground predator odors and respond defensively. We investigated changes in behavior, hormones, and the hypothalamic corticotropin-releasing factor and c-fos mRNA expression levels of the plateau zokor when exposed to different amounts of eagle owl feces. We believe that our study makes a significant contribution to the literature because our results support the predation risk allocation hypothesis, showing that eagle owl feces inhibit foraging and induce various anti-predatory behavioral and neuroendocrine responses in plateau zokors.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
References
Apfelbach R, Blanchard CD, Blanchard RJ, Hayes RA, McGregor IS (2005) The effects of predator odors in mammalian prey species: a review of field and laboratory studies. Neurosci Biobehav Rev 29:1123–1144. https://doi.org/10.1016/j.neubiorev.2005.05.005
Apfelbach R, Parsons MH, Soini HA, Novotny MV (2015) Are single odorous components of a predator sufficient to elicit defensive behaviors in prey species? Front Neurosci 9:263. https://doi.org/10.3389/fnins.2015.00263
Apps PJ, Weldon PJ, Kramer M (2015) Chemical signals in terrestrial vertebrates: search for design features. Nat Prod Rep 32:1131–1153. https://doi.org/10.1039/c5np00029g
Berger-Tal O, Mukherjee S, Kotler BP, Brown JS (2010) Complex state-dependent games between owls and gerbils. Ecol Lett 13:302–310. https://doi.org/10.1111/j.1461-0248.2010.01447.x
Berton F, Vogel E, Belzung C (1998) Modulation of mice anxiety in response to cat odor as a consequence of predators diet. Physiol Behav 65:247–254. https://doi.org/10.1016/S0031-9384(98)00126-7
Blanchard RJ, Nikulina JN, Sakai RR, McKittrick C, McEwen B, Blanchard DC (1998) Behavioral and endocrine change following chronic predatory stress. Physiol Behav 63:561–569. https://doi.org/10.1016/S0031-9384(97)00508-8
Blanchard DC, Blanchard RJ, Griebel G (2005) Defensive responses to predator threat in the rat and mouse. Curr Protoc Neurosci 30:8.19.1–18.19.20. https://doi.org/10.1002/0471142301.ns0819s30
Bowen MT, Kevin RC, May M, Staples LG, Hunt GE, McGregor IS (2013) Defensive aggregation (huddling) in Rattus norvegicus toward predator odor: individual differences, social buffering effects and neural correlates. PLoS ONE 8:e68483. https://doi.org/10.1371/journal.pone.0068483
Broberger C (2005) Brain regulation of food intake and appetite: molecules and networks. J Intern Med 258:301–327. https://doi.org/10.1111/j.1365-2796.2005.01553.x
Cai WH (2006) Postreactivation glucocorticoids impair recall of established fear memory. J Neurosci 26:9560–9566. https://doi.org/10.1523/jneurosci.2397-06.2006
Campos AC, Fogaca MV, Aguiar DC, Guimaraes FS (2013) Animal models of anxiety disorders and stress. Braz J Psychiatr 35:S101–S111. https://doi.org/10.1590/1516-4446-2013-1139
Canteras NS (2002) The medial hypothalamic defensive system: hodological organization and functional implications. Pharmacol Biochem Behav 71:481–491. https://doi.org/10.1016/S0091-3057(01)00685-2
Chabot D, Gagnon P, Dixon EA (1996) Effect of predator odors on heart rate and metabolic rate of wapiti (Cervus elaphus canadensis). J Chem Ecol 22:839–868. https://doi.org/10.1007/BF02033590
Cook CJ (2002) Glucocorticoid feedback increases the sensitivity of the limbic system to stress. Physiol Behav 75:455–464. https://doi.org/10.1016/S0031-9384(02)00650-9
Creel S, Winnie JA, Christianson D (2009) Glucocorticoid stress hormones and the effect of predation risk on elk reproduction. Proc Natl Acad Sci U S A 106:12388–12393. https://doi.org/10.1073/pnas.0902235106
Cui Q, Lian X, Zhang T, Su J (2003) Food habits comparison between Buteo hemilasius and Bubo bubo. China J Zool 38:57–63 [In Chinese with English summary]
Dautzenberg FM, Hauger RL (2002) The CRF peptide family and their receptors: yet more partners discovered. Trends Pharmacol Sci 23:71–77. https://doi.org/10.1016/S0165-6147(02)01946-6
Dielenberg RA, McGregor IS (2001) Defensive behavior in rats towards predatory odors: a review. Neurosci Biobehav Rev 25:597–609. https://doi.org/10.1016/S0149-7634(01)00044-6
Drakeley M, Lapiedra O, Kolbe JJ (2015) Predation risk perception, food density and conspecific cues shape foraging decisions in a tropical lizard. PLoS ONE 10:e0138016. https://doi.org/10.1371/journal.pone.0138016
Dunn AJ, Berridge CW (1990) Physiological and behavioral responses to corticotropin-releasing factor administration: is CRF a mediator of anxiety or stress responses? Brain Res Brain Res Rev 15:71–100. https://doi.org/10.1016/0165-0173(90)90012-D
Elvidge CK, Ramnarine I, Brown GE (2015) Compensatory foraging in Trinidadian guppies: effects of acute and chronic predation threats. Curr Zool 60:323–332. https://doi.org/10.1093/czoolo/60.3.323
Endres T, Apfelbach R, Fendt M (2005) Behavioral changes induced in rats by exposure to trimethylthiazoline, a component of fox odor. Behav Neurosci 119:1004–1010. https://doi.org/10.1037/0735-7044.119.4.1004
Eriksen A, Wabakken P (2018) Activity patterns at the Arctic Circle: nocturnal eagle owls and interspecific interactions during continuous midsummer daylight. J Avian Biol 49:e01781. https://doi.org/10.1111/jav.01781
Farnworth B, Meitern R, Innes J, Waas JR (2019) Increasing predation risk with light reduces speed, exploration and visit duration of invasive ship rats (Rattus rattus). Sci Rep 9:3739. https://doi.org/10.1038/s41598-019-39711-3
Fendt M, Brosch M, Wernecke KEA, Willadsen M, Wöhr M (2018) Predator odour but not TMT induces 22-kHz ultrasonic vocalizations in rats that lead to defensive behaviours in conspecifics upon replay. Sci Rep 8:11041. https://doi.org/10.1038/s41598-018-28927-4
Ferrari MCO, Sih A, Chivers DP (2009) The paradox of risk allocation: a review and prospectus. Anim Behav 78:579–585. https://doi.org/10.1016/j.anbehav.2009.05.034
Friard O, Gamba M (2016) BORIS: a free, versatile open-source event-logging software for video/audio coding and live observations. Methods Ecol Evol 7:1325–1330. https://doi.org/10.1111/2041-210X.12584
Hegab IM, Wei W (2014) Neuroendocrine changes upon exposure to predator odors. Physiol Behav 131:149–155. https://doi.org/10.1016/j.physbeh.2014.04.041
Hegab IM, Jin Y, Ye M, Wang A, Yin B, Yang S, Wei W (2014) Defensive responses of Brandt's voles (Lasiopodomys brandtii) to stored cat feces. Physiol Behav 123:193–199. https://doi.org/10.1016/j.physbeh.2013.10.030
Hegab IM, Kong S, Yang S, Mohamaden WI, Wei W (2015) The ethological relevance of predator odors to induce changes in prey species. Acta Ethol 18:1–9. https://doi.org/10.1007/s10211-014-0187-3
Hegab IM, Zhang Q, Pu QS, Wang ZC, Kang YK, Cai ZY, Guo H, Wang H, Ji W, Hanafy AM, Su J (2018a) Gender difference in unconditioned and conditioned predator fear responses in Smith's zokors (Eospalax smithii). Glob Ecol Conserv 16:e00503. https://doi.org/10.1016/j.gecco.2018.e00503
Hegab IM, Tan Y, Wang C, Yao B, Wang H, Ji W, Su J (2018b) Examining object recognition and object-in-Place memory in plateau zokors, Eospalax baileyi. Behav Process 146:34–41. https://doi.org/10.1016/j.beproc.2017.11.007
Hogan LA, Lisle AT, Johnston SD, Robertson H (2012) Non-invasive assessment of stress in captive numbats, Myrmecobius fasciatus (Mammalia: Marsupialia), using faecal cortisol measurement. Gen Comp Endocrinol 179:376–383. https://doi.org/10.1016/j.ygcen.2012.09.020
Jolles JW, Boogert NJ, van den Bos R (2015) Sex differences in risk-taking and associative learning in rats. R Soc Open Sci 2:150485. https://doi.org/10.1098/rsos.150485
Kindermann T, Siemers BM, Fendt M (2009) Innate or learned acoustic recognition of avian predators in rodents? J Exp Biol 212:506–513. https://doi.org/10.1242/jeb.024174
Koressaar T, Lepamets M, Kaplinski L, Raime K, Andreson R, Remm M (2018) Primer3_masker: integrating masking of template sequence with primer design software. Bioinformatics 34:1937–1938. https://doi.org/10.1093/bioinformatics/bty036
Lima SL, Bednekoff PA (1999a) Temporal variation in danger drives antipredator behavior: the predation risk allocation hypothesis. Am Nat 153:649–659. https://doi.org/10.1086/303202
Lima SL, Bednekoff PA (1999b) Back to the basics of antipredatory vigilance: can nonvigilant animals detect attack? Anim Behav 58:537–543. https://doi.org/10.1006/anbe.1999.1182
Maroun M, Akirav I (2008) Arousal and stress effects on consolidation and reconsolidation of recognition memory. Neuropsychopharmacology 33:394–405. https://doi.org/10.1038/sj.npp.1301401
Martin PR, Bateson PPG (2017) Measuring behaviour: an introductory guide. Cambridge University Press, Cambridge
McGregor IS, Schrama L, Ambermoon P, Dielenberg RA (2002) Not all 'predator odours' are equal: cat odour but not 2,4,5 trimethylthiazoline (TMT; fox odour) elicits specific defensive behaviours in rats. Behav Brain Res 129:1–16. https://doi.org/10.1016/S0166-4328(01)00324-2
Mitchell MD, Bairos-Novak KR, Ferrari MCO (2017) Mechanisms underlying the control of responses to predator odours in aquatic prey. J Exp Biol 220:1937–1946. https://doi.org/10.1242/jeb.135137
Muñoz-Abellán C, Rabasa C, Daviu N, Nadal R, Armario A (2011) Behavioral and endocrine consequences of simultaneous exposure to two different stressors in rats: interaction or independence? PLoS ONE 6:e21426. https://doi.org/10.1371/journal.pone.0021426
Navarro-Castilla A, Sanchez-Gonzalez B, Barja I (2019) Latrine behaviour and faecal corticosterone metabolites as indicators of habitat-related responses of wild rabbits to predation risk. Ecol Indic 97:175–182. https://doi.org/10.1016/j.ecolind.2018.10.016
Pagani JH, Rosen JB (2009) The medial hypothalamic defensive circuit and 2,5-dihydro-2,4,5-trimethylthiazoline (TMT) induced fear: Comparison of electrolytic and neurotoxic lesions. Brain Res 1286:133–146. https://doi.org/10.1016/j.brainres.2009.06.062
Perrot-Sinal TS, Ossenkopp KP, Kavaliers M (1999) Brief predator odour exposure activates the HPA axis independent of locomotor changes. Neuroreport 10:775–780. https://doi.org/10.1097/00001756-199903170-00021
Roelofs K (2017) Freeze for action: neurobiological mechanisms in animal and human freezing. Philos Trans R Soc B 372:20160206. https://doi.org/10.1098/rstb.2016.0206
Roozendaal B (2002) Stress and memory: Opposing effects of glucocorticoids on memory consolidation and memory retrieval. Neurobiol Learn Mem 78:578–595. https://doi.org/10.1006/nlme.2002.4080
Roseboom PH, Nanda SA, Bakshi VP, Trentani A, Newman SM, Kalin NH (2007) Predator threat induces behavioral inhibition, pituitary-adrenal activation and changes in amygdala CRF-binding protein gene expression. Psychoneuroendocrinology 32:44–55. https://doi.org/10.1016/j.psyneuen.2006.10.002
Sánchez-González B, Barja I, Navarro-Castilla Á (2017) Wood mice modify food intake under different degrees of predation risk: influence of acquired experience and degradation of predator’s faecal volatile compounds. Chemoecology 27:115–122. https://doi.org/10.1007/s00049-017-0237-1
Sánchez-González B, Planillo A, Navarro-Castilla Á, Barja I (2018) The concentration of fear: mice’s behavioural and physiological stress responses to different degrees of predation risk. Sci Nat 105:16. https://doi.org/10.1007/s00114-018-1540-6
Schaller GB (1998) Wildlife of the Tibetan Steppe. University of Chicago Press, Chicago
Schmidt MV (2019) Stress-hyporesponsive period. In: Fink G (ed) Stress: Physiology, Biochemistry, and Pathology. Academic Press, London, pp 49–56
Sinclair ARE, Arcese P (1995) Population consequences of predation-sensitive foraging: the Serengeti wildebeest. Ecology 76:882–891. https://doi.org/10.2307/1939353
Staples LG, Hunt GE, van Nieuwenhuijzen PS, McGregor IS (2008) Rats discriminate individual cats by their odor: Possible involvement of the accessory olfactory system. Neurosci Biobehav Res 32:1209–1217. https://doi.org/10.1016/j.neubiorev.2008.05.011
St-Cyr S, Abuaish S, Spinieli RL, McGowan PO (2018) Maternal predator odor exposure in mice programs adult offspring social behavior and increases stress-induced behaviors in semi-naturalistic and commonly-used laboratory tasks. Front Behav Neurosci 12:136. https://doi.org/10.3389/fnbeh.2018.00136
Stryjek R, Mioduszewska B, Spaltabaka-Gędek E, Juszczak GR (2018) Wild Norway rats do not avoid predator scents when collecting food in a familiar habitat: a field study. Sci Rep 8:9475. https://doi.org/10.1038/s41598-018-27054-4
Su J, Hegab IM, Ji W, Nan Z (2018a) Function-related drivers of skull morphometric variation and sexual size dimorphism in a subterranean rodent, Plateau Zokor (Eospalax baileyi). Ecol Evol 8:4631–4643. https://doi.org/10.1002/ece3.3986
Su JH, Peng R, Nan ZB, Ji WH, Cai ZS (2018b) Age determination and composition analyses of plateau zokor (Eospalax baileyi) in gannan meadow. China J Zool 53:46–54 [In Chinese with English summary]
Takahashi L (2014) Olfactory systems and neural circuits that modulate predator odor fear. Front Behav Neurosci 8:72. https://doi.org/10.3389/fnbeh.2014.00072
Trimmer PC, Ehlman SM, McNamara JM, Sih A (2017) The erroneous signals of detection theory. Proc R Soc B 284:20171852. https://doi.org/10.1098/rspb.2017.1852
Weinberg MS, Bhatt AP, Girotti M, Masini CV, Day HEW, Campeau S, Spencer RL (2009) Repeated ferret odor exposure induces different temporal patterns of same-stressor habituation and novel-stressor sensitization in both hypothalamic-pituitary-adrenal axis activity and forebrain c-fos expression in the rat. Endocrinology 150:749–761. https://doi.org/10.1210/en.2008-0958
Wernecke KEA, Vincenz D, Storsberg S, D'Hanis W, Goldschmidt J, Fendt M (2015) Fox urine exposure induces avoidance behavior in rats and activates the amygdalar olfactory cortex. Behav Brain Res 279:76–81. https://doi.org/10.1016/j.bbr.2014.11.020
Whitham J, Mathis A (2000) Effects of hunger and predation risk on foraging behavior of graybelly salamanders, Eurycea multiplicata. J Chem Ecol 26:1659–1665
Xu JF, Chen XQ, Du JZ, Wang TY (2005) CRF receptor type 1 mediates continual hypoxia-induced CRF peptide and CRF mRNA expression increase in hypothalamic PVN of rats. Peptides 26:639–646. https://doi.org/10.1016/j.peptides.2004.11.014
Yin BF, Gu C, Lu Y, Hegab IM, Yang SM, Wang AQ, Wei WH (2017) Repeated exposure to cat urine induces complex behavioral, hormonal, and c-fos mRNA responses in Norway rats (Rattus norvegicus). Sci Nat 104:64. https://doi.org/10.1007/s00114-017-1484-2
Zhang Y (2007) The biology and ecology of plateau zokors (Eospalax fontanierii). In: Begall S, Burda H, Schleich CE (eds) Subterranean Rodents: news from underground. Springer, Berlin, pp 237–249
Zhou WY, Dou FM (1990) Studies on activity and home range of plateau zokor. Acta Theriol 10:31–39 [In Chinese with English summary]
Acknowledgments
Funding for this study was supported by the National Natural Science Foundation of China (31760706 and 31460566), the Program for Longyuan Youth Innovation Talents of Gansu Province (LQ20180916-2) of China, and Innovation Funds (GSAU-XKJS-2018-003) and “Fuxi Talent” Plan (Gaufx-02J03) of Gansu Agricultural University. Thanks to two anonymous reviewers for their comments on this article
Code availability
Not applicable.
Funding
Funding for this study was supported by the National Natural Science Foundation of China (31760706 and 31460566), the Program for Longyuan Youth Innovation Talents of Gansu Province (LQ20180916-2) of China, and Innovation Funds (GSAU-XKJS-2018-003) and “Fuxi Talent” Plan (Gaufx-02J03) of Gansu Agricultural University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethics approval
The Animal Ethics Committee of Gansu Agricultural University approved the experimental procedure, and the local authorities approved our research (GAU-LC-2020-014). Institutional guidelines for the use of animals were followed.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by C. Soulsbury
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Guo, H., Hegab, I.M., Tan, Y. et al. Exposure to eagle owl feces induces anti-predator behavior, physiology, and hypothalamic gene responses in a subterranean rodent, the plateau zokor (Eospalax baileyi). Behav Ecol Sociobiol 74, 145 (2020). https://doi.org/10.1007/s00265-020-02934-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-020-02934-w