Abstract
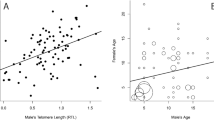
The composition of breeding pairs can deviate from random, resulting in a positive association between homologous traits between mates. Assortative mating can arise either as a by-product of constraints imposed on random pairing by variation in individual life histories or because of mutual sex preferences. Because individuals that prefer high-quality mates gain fitness advantages, mate choice should result in positive assortative mating for traits that are associated with individual quality. Telomeres are nucleoprotein complexes that function to maintain chromosome integrity. Telomeres shorten at each cell division, but the rate of shortening depends on exogenous and endogenous effects, including antioxidant defense mechanisms that reduce the negative impact of pro-oxidants on telomere length. Once a critical length is attained, cells enter either senescence or apoptosis, causing a decline in tissue renewal and ultimately in individual performance. Assortative mating is therefore expected both for telomere length and for antioxidant defense because both males and females may accrue benefits from choosing a mate with long telomeres and efficient antioxidant defense. Consistent with this prediction, in the barn swallow (Hirundo rustica), we found a positive correlation between telomere length (RTL) and total antioxidant capacity (TAC) of social mates which was not the spurious result of age or breeding date. Because parents with relatively long telomeres produce offspring which also have relatively long telomeres, assortative mating seems to be adaptive. The evidence of assortative mating for telomere length and antioxidant capacity suggests that epigamic signals exist that reliably reflect these traits and may mediate mutual mating preferences.
Significance statement
Telomeres protect chromosome integrity but shorten at each cell division, impairing organismal functions. Because individuals vary in telomere length and rate of shortening, as well as in antioxidant defenses reducing the negative impact of pro-oxidants on telomeres, adaptive mate choice should result in assortative mating for telomere length and antioxidants. Consistent with this prediction, in barn swallows, telomere length and antioxidant capacity were positively correlated between social mates.




Similar content being viewed by others
References
Andersson M (1994) Sexual selection. Princeton University Press, Princeton
Badás EP, Martínez J, Rivero de Aguilar Cachafeiro J, Miranda F, Figuerola J, Merino S (2015) Ageing and reproduction: antioxidant supplementation alleviates telomere loss in wild birds. J Evol Biol 28:896–905
Bakaysa SL, Mucci LA, Slagboom PE, Boomsma DI, McClearn GE, Johansson B, Pedersen NL (2007) Telomere length predicts survival independent of genetic influences. Aging Cell 6:769–774
Barrett EL, Burke TA, Hammers M, Komdeur J, Richardson DS (2013) Telomere length and dynamics predict mortality in a wild longitudinal study. Mol Ecol 22:249–259
Bearhop S, Fiedler W, Furness RW, Votier SC, Waldron S, Newton J, Bowen GJ, Berthold P, Farnsworth K (2005) Assortative mating as a mechanism for rapid evolution of a migratory divide. Science 310:502–504
Bize P, Criscuolo F, Metcalfe NB, Nasir L, Monaghan P (2009) Telomere dynamics rather than age predict life expectancy in the wild. Proc R Soc Lond B 276:1679–1683
Campisi J, Kim SH, Lim CS, Rubio M (2001) Cellular senescence, cancer and aging: the telomere connection. Exp Gerontol 36:1619–1637
Caprioli M, Romano M, Romano A, Rubolini D, Motta R, Folini M, Saino N (2013) Nestling telomere length does not predict longevity, but covaries with adult body size in wild barn swallows. Biol Lett 23:20130340
Cawthon RM (2009) Telomere length measurement by a novel monochrome multiplex quantitative PCR method. Nucleic Acids Res 37:e21
Cawthon RM, Smith KR, O'Brien E, Sivatchenko A, Kerber RA (2003) Association between telomere length in blood and mortality in people aged 60 years or older. Lancet 361:393–395
Costanzo A, Parolini M, Bazzi G et al (2016) Brood size, telomere length, and parent-offspring color signaling in barn swallows. Behav Ecol 28:204–211
Cramp S (1998) The complete birds of the Western Palearctic on CD-ROM. Oxford University Press, Oxford
Crespi BJ (1989) Causes of assortative mating in arthropods. Anim Behav 38:980–1000
Crow JF, Felsenstein J (1982) The effect of assortative mating on the genetic composition of a population. Soc Biol 29:22–35
Deng Y, Chan SS, Chang S (2008) Telomere dysfunction and tumour suppression: the senescence connection. Nat Rev Cancer 8:450–458
Dingemanse NJ, Dochtermann NA (2013) Quantifying individual variation in behaviour: mixed-effect modelling approaches. J Anim Ecol 82:39–54
Drès M, Mallet J (2002) Host races in plant–feeding insects and their importance in sympatric speciation. Philos Trans R Soc Lond B 357:471–492
Edelaar P, Siepielski AM, Clobert J (2008) Matching habitat choice causes directed gene flow: a neglected dimension in evolution and ecology. Evolution 62:2462–2472
Epel ES, Blackburn EH, Lin J, Dhabhar FS, Adler NE, Morrow JD (2004) Accelerated telomere shortening in response to life stress. Proc Natl Acad Sci USA 101:17312–17315
Erel O (2004) A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin Biochem 37:277–285
Faravelli M, Azzalin CM, Bertoni L, Chernova O, Attolini C, Mondello C, Giulotto E (2002) Molecular organization of internal telomeric sequences in Chinese hamster chromosomes. Gene 283:11–16
Foote CG, Vleck D, Vleck C (2013) Extent and variability of interstitial telomeric sequences and their effects on estimates of telomere length. Mol Ecol Resour 13:417–428
Giraudeau M, Sweazea K, Butler MW, McGraw KJ (2013) Effects of carotenoid and vitamin E supplementation on oxidative stress and plumage coloration in house finches (Haemorhous mexicanus). Comp Biochem Physiol A Mol Integr Physiol 166:406–413
Hall ME, Nasir L, Daunt F, Gault EA, Croxall JP, Wanless S, Monaghan P (2004) Telomere loss in relation to age and early environment in long-lived birds. Proc R Soc Lond B 271:1571–1576
Harari AR, Handler AM, Landolt PJ (1999) Size-assortative mating, male choice and female choice in the curculionid beetle Diaprepes abbreviates. Anim Behav 58:1191–1200
Haussmann MF, Winkler DW, Vleck CM (2005) Longer telomeres associated with higher survival in birds. Biol Lett 1:212–214
Heidinger BJ, Blount JD, Boner W, Griffiths K, Metcalfe NB, Monaghan P (2012) Telomere length in early life predicts lifespan. Proc Natl Acad Sci USA 109:1743–1748
Houben JMJ, Moonen HJJ, van Schooten FJ, Hageman GJ (2008) Telomere length assessment: bio-marker of chronic oxidative stress? Free Radic Biol Med 44:235–246
Jiang Y, Bolnick DI, Kirkpatrick M (2013) Assortative mating in animals. Am Nat 181:E125–E138
Kim S-Y, Velando A (2015) Antioxidants safeguard telomeres in bold chicks. Biol Lett 11(5):20150211–20150211
Kingsolver JG, Hoekstra HE, Hoekstra JM, Berrigan D, Vignieri SN, Hill CE, Hoang A, Gibert P, Beerli P (2001) The strength of phenotypic selection in natural populations. Am Nat 157:245–261
Kondrashov AS, Shpak M (1998) On the origin of species by means of assortative mating. Proc R Soc Lond B 265:2273–2278
Lewontin R, Kirk D, Crow J (1968) Selective mating, assortative mating, and inbreeding: definitions and implications. Eugen Q 15:141–143
Martínez P, Blasco MA (2015) Replicating through telomeres: a means to an end. Trends Biochem Sci 40:504–515
Meyne J, Ratliff RL, Moyzis RK (1989) Conservation of the human telomere sequence (TTAGGG)n among vertebrates. Proc Natl Acad Sci USA 86:7049–7053
Møller AP (1993) Sexual selection in the barn swallow Hirundo rustica III. Female tail ornaments. Evolution 47:417–431
Møller AP (1994) Sexual selection and the barn swallow. Oxford University Press, Oxford
Møller AP (2004) Protandry, sexual selection and climate change. Glob Change Biol 10:2028–2035
Monaghan P (2010) Telomeres and life histories: the long and the short of it. Ann N Y Acad Sci 1206:130–142
Monaghan P (2014) Organismal stress, telomeres and life histories. J Exp Biol 217:57–66
Noguera JC, Metcalfe NB, Boner W, Monaghan P (2015) Sex-dependent effects of nutrition on telomere dynamics in zebra finches (Taeniopygia guttata). Biol Lett 11:20140938
Palm W, de Lange T (2008) How shelterin protects mammalian telomeres. Annu Rev Genet 42:301–334
Parolini M, Romano A, Khoriauli L, Nergadze SG, Caprioli M, Rubolini D, Santagostino M, Saino N, Giulotto E (2015) Early-life telomere dynamics differ between the sexes and predict growth in the barn swallow (Hirundo rustica). PLoS One 10:e0142530
Roulin A (2004) The evolution, maintenance and adaptive function of genetic colour polymorphism in birds. Biol Rev 79:815–848
Ruiz-Herrera A, Nergadze SG, Santagostino M, Giulotto E (2008) Telomeric repeats far from the ends: mechanisms of origin and role in evolution. Cytogenet Genome Res 122:219–228
Saino N, Caprioli M, Romano M, Boncoraglio G, Rubolini D, Ambrosini R, Bonisoli-Alquati A, Romano A (2011) Antioxidant defenses predict long-term survival in a passerine bird. PLoS One 6:e19593
Saino N, Romano M, Ambrosini R, Rubolini D, Boncoraglio G, Caprioli M, Romano A (2012) Longevity and lifetime reproductive success of barn swallow offspring are predicted by their hatching date and phenotypic quality. J Anim Ecol 81:1004–1012
Salomons HM, Mulder GA, van de Zande L, Haussmann MF, Linkens MHK, Verhulst S (2009) Telomere shortening and survival in free-living corvids. Proc R Soc Lond B 276:3157–3165
Shine R, O'connor D, Lemaster MP, Masonb RT (2001) Pick on someone your own size: ontogenetic shifts in mate choice by male garter snakes result in size-assortative mating. Anim Behav 61:1133–1141
Simons MJ (2015) Questioning causal involvement of telomeres in ageing. Ageing Res Rev 24:191–196
Snowberg LK, Bolnick DI (2008) Assortative mating by diet in a phenotypically unimodal but ecologically variable population of stickleback. Am Nat 172:733–739
Stier A, Reichert S, Criscuolo F, Bize P (2015) Red blood cells open promising avenues for longitudinal studies of ageing in laboratory, non-model and wild animals. Exp Gerontol 71:118–134
Turner A (2006) The barn swallow. T & AD Poyser, London
von Zglinicki T (2002) Oxidative stress shortens telomeres. Trends Biochem Sci 27:339–344
Waser NM (1993) Population structure, optimal outbreeding, and assortative mating in angiosperms. In: Thornhill NW (ed) The natural history of inbreeding and outbreeding: theoretical and empirical perspectives. University of Chicago Press, Chicago, pp 173–199
Webb CJ, Wu Y, Zakian VA (2013) DNA repair at telomeres: keeping the ends intact. Cold Spring Harb Perspect Biol 5:a012666
Weis A, Winterer J, Vacher C, Kossler T, Young C, LeBuhn G (2005) Phenological assortative mating in flowering plants: the nature and consequences of its frequency dependence. Evol Ecol Res 7:161–181
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical approval
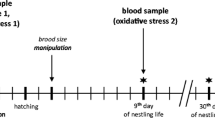
Capture, handling, and blood sampling of barn swallows were authorized by Regione Lombardia (Decreto n° 2141, issued on March 9, 2011). As no manipulative experiments were carried out, no approval from an ethical committee was required for this study. However, all applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Conflict of interest
The authors declare that they have no conflict of interest.
Data availability
The datasets analyzed during the current study will be available from the corresponding author on reasonable request. In addition, all data analyzed during this study will be included in the supplementary information files.
Additional information
Communicated by C. M. Garcia
Electronic supplementary material
ESM 1
(XLS 37 kb)
Rights and permissions
About this article
Cite this article
Khoriauli, L., Romano, A., Caprioli, M. et al. Assortative mating for telomere length and antioxidant capacity in barn swallows (Hirundo rustica). Behav Ecol Sociobiol 71, 124 (2017). https://doi.org/10.1007/s00265-017-2352-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-017-2352-y