Abstract
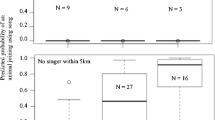
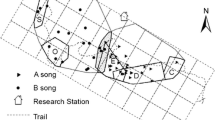
Maynard-Smith’s game-theoretical framework suggests that conflicts should be resolved through an exchange of agonistic displays which can be as effective as physical attack in securing contested resources. Hence, participants use signaling tactics that are governed by game-theoretical competition rules to maximize their benefit from the interaction. In rock hyraxes, males initiate singing in response to conspecific male songs, possibly to self-advertise and to facilitate comparison of performance by receivers. We hypothesized that hyrax counter-singing strategy can be explained by resident-intruder model and its outcome is determined by the identity and ability of the participants. Natural hyrax singing events and playback-induced events were classified according to the participants’ residence status—resident or bachelor (i.e., non-resident). As predicted by the resident-intruder model, resident listeners tended to reply to conspecific males at high rates (≈40 %), while bachelors showed low reply rates (17 %) to resident initiators. Unexpectedly, the lowest-ranking bachelors showed a tendency to reply to the highest-ranking residents. One possible explanation for this counter-intuitive behavior might be that weaker individuals exploit a situation in which it is unremunerated for a stronger opponent to retaliate (i.e., Napoleon strategy) and seize a self-advertisement opportunity in front of a large audience attending to the high-ranking males’ signal. Overall, we show that male hyraxes employ different counter-singing tactics depending on the residence and rank differences between the participants. This choice of competition tactics can be based on opponents’ individual recognition or/and on direct decoding of opponents’ traits encoded in the song.
Significance statement
In nature, conflicts are usually resolved through signaling rather than by actual fighting. Rock hyrax males often participate in vocal competitions, possibly to reduce the need for physical confrontations. During our study on the dynamics of male hyrax vocal competition, we discovered that hyraxes employ mixed counter-singing strategy dependent on the residence status and social rank differences between the participants. A paradoxical “Napoleon-complex” tactic was observed in interactions between males, when low ranking bachelors were found to interact and challenge the highest-ranking residents. On the other hand, residents were found to interact with opponents regardless of residency and/or rank. On top of the cognitive abilities required to individually recognize an opponent and possibly decode its vocal signals, our results suggest that male hyraxes compare own individual traits to those of the opponent in a mutual signal assessment interaction.


Similar content being viewed by others
References
Adkins-Regan E (2005) Hormones and animal social behavior. Princeton University Press, Princeton
Aragon P, Lopez P, Martin J (2006) Roles of male residence and relative size in the social behavior of Iberian rock lizards, Lacerta monticola. Behav Ecol Sociobiol 59:762–769
Bar Ziv E, Ilany A, Demartsev V, Barocas A, Geffen E, Koren L (2016) Individual, social, and sexual niche traits affect copulation success in a polygynandrous mating system. Behav Ecol Sociobiol 70:901–912
Barlow G, Rogers W, Fraley N (1986) Do Midas cichlids win through prowess or daring? It depends. Behav Ecol Sociobiol 19:1–8
Barocas A, Ilany A, Koren L, Kam M, Geffen E (2011) Variance in centrality within rock hyrax social networks predicts adult longevity. PLoS One 6:e22375
Behr O, Knörnschild M, von Helversen O (2009) Territorial counter-singing in male sac-winged bats (Saccopteryx bilineata): low-frequency songs trigger a stronger response. Behav Ecol Sociobiol 63:433–442
Bergman M, Olofsson M, Wiklund C (2010) Contest outcome in a territorial butterfly: the role of motivation. Proc R Soc Lond B 277:3027–3033
Blas J, Cabezas S, Figuerola J, Lopez L, Tanferna A, Hiraldo F (2013) Carotenoids and skin coloration in a social raptor. J Raptor Res 47:174–184
Bolyard KJ, Rowland WJ (2000) The effects of spatial context and social experience on the territorial aggression of male threespine stickleback. Behaviour 137:845–864
Demartsev V, Kershenbaum A, Ilany A, Barocas A, Bar Ziv E, Koren L, Geffen E (2014) Male hyraxes increase song complexity and duration in the presence of alert individuals. Behav Ecol 25:1451–1458
Dirienzo N, Hedrick AV (2014) Animal personalities and their implications for complex signaling. Curr Zool 60:381–386
Elias DO, Botero CA, Andrade MCB, Mason AC, Kasumovic MM (2010) High resource valuation fuels “desperado” fighting tactics in female jumping spiders. Behav Ecol 21:868–875
Fischer J, Kitchen D, Seyfarth R, Cheney D (2004) Baboon loud calls advertise male quality: acoustic features and their relation to rank, age, and exhaustion. Behav Ecol Sociobiol 56:140–148
Fourie PB (1977) Acoustic communication in the Rock Hyrax (Procavia capensis. Z Tierpsychol 44:194–219
Frommolt K, Goltsman M, MacDonald D (2003) Barking foxes, Alopex lagopus: field experiments in individual recognition in a territorial mammal. Anim Behav 65:509–518
Gammell MP, De Vries H, Jennings DJ, Carlin CM, Hayden TJ (2003) David's score: a more appropriate dominance ranking method than Clutton-Brock et al.'s index. Anim Behav 66:601–605
Getty T (1987) Dear enemies and the prisoners-dilemma—why should territorial neighbors form defensive coalitions. Am Zool 27:327–336
Getty T (1989) Are dear enemies in a war of attrition? Anim Behav 37:337–339
Grafen A (1987) The logic of divisively asymmetric contests—respect for ownership and the desperado effect. Anim Behav 35:462–467
Hurd PL (2006) Resource holding potential, subjective resource value, and game theoretical models of aggressiveness signalling. J Theor Biol 241:639–648
Ilany A, Barocas A, Koren L, Kam M, Geffen E (2011) Do singing rock hyraxes exploit conspecific calls to gain attention? PLoS One 6:e28612
Ilany A, Barocas A, Kam M, Ilany T, Geffen E (2013) The energy cost of singing in wild rock hyrax males: evidence for an index signal. Anim Behav 85:995–1001
Innocent TM, West SA, Sanderson JL, Hyrkkanen N, Reece SE (2011) Lethal combat over limited resources: testing the importance of competitors and kin. Behav Ecol 22:923–931
Jennings DJ, Elwood RW, Carlin CM, Hayden TJ, Gammell MP (2012) Vocal rate as an assessment process during fallow deer contests. Behav Process 91:152–158
Jenssen TA, Decourcy KR, Congdon JD (2005) Assessment in contests of male lizards (Anolis carolinensis): how should smaller males respond when size matters? Anim Behav 69:1325–1336
Just W, Morris MR (2003) The Napoleon Complex: why smaller males pick fights. Evol Ecol 17:509–522
Kitchen DM, Seyfarth RM, Fischer J, Cheney DL (2003) Loud calls as indicators of dominance in male baboons (Papio cynocephalus ursinus. Behav Ecol Sociobiol 53:374–384
Kokko H, Rintamaki PT, Alatalo RV, Hoglund J, Karvonen E, Lundberg A (1999) Female choice selects for lifetime lekking performance in black grouse males. Proc R Soc Lond B 266:2109–2115
Kokko H, Lopez-Sepulcre A, Morrell LJ (2006) From hawks and doves to self-consistent games of territorial behavior. Am Nat 167:901–912
Koren L, Geffen E (2009) Complex call in male rock hyrax (Procavia capensis): a multi-information distributing channel. Behav Ecol Sociobiol 63:581–590
Koren L, Geffen E (2011) Individual identity is communicated through multiple pathways in male rock hyrax (Procavia capensis) songs. Behav Ecol Sociobiol 65:675–684
Koren L, Mokady O, Geffen E (2008) Social status and cortisol levels in singing rock hyraxes. Horm Behav 54:212–216
Lindstrom K (2001) Effects of resource distribution on sexual selection and the cost of reproduction in sandgobies. Am Nat 158:64–74
Lotem A (1998) Higher levels of begging behavior by small nestlings: a case of a negatively correlated handicap. Isr J Zool 44:29–45
Mateos C, Carranza J (1997) The role of bright plumage in male-male interactions in the ring-necked pheasant. Anim Behav 54:1205–1214
Maynard-Smith J, Parker GA (1976) Logic of asymmetric contests. Anim Behav 24:159–175
Michod RE, Hasson O (1990) On the evolution of reliable indicators of fitness. Am Nat 135:788–808
Miyai CA, Carretero Sanches FH, Costa TM, Colpo KD, Volpato GL, Barreto RE (2011) The correlation between subordinate fish eye colour and received attacks: a negative social feedback mechanism for the reduction of aggression during the formation of dominance hierarchies. Zoology 114:335–339
Morrell LJ, Lindstrom J, Ruxton GD (2005) Why are small males aggressive? Proc R Soc Lond B 272:1235–1241
Olsson M, Shine R (2000) Ownership influences the outcome of male-male contests in the scincid lizard, Niveoscincus microlepidotus. Behav Ecol 11:587–590
Reby D, McComb K, Cargnelutti B, Darwin C, Fitch WT, Clutton-Brock T (2005) Red deer stags use formants as assessment cues during intrasexual agonistic interactions. Proc R Soc Lond B 272:941–947
Reddon AR, Balk D, Balshine S (2013) Probing aggressive motivation during territorial contests in a group-living cichlid fish. Behav Process 92:47–51
Russ BE, Comins JA, Smith R, Hauser MD (2010) Recognizing and respecting claims over resources in free-ranging rhesus monkeys, Macaca mulatta. Anim Behav 80:563–569
Schradin C, Schneider C, Lindholm AK (2010) The nasty neighbour in the striped mouse (Rhabdomys pumilio) steals paternity and elicits aggression. Front Zool 7:19
Searcy WA, Beecher MD (2009) Song as an aggressive signal in songbirds. Anim Behav 78:1281–1292
Shuster SM, Wade MJ (2003) Mating systems and strategies. Princeton University Press, Princeton
Sih A, Hanser SF, McHugh KA (2009) Social network theory: new insights and issues for behavioral ecologists. Behav Ecol Sociobiol 63:975–988
Smith IP, Huntingford FA, Atkinson RJA, Taylor AC (1994) Strategic decisions during agonistic behaviour in the velvet swimming crab (Necora puber L. Anim Behav 47:885–894
Svensson PA, Lehtonen TK, Wong BBM (2012) A high aggression strategy for smaller males. PLoS One 7:e43121
Temeles EJ (1994) The role of neighbors in territorial systems—when are they dear enemies. Anim Behav 47:339–350
Umbers KDL, Osborne L, Keogh JS (2012) The effects of residency and body size on contest initiation and outcome in the territorial dragon, Ctenophorus decresii. PLoS One 7:e47143
Yack JE, Gill S, Drummond-Main C, Sherratt TN (2014) Residency duration and shelter quality influence vibratory signalling displays in a territorial caterpillar. Ethology 120:354–364
Ydenberg RC, Giraldeau LA, Falls JB (1988) Neighbours, strangers, and the asymmetric war of attrition. Anim Behav 36:343–347
Acknowledgments
We wish to thank two anonymous reviewers for their constructive remarks on the earlier versions of this paper. We are grateful to the Ein Gedi Nature Reserve and Ein Gedi Field School staff for their hospitality and logistic support. We appreciate the valuable help in the field received from all field technicians, project students, and guests. We are grateful to the Nature and Park Authority for permission to work in the Ein Gedi Nature Reserve. We thank M. Samuni-Blank for a fruitful discussion on early drafts of the manuscript and N. Paz for her editorial services.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was supported by three grants from the Israel Science Foundation (577/99, 488/05, 461/09).
Conflict of interest
The authors declare that they have no conflict of interests.
Ethical approval
This study was conducted under annual permits from the Israeli Nature and Parks Authority (NPA) for capturing, handling, and sampling the hyraxes at the Ein Gedi Nature Reserve (2000/8871, 2001/8871, 2002/14674, 2003/14674, 2004/17687, 2005/17687, 2007/27210, 2008/31138, 2009/32871, 2010/37520, 2011/38061, 2012/38400, 2013/38803, 2014/40185). All procedures performed in this study involving animals were in accordance with the ethical standards of the NPA. During the whole course of this 15-year field study, no observable long-term stress or interference effects were detected to individual animals or the whole study population. Both general population numbers and the integrity of the specific social groups in the research area remained stable.
Additional information
Communicated by C. Voigt
Rights and permissions
About this article
Cite this article
Demartsev, V., Ilany, A., Barocas, A. et al. A mixed strategy of counter-singing behavior in male rock hyrax vocal competitions. Behav Ecol Sociobiol 70, 2185–2193 (2016). https://doi.org/10.1007/s00265-016-2222-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-016-2222-z