Abstract
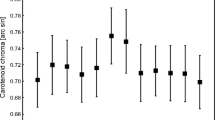
It was long thought that the colour of bird feathers does not change after plumage moult. However, there is increasing evidence that the colour of feathers may change due to abrasion, photochemical change and staining, either accidental or deliberate. The coloration of plumage due to deliberate staining, i.e. with cosmetic purposes, may help individuals to communicate their quality to conspecifics. The presence of carotenoids in preen oils has been previously only suggested, and here we confirm for the first time its presence in such oils. Moreover, the carotenoids in the uropygial secretions were the same specific pigments found in feathers. We show not only that the colour of feathers of greater flamingos Phoenicopterus roseus became more colourful due to the application of carotenoids from uropygial secretions over the plumage but also that the feathers became more colourful with the quantity of pigments applied over them, thus providing evidence of cosmetic coloration. Flamingos used uropygial secretions as cosmetic much more frequently during periods when they were displaying in groups than during the rest of the year, suggesting that the primary function of cosmetic coloration is mate choice. Individuals with more colourful plumage initiated nesting earlier. There was a correlation between plumage coloration before and after removal of uropygial secretions from feathers’ surfaces, suggesting that the use of these pigmented secretions may function as a signal amplifier by increasing the perceptibility of plumage colour, and hence of individual quality. As the cosmetic coloration strengthens signal intensity by reinforcing base-plumage colour, its use may help to the understanding of selection for signal efficacy by making interindividual differences more apparent.





Similar content being viewed by others
References
Altmann J (1974) Observational study of behaviour: sampling methods. Behaviour 49:227–267
Amat JA, Rendón MA, Rendón-Martos M, Garrido A, Ramírez JM (2005) Ranging behaviour of greater flamingos during the breeding and post-breeding periods: linking connectivity to biological processes. Biol Conserv 125:183–192
Bennett ATD, Cuthill IC (1994) Ultraviolet vision in birds: what is its function? Vis Res 34:1471–1478
Blanco G, Frías O, Garrido-Fernández J, Hornero-Méndez D (2005) Environmental-induced acquisition of nuptial plumage expression: a role of denaturation of feathers caroproteins? Proc R Soc B 272:1893–1900
Britton G (1995) UV/visible spectroscopy. In: Britton G, Liaan-Jensen S, Pfander H (eds) Carotenoids: spectroscopy, vol 1B. Birkäuser, Basel, pp 13–63
Cézilly F, Johnson AR (1995) Re-mating between and within breeding seasons in the greater flamingo Phoenicopterus ruber roseus. Ibis 137:543–546
Cramp S, Simmons KEL (1977) The birds of the Western Palearctic, vol 1. Oxford University Press, Oxford
Davies BH, Köst HP (1988) Carotenoids. In: Köst HP (ed) Handbook of chromatography, vol 1. CRC, Boca Raton, pp 3–188
Delhey K, Peters A, Kempenaers B (2007) Cosmetic coloration in birds: occurrence, function, and evolution. Am Nat 169:S145–S158
Delhey K, Peters A, Biedermann P, Kempenaers K (2008) Optical properties of the uropygial gland secretion: no evidence for UV cosmetics in birds. Naturwissenchaften 95:939–946
Eugster CH (1995) Chemical derivatization: microscale tests for the presence of common functional groups in carotenoids. In: Britton G, Liaan-Jensen S, Pfander H (eds) Carotenoids: isolation and analysis, vol. 1A. Birkäuser, Basel, pp 71–80
Figuerola J, Senar JC (2005) Seasonal changes in carotenoid- and melanin-based plumage coloration in the great tit Parus major. Ibis 147:797–802
Foppen FH (1971) Tables for identification of carotenoid pigments. Chromatogr Rev 14:133–298
Fox DL (1975) Carotenoids in pigmentation. In: Kear J, Duplaix-Hall N (eds) Flamingos. Poyser, Berkhamsted, pp 162–182
Grafen A (1990) Sexual selection unhandicapped by the Fisher process. J Theor Biol 144:473–516
Griggio M, Hoi H, Pilastro A (2010) Plumage maintenance affects ultraviolet colour and female preference in the budgerigar. Behav Proc 84:739–744
Guilford T, Dawkins MS (1991) Receiver psychology and the evolution of animal signals. Anim Behav 42:1–14
Hasson O (1991) Sexual displays as amplifiers: practical examples with an emphasis on feather decorations. Behav Ecol 2:189–197
Hays H, Hudon J, Cormons G, Diconstanzo J, Lima P (2006) The pink feather blush of the roseate tern. Waterbirds 29:296–301
Hill GE (2002) A red bird in a brown bag: the function and evolution of ornamental plumage coloration in the house finch. Oxford University Press, Oxford
Hill GE, Nolan PM, Stoehr AM (1999) Pairing success relative to male plumage redness and pigment symmetry in the house finch: temporal and geographic constancy. Behav Ecol 10:48–53
Johnson A, Cézilly F (2007) The greater flamingo. Poyser, London
Kahl MP (1972) Comparative ethology of the Ciconiidae. The wood-storks (genera Mycteria and Ibis). Ibis 114:15–29
López-Rull I, Pagán I, Macías García C (2010) Cosmetic enhancement of signal coloration: experimental evidence in the house finch. Behav Ecol 21:781–787
Maynard Smith J, Harper D (2003) Animal signals. Oxford University Press, Oxford
McGraw KJ, Hill GE (2004) Plumage color as a dynamic trait: carotenoid pigmentation of male house finches (Carpocadacus mexicanus) fades during the breeding season. Can J Zool 82:734–738
Mínguez-Mosquera MI, Hornero-Méndez D (1993) Separation and quantification of the carotenoid pigments in red peppers (Capsicum annuum L), paprika and oleoresin by reversed-phase HPLC. J Agric Food Chem 41:1616–1620
Møller AP, Biard C, Blount JD, Houston DC, Ninni P, Saino N, Surai PF (2000) Carotenoid-dependent signals: indicators of foraging efficiency, immunocompetence or detoxification ability? Avian Poult Biol Rev 11:137–159
Montgomerie R (2006) Cosmetic and adventitious colors. In: Hill GE, McGraw KJ (eds) Bird coloration, vol. I. Harvard University Press, Cambridge, pp 399–427
Negro JJ, Margalida A, Hiraldo F, Heredia R (1999) The function of the cosmetic coloration of bearded vultures: when art imitates life. Anim Behav 58:F14–F17
Perdeck AC, Cavé AJ (1992) Laying date in the coot: effects of age and mate choice. J Anim Ecol 61:13–19
Piault R, Gasparini J, Bize P, Paulet M, McGraw KJ, Roulin A (2008) Experimental support for the make-up hypothesis in nestling tawny owls (Strix aluco). Behav Ecol 19:703–709
Piersma T, Dekker M, Damsté JSS (1999) An avian equivalent of make-up? Ecol Lett 2:201–203
Rendón MA, Garrido A, Ramírez JM, Rendón-Martos M, Amat JA (2001) Despotic establishment of breeding colonies of greater flamingos, Phoenicopterus ruber, in southern Spain. Behav Ecol Sociobiol 50:55–60
Reneerkens J, Korsten P (2004) Plumage reflectance is not affected by preen wax composition in red knots Calidris canutus. J Avian Biol 35:405–409
Reneerkens J, Piersma T, Damsté JSS (2002) Sandpipers (Scolopacidae) switch from monoester to diester preen waxes during courtship and incubation, but why? Proc R Soc Lond B 269:2135–2139
Searcy WA, Nowicki S (2005) The evolution of animal communication: reliability and deception in signaling systems. Princeton University Press, Princeton
Seddon N, Tobias JA, Eaton M, Ödeen A (2010) Human vision can provide a valid proxy for avian perception of sexual dichromatism. Auk 127:283–292
Shannon PW (2000) Plumages and molt patterns in captive Caribbean flamingos. Waterbirds 23(Spec Publ 1):160–172
StatSoft Inc (2001) STATISTICA (data analysis software system), version 6. StatSoft, Tulsa
Stegmann B (1956) Über die Herkunft des flüchtigen rosenroten Federpigments. J Ornithol 97:204–205
Surmacki A (2008) Preen waxes do not protect carotenoid plumage from bleaching by sunlight. Ibis 150:335–341
Surmacki A, Nowakowski JK (2007) Soil and preen waxes influence the expression of carotenoid-based plumage coloration. Naturwissenschaften 94:829–835
Uchida Y (1970) On the color change in Japanese crested ibis. A new type of cosmetic coloration in birds. Misc Rep Yamashina Inst Ornithol 6:54–72, In Japanese, with English summary
Vevers HG (1985) Colour. In: Campbell BC, Lack E (eds) A dictionary of birds. Poyser, Berkhamsted, pp 99–100
Walter BA, Clayton DH (2005) Elaborate ornaments are costly to maintain: evidence for high maintenance handicaps. Behav Ecol 16:89–95
Wiki W (1991) Biological functions and activities of animal carotenoids. Pure Appl Chem 63:141–146
Zahavi A (1975) Mate selection—a selection for a handicap. J Theor Biol 53:205–214
Acknowledgements
We thank M. Adrián, O. González, P. Rodríguez, N. Varo and M. Vázquez for helping to capture flamingos and taking samples, and F. Cézilly, K. Delhey, K.J. McGraw, J.J. Negro, J.C. Senar and two referees for commenting on the manuscript. The Consejería de Medio Ambiente of the Junta de Andalucía and ‘Cañada de los Pájaros’ provided facilities. Funding was provided by Ministerio de Educación y Ciencia of Spain with EU–ERDF support (research grants BOS2002-04695 and CGL2005-01136/BOS).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J. Lindström
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 2252 kb)
Rights and permissions
About this article
Cite this article
Amat, J.A., Rendón, M.A., Garrido-Fernández, J. et al. Greater flamingos Phoenicopterus roseus use uropygial secretions as make-up. Behav Ecol Sociobiol 65, 665–673 (2011). https://doi.org/10.1007/s00265-010-1068-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-010-1068-z