Abstract
Purpose
The haematoma at a fracture site plays an important role in fracture healing. Previously, we demonstrated that a fracture haematoma contains multilineage mesenchymal progenitor cells. We postulated that the haematoma provided a source of chondrogenic cells for endochondral ossification during fracture healing and preservation of the cells contributed to biological fracture healing. In this study, we investigated whether haematoma-derived cells (HCs) could differentiate into hypertrophic chondrocytes and finally induce calcification of the extracellular matrix in vitro.
Methods
Fracture haematomas were obtained from four patients. HCs were cultured for five weeks under conditions that induce chondrogenic differentiation, followed by two weeks of hypertrophic induction using a pellet culture system. The pellets were analysed histologically and immunohistochemically. The gene expression levels of chondrogenic, hypertrophic, osteogenic, and angiogenic markers were measured by real-time PCR.
Results
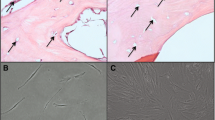
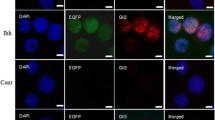
The histological and immunohistochemical analyses revealed that HCs differentiated into chondrocytes and hypertrophic chondrocytes, followed by calcification of the extracellular matrix. This sequential differentiation was also reflected in the gene expression profiles. After chondrogenic induction, expression of osteogenic and angiogenic markers was not significantly upregulated. However, the expression of these markers was significantly upregulated following hypertrophic induction. These in vitro observations mimicked the process of endochondral ossification during fracture healing.
Conclusions
Our results suggest that the fracture haematoma may offer a source of cells with chondrogenic potential that play key roles in endochondral ossification during fracture healing. These findings support the opinion that the haematoma should be preserved for biological fracture healing.


Similar content being viewed by others
References
Mizuno K, Mineo K, Tachibana T, Sumi M, Matsubara T, Hirohata K (1990) The osteogenetic potential of fracture hematoma: subperiosteal and intramuscular transplantation of the hematoma. J Bone Joint Surg Br 72:822–829
Marsell R, Einhorn TA (2011) The biology of fracture healing. Injury 42:551–555
Dimitriou R, Tsiridis E, Giannoudis PV (2005) Current concepts of molecular aspects of bone healing. Injury 36:1392–1404
Sarahrudi K, Mousavi M, Thomas A, Eipeldauer S, Vécsei V, Pietschmann P, Aharinejad S (2010) Elevated levels of macrophage colony: stimulating factor in human fracture healing. J Orthop Res 28:671–676
Kolar P, Gaber T, Perka C, Duda GN, Buttgereit F (2011) Human early fracture hematoma is characterized by inflammation and hypoxia. Clin Orthop Relat Res 469:3118–3126
Groothuis A, Duda GN, Wilson CJ, Thompson MS, Hunter MR, Simon P, Bail HJ, van Scherpenzeel KM, Kasper G (2010) Mechanical stimulation of the pro-angiogenic capacity of human fracture haematoma: involvement of VEGF mechano-regulation. Bone 47:438–444
Oe K, Miwa M, Sakai Y, Lee SY, Kuroda R, Kurosaka M (2007) An in vitro study demonstrating that haematomas found at the site of human fractures contain progenitor cells with multilineage capacity. J Bone Joint Surg Br 89:133–138
Bielby R, Jones E, McGonagle D (2007) The role of mesenchymal stem cells in maintenance and repair of bone. Injury 38:S26–S32
Ueno M, Uchida K, Takaso M, Minehara H, Suto K, Takahira N, Steck R, Schuetz MA, Itoman M (2011) Distribution of bone marrow-derived cells in the fracture callus during plate fixation in a green fluorescent protein-chimeric mouse model. Exp Anim 60:455–462
Ozaki A, Tsunoda M, Kinoshita S, Saura R (2000) Role of fracture hematoma and periosteum during fracture healing in rats: interaction of fracture hematoma and the periosteum in the initial step of the healing process. J Orthop Sci 5:64–70
Mackay AM, Beck SC, Murphy JM, Barry FP, Chichester CO, Pittenger MF (1998) Chondrogenic differentiation of cultured human mesenchymal stem cells from marrow. Tissue Eng 4:415–428
Mueller MB, Fischer M, Zellner J, Berner A, Dienstknecht T, Prantl L, Kujat R, Nerlich M, Tuan RS, Angele P (2010) Hypertrophy in mesenchymal stem cell chondrogenesis: effect of TGF-beta isoforms and chondrogenic conditioning. Cells Tissues Organs 192:158–166
Scotti C, Tonnarelli B, Papadimitropoulos A, Scherberich A, Schaeren S, Schauerte A, Lopez-Rios J, Zeller R, Barbero A, Martin I (2010) Recapitulation of endochondral bone formation using human adult mesenchymal stem cells as a paradigm for developmental engineering. Proc Natl Acad Sci USA 107:7251–7256
Hasegawa T, Miwa M, Sakai Y, Niikura T, Kurosaka M, Komori T (2009) Osteogenic activity of human fracture haematoma-derived progenitor cells is stimulated by low-intensity pulsed ultrasound in vitro. J Bone Joint Surg Br 91:264–270
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Ivkovic A, Marijanovic I, Hudetz D, Porter R, Pecina M, Evans C (2011) Regenerative medicine and tissue engineering in orthopaedic surgery. Front Biosci (Elite Ed) 3:923–944
Pecina M, Vukicevic S (2007) Biological aspects of bone, cartilage and tendon regeneration. Int Orthop 31:719–720
Borovecki F, Pecina-Slaus N, Vukicevic S (2007) Biological mechanisms of bone and cartilage remodelling—genomic perspective. Int Orthop 31:799–805
Behonick DJ, Xing ZQ, Lieu S, Buckley JM, Lotz JC, Marcucio RS, Werb Z, Miclau T, Colnot C (2007) Role of matrix metalloproteinase 13 in both endochondral and Intramembranous ossification during skeletal regeneration. PLoS One 2:e1150
D’Angelo M, Yan Z, Nooreyazdan M, Pacifici M, Sarment D, Billings PC, Leboy PS (2000) MMP-13 is induced during chondrocyte hypertrophy. J Cell Biochem 77:678–693
Winter A, Breit S, Parsch D, Benz K, Steck E, Hauner H, Weber RM, Ewerbeck V, Richter W (2003) Cartilage-like gene expression in differentiated human stem cell spheroids—a comparison of bone marrow-derived and adipose tissue-derived stromal cells. Arthritis Rheum 48:418–429
Wu CW, Tchetina EV, Mwale F, Hasty K, Pidoux I, Reiner A, Chen J, Van Wart HE, Poole AR (2002) Proteolysis involving matrix metalloproteinase 13 (collagenase-3) is required for chondrocyte differentiation that is associated with matrix mineralization. J Bone Miner Res 17:639–651
Baumgaertel F, Buhl M, Rahn BA (1998) Fracture healing in biological plate osteosynthesis. Injury 29:3–6
Farouk O, Krettek C, Miclau T, Schandelmaier P, Guy P, Tscherne H (1999) Minimally invasive plate osteosynthesis: does percutaneous plating disrupt femoral blood supply less than the traditional technique? J Orthop Trauma 13:401–406
Kesemenli C, Subasi M, Necmioglu S, Kapukaya A (2002) Treatment of multifragmentary fractures of the femur by indirect reduction (biological) and plate fixation. Injury 33:691–699
Collinge C, Kuper M, Larson K, Protzman R (2007) Minimally invasive plating of high-energy metaphyseal distal tibia fractures. J Orthop Trauma 21:355–361
Oh CW, Kyung HS, Park IH, Kim PT, Ihn JC (2003) Distal tibia metaphyseal fractures treated by percutaneous plate osteosynthesis. Clin Orthop Relat Res 408:286–291
Redfern DJ, Syed SU, Davies SJM (2004) Fractures of the distal tibia: minimally invasive plate osteosynthesis. Injury 35:615–620
Acknowledgments
The authors wish to thank Ms. M. Yasuda, Ms. K. Tanaka, and Ms. M. Nagata (Department of Orthopaedic Surgery, Kobe University Graduate School of Medicine) for their excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koga, T., Niikura, T., Lee, S.Y. et al. In vitro hypertrophy and calcification of human fracture haematoma-derived cells in chondrogenic differentiation. International Orthopaedics (SICOT) 37, 961–967 (2013). https://doi.org/10.1007/s00264-013-1813-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-013-1813-9