Abstract
Background
Although proteoglycan (PG) is one of the major components of cartilage matrices, its biological function is not fully elucidated.
Methods
The objectives of this study were to investigate the proliferation and differentiation of chondrocytes embedded in atelocollagen gel with exogenous cartilage PG (PG-atelocollagen gel) in vitro, and also to evaluate the repair of cartilage defects by PG-atelocollagen gel in vivo. In the in vitro study, rabbit chondrocytes were cultured in the PG-atelocollagen gel. Cell proliferation and mRNA expression levels were measured, and gels were histologically evaluated. In the in vivo study, cultured PG-atelocollagen gel containing chondrocytes were transplanted into full-thickness articular cartilage defects in rabbit knees, and evaluated macroscopically and histologically.
Results
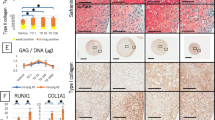
For the in vitro study, chondrocyte proliferation in 5.0 mg/ml PG-atelocollagen gel was enhanced, and the gene expression of Col2a1 and Aggrecan were decreased. In contrast, chondrocyte proliferation in 0.1 and 1.0 mg/ml PG-atelocollagen gel was not enhanced. The gene expression of Aggrecan in 0.1 and 1.0 mg/ml PG-atelocollagen gel was increased. For the in vivo study, the histological average total score of the 0.1 mg/ml PG-atelocollagen gel was significantly better than that of the group without PG.
Conclusions
Although the appropriate concentration of PG has not been defined, this study suggests the efficacy of PG for cartilage repair.





Similar content being viewed by others
References
Smith GD, Knutsen G, Richardson JB (2005) A clinical review of cartilage repair techniques. J Bone Joint Surg Br 87:445–449
Borovecki F, Pecina-Slaus N, Vukicevic S (2007) Biological mechanisms of bone and cartilage remodelling—genomic perspective. Int Orthop 31:799–805
Ivkovic A, Pascher A, Hudetz D, Maticic D, Jelic M, Dickinson S, Loparic M, Haspl M, Windhager R, Pecina M (2010) Articular cartilage repair by genetically modified bone marrow aspirate in sheep. Gene Ther 17:779–789
Ivkovic A, Marijanovic I, Hudetz D, Porter RM, Pecina M, Evans CH (2011) Regenerative medicine and tissue engineering in orthopaedic surgery. Front Biosci (Elite Ed) 3:923–944
Grande DA, Pitman MI, Peterson L, Menche D, Klein M (1989) The repair of experimentally produced defects in rabbit articular cartilage by autologous chondrocyte transplantation. J Orthop Res 7:208–218
Brittberg M, Lindahl A, Nilsson A, Ohlsson C, Isaksson O, Peterson L (1994) Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N Engl J Med 331:889–895
Peterson L, Minas T, Brittberg M, Lindahl A (2003) Treatment of osteochondritis dissecans of the knee with autologous chondrocyte transplantation: results at two to ten years. J Bone Joint Surg Am 85:17–24
Grigolo B, Roseti L, De Franceschi L, Piacentini A, Cattini L, Manfredini M, Faccini R, Facchini A (2005) Molecular and immunohistological characterization of human cartilage. Two years following autologous cell transplantation. J Bone Joint Surg Am 87:46–57
Buckwalter JA, Mankin HJ (1998) Articular cartilage: tissue design and chondrocyte-matrix interactions. Instr Course Lect 47:477–486
Yang BB, Zhang Y, Cao L, Yang BL (1998) Aggrecan and link protein affect cell adhesion to culture plates and to type II collagen. Matrix Biol 16:541–561
Funderburg FM, Markwald RR (1986) Conditioning of native substrates by chondroitin sulfate proteoglycans during cardiac mesenchymal cell migration. J Cell Biol 103:2475–2487
Zhang Y, Wu Y, Cao L, Lee V, Chen L, Lin Z, Kiani C, Adams ME, Yang BB (2001) Versican modulates embryonic chondrocyte morphology via the epidermal growth factor-like motifs in G3. Exp Cell Res 263:33–42
Kamiya N, Watanabe H, Habuchi H, Takagi H, Shinomura T, Shimizu K, Kimata K (2006) Versican/PG-M regulates chondrogenesis as an extracellular matrix molecule crucial for mesenchymal condensation. J Biol Chem 281:2390–2400
Watanabe H, Kimata K, Line S, Strong D, Gao LY, Kozak CA, Yamada Y (1994) Mouse cartilage matrix deficiency (cmd) caused by a 7 bp deletion in the aggrecan gene. Nat Genet 7:154–157
French MM, Smith SE, Akanbi K, Sanford T, Hecht J, Farach-Carson MC, Carson DD (1999) Expression of the heparan sulfate proteoglycan, perlecan, during mouse embryogenesis and perlecan chondrogenic activity in vitro. J Cell Biol 145:1103–1115
Nishimoto S, Takagi M, Wakitani S, Nihira T, Yoshida T (2005) Effect of chondroitin sulfate and hyaluronic acid on gene expression in a three-dimensional culture of chondrocytes. J Biosci Bioeng 100:123–126
Kagita E, Ikeda M, Wakitani S, Takagi M (2010) Effect of monosaccharides composing glycosaminoglycans on type 2 collagen accumulation in a three-dimensional culture of chondrocytes. J Biosci Bioeng 109:51–54
Wu CH, Ko CS, Huang JW, Huang HJ, Chu IM (2010) Effect of exogenous glycosaminoglycans on human chondrocytes cultivated on type II collagen scaffolds. J Mater Sci Mater Med 21:725–729
French MM, Gomes RR Jr, Timpl R, Höök M, Czymmek K, Farach-Carson MC, Carson DD (2002) Chondrogenic activity of the heparan sulfate proteoglycan perlecan maps to the N-terminal domain I. J Bone Miner Res 17:48–55
French MM, Rose S, Canseco J, Athanasiou KA (2004) Chondrogenic differentiation of adult dermal fibroblasts. Ann Biomed Eng 32:50–56
Uchio Y, Ochi M, Matsusaki M, Kurioka H, Katsube K (2000) Human chondrocyte proliferation and matrix synthesis cultured in Atelocollagen® gel. J Biomed Mater Res 50:138–143
Katsube K, Ochi M, Uchio Y, Maniwa S, Matsusaki M, Tobita M, Iwasa J (2000) Repair of articular cartilage defects with cultured chondrocytes in Atelocollagen gel. Comparison with cultured chondrocytes in suspension. Arch Orthop Trauma Surg 120:121–127
Ochi M, Uchio Y, Kawasaki K, Wakitani S, Iwasa J (2002) Transplantation of cartilage-like tissue made by tissue engineering in the treatment of cartilage defects of the knee. J Bone Joint Surg Br 84:571–578
Majima M, Takagaki K, Sudo S, Yoshihara S, Kudo Y, Yamagishi S (2001) Effect of proteoglycan on experimental colitis. In: Endo M, Harata S, Saito Y, Munakata A, Sasaki M, Tsuchida S (eds) New developments in glycomedicine. Elsevier Science BV, Amsterdam, pp 221–224
Wachsmuth L, Keiffer R, Juretschke HP, Raiss RX, Kimmig N, Lindhorst E (2003) In vivo contrast-enhanced micro MR-imaging of experimental osteoarthritis in the rabbit knee joint at 7.1T1. Osteoarthr Cartil 11:891–902
Wakitani S, Goto T, Pineda SJ, Young RG, Mansour JM, Caplan AI, Goldberg VM (1994) Mesenchymal cell-based repair of large, full-thickness defects of articular cartilage. J Bone Joint Surg Am 76:579–592
Tohyama H, Yasuda K, Minami A, Majima T, Iwasaki N, Muneta T, Sekiya I, Yagishita K, Takahashi S, Kurokouchi K, Uchio J, Iwasa J, Deie M, Adachi N, Sugawara K, Ochi M (2009) Atelocollagen-associated autologous chondrocyte implantation for the repair of chondral defects of the knee: a prospective multicenter clinical trial in Japan. J Orthop Sci 14:579–588
Zhang Y, Cao L, Kiani C, Yang BL, Hu W, Yang BB (1999) Promotion of chondrocyte proliferation by versican mediated by G1 domain and EGF-like motifs. J Cell Biochem 73:445–447
Acknowledgements
This work was supported in part by the Ministry of Education, Culture, Sports, Science and Technology Cooperation for innovation technology and advanced research in an urban area, the Hirosaki City Area (Proteoglycan application research project).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ohshika, S., Ishibashi, Y., Kon, A. et al. Potential of exogenous cartilage proteoglycan as a new material for cartilage regeneration. International Orthopaedics (SICOT) 36, 869–877 (2012). https://doi.org/10.1007/s00264-011-1335-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-011-1335-2