Abstract
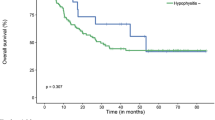
Combined immune checkpoint blockade (ICB) is effective therapy for renal cell carcinoma (RCC). However, the dynamic changes in circulating B cells induced by combined ICB have not been clarified. The present study prospectively examined 22 patients scheduled to receive ICB for unresectable or metastatic RCC between March 2018 and August 2021. Eleven patients received combined therapy with anti-PD-1 (nivolumab) and anti-CTLA-4 (ipilimumab), and the other 11 patients received nivolumab monotherapy. Comprehensive phenotypes of circulating immune cells obtained prior to and after ICB therapy were analyzed by flow cytometry. Although the proportion of naïve B cells among total B cells was significantly decreased, that of switched memory B cells was significantly increased after combined therapy. In responders, the proportion of B cells among peripheral blood mononuclear cells was significantly higher prior to ICB therapy, and the proportion of switched memory B cells among total B cells tended to increase after ICB therapy. Of note, the proportion of plasmablasts among total B cells was significantly increased after ICB therapy in patients who developed severe immune-related adverse events (irAEs), and the proportion of B cells among peripheral blood decreased significantly. Furthermore, in four of five patients who developed immune-related hypophysitis following combined therapy, anti-pituitary antibody was detected in the serum. These results suggested that immune-related hypophysitis was closely related to the increase in circulating plasmablasts. Collectively, this study suggests that combined ICB promotes the differentiation of B cell populations, which is associated with efficient tumor suppression and development of irAEs.







Similar content being viewed by others
Data availability
Data are available upon reasonable request.
References
Motzer RJ, Tannir NM, McDermott DF, Arén Frontera O, Melichar B, Choueiri TK, Plimack ER, Barthélémy P, Porta C, George S et al (2018) Nivolumab plus ipilimumab versus sunitinib in advanced renal-cell carcinoma. N Engl J Med 378:1277–1290. https://doi.org/10.1056/NEJMoa1712126
Motzer RJ, Escudier B, McDermott DF, George S, Hammers HJ, Srinivas S, Tykodi SS, Sosman JA, Procopio G, Plimack ER et al (2015) Nivolumab versus everolimus in advanced renal-cell carcinoma. N Engl J Med 373:1803–1813. https://doi.org/10.1056/NEJMoa1510665
Wang C, Thudium KB, Han M, Wang XT, Huang H, Feingersh D, Garcia C, Wu Y, Kuhne M, Srinivasan M et al (2014) In vitro characterization of the anti-PD-1 antibody nivolumab, BMS-936558, and in vivo toxicology in non-human primates. Cancer lmmunol Res 2:846–856. https://doi.org/10.1158/2326-6066.CIR-14-0040
Wolchok JD, Hoos A, O’Day S, Weber JS, Hamid O, Lebbé C, Maio M, Binder M, Bohnsack O, Nichol G et al (2009) Guidelines for the evaluation of immune therapy activity in solid tumors: immune-related response criteria. Clin Cancer Res 15:7412–7420. https://doi.org/10.1158/1078-0432.CCR-09-1624
Chen DS, Mellman I (2013) Oncology meets immunology: the cancer-immunity cycle. Immunity 39:1–10. https://doi.org/10.1016/j.immuni.2013.07.012
Helmink BA, Reddy SM, Gao J, Zhang S, Basar R, Thakur R, Yizhak K, Sade-Feldman M, Blando J, Han G et al (2020) B cells and tertiary lymphoid structures promote immunotherapy response. Nature 577:549–555. https://doi.org/10.1038/s41586-019-1922-8
Ohmura H, Yamaguchi K, Hanamura F, Ito M, Makiyama A, Uchino K, Shimokawa H, Tamura S, Esaki T, Mitsugi K et al (2020) OX40 and LAG3 are associated with better prognosis in advanced gastric cancer patients treated with anti-programmed death-1 antibody. Br J Cancer 122:1507–1517. https://doi.org/10.1038/s41416-020-0810-1
Yamaguchi K, Mishima K, Ohmura H, Hanamura F, Ito M, Nakano M, Tsuchihashi K, Ota SI, Wada N, Uchi H et al (2018) Activation of central/effector memory T cells and T-helper 1 polarization in malignant melanoma patients treated with anti-programmed death-1 antibody. Cancer Sci 109:3032–3042. https://doi.org/10.1111/cas.13758
Das R, Bar N, Ferreira M, Newman AM, Zhang L, Bailur JK, Bacchiocchi A, Kluger H, Wei W, Halaban R et al (2018) Early B cell changes predict autoimmunity following combination immune checkpoint blockade. J Clin Invest 128:715–720. https://doi.org/10.1172/JCI96798
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline version 1.1. Eur J Cancer 45:228–247. https://doi.org/10.1016/j.ejca.2008.10.026
Common Terminology Criteria for Adverse Events (CTCAE) Version 5. Published: November 27, 2017. US Department of Health and Human Services, National Institutes of Health, National Cancer Institute.
Ricciuti A, De Remigis A, Landek-Salgado MA, De Vincentiis L, Guaraldi F, Lupi I, Iwama S, Wand GS, Salvatori R, Caturegli P (2014) Detection of pituitary antibodies by immunofluorescence: approach and results in patients with pituitary diseases. J Clin Endocrinol Metab 99:1758–1766. https://doi.org/10.1210/jc.2014-1049
Kobayashi T, Iwama S, Sugiyama D, Yasuda Y, Okuji T, Ito M, Ito S, Sugiyama M, Onoue T, Takagi H et al (2021) Anti-pituitary antibodies and susceptible human leukocyte antigen alleles as predictive biomarkers for pituitary dysfunction induced by immune checkpoint inhibitors J Immunother. Cancer 9:e002493. https://doi.org/10.1136/jitc-2021-002493
Heng DY, Xie W, Regan MM, Harshman LC, Bjarnason GA, Vaishampayan UN, Mackenzie M, Wood L, Donskov F, Tan MH et al (2013) External validation and comparison with other models of the International Metastatic Renal-Cell Carcinoma Database Consortium prognostic model: a population-based study. Lancet Oncol 14:141–148. https://doi.org/10.1016/S1470-2045(12)70559-4
Motzer RJ, Jonasch E, Agarwal N, Alva A, Baine M, Beckermann K, Carlo MI, Choueiri TK, Costello BA, Derweesh IH et al (2022) Kidney cancer, version 3.2022, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw 20:71–90. https://doi.org/10.6004/jnccn.2022.0001
Kobayashi T, Iwama S, Yasuda Y, Okada N, Okuji T, Ito M, Onoue T, Goto M, Sugiyama M, Tsunekawa T et al (2020) Pituitary dysfunction induced by immune checkpoint inhibitors is associated with better overall survival in both malignant melanoma and non-small cell lung carcinoma: a prospective study. J Immunother Cancer 8:e000779. https://doi.org/10.1136/jitc-2020-000779
Freeman-Keller M, Kim Y, Cronin H, Richards A, Gibney G, Weber JS (2016) Nivolumab in resected and unresectable metastatic melanoma: characteristics of immune-related adverse events and association with outcomes. Clin Cancer Res 15(22):886–894. https://doi.org/10.1158/1078-0432.CCR-15-1136
Valeria H, Ilaria B, Marianna M, Sada V, Sbardella E, Isidori AM (2021) Non-canonical effects of ACTH: insights into adrenal insufficiency. Front Endocrinol 12:701263. https://doi.org/10.3389/fendo.2021.701263
Chen P-Y, Wu CY-J, Fang J-H, Chen HC, Feng LY, Huang CY, Wei KC, Fang JY, Lin CY (2019) Functional change of effector tumor-infiltrating CCR5 + CD38 + HLA-DR + CD8 + T cells in glioma microenvironment. Front Immunol 10:2395. https://doi.org/10.3389/fimmu.2019.02395
McCoy MJ, Nowak AK, van der Most RG, Dick IM, Lake RA (2013) Peripheral CD8(+) T cell proliferation is prognostic for patients with advanced thoracic malignancies. Cancer Immunol Immunother 62:529–539. https://doi.org/10.1007/s00262-012-1360-z
Finke JH, Rini B, Ireland J, Rayman P, Richmond A, Golshayan A, Wood L, Elson P, Garcia J, Dreicer R et al (2008) Sunitinib reverses type-1 immune suppression and decreases T-regulatory cells in renal cell carcinoma patients. Clin Cancer Res 14:6674–6682. https://doi.org/10.1158/1078-0432.CCR-07-5212
Ocadlikova D, Lecciso M, Broto JM, Scotlandi K, Cavo M, Curti A, Palmerini E et al (2021) Sunitinib exerts in vitro immunomodulatory activity on sarcomas via dendritic cells and synergizes with PD-1 blockade. Front Immunol 12:577766. https://doi.org/10.3389/fimmu.2021.577766
Huang B, Pan P-Y, Li Q, Sato AI, Levy DE, Bromberg J, Divino CM, Chen SH (2006) Gr-1+CD115+ immature myeloid suppressor cells mediate the development of tumor-induced T regulatory cells and T-cell anergy in tumor-bearing host. Cancer Res 66:1123–1131. https://doi.org/10.1158/0008-5472.CAN-05-1299
Wishnie AJ, Chwat-Edelstein T, Attaway M, Vuong BQ (2021) BCR Affinity influences T-b interactions and B cell development in secondary lymphoid organs. Front Immunol 12:703918. https://doi.org/10.3389/fimmu.2021.703918
Crotty S (2014) T follicular helper cell differentiation, function, and roles in disease. Immunity 41:529–542. https://doi.org/10.1016/j.immuni.2014.10.004
Kurata I, Matsumoto I, Ohyama A, Osada A, Ebe H, Kawaguchi H, Kaneko S, Kondo Y, Tsuboi H, Tomioka A et al (2019) Potential involvement of OX40 in the regulation of autoantibody sialylation in arthritis. Ann Rheum Dis 78:1488–1496. https://doi.org/10.1136/annrheumdis-2019-215195
Szabo K, Papp G, Barath S, Gyimesi E, Szanto A, Zeher M (2013) Follicular helper T cells may play an important role in the severity of primary Sjögren’s syndrome. Clin Immunol 147:95–104. https://doi.org/10.1016/j.clim.2013.02.024
Zhu S, Lin J, Qiao G, Wang X, Xu Y (2016) Tim-3 identifies exhausted follicular helper T cells in breast cancer patients. Immunobiology 221:986–993. https://doi.org/10.1016/j.imbio.2016.04.005
Zhou ZQ, Tong DN, Guan J, Tan HW, Zhao LD, Zhu Y, Yao J, Yang J, Zhang ZY et al (2016) Follicular helper T cell exhaustion induced by PD-L1 expression in hepatocellular carcinoma results in impaired cytokine expression and B cell help, and is associated with advanced tumor stages. Am J Transl Res 8:2926–2936
Sage PT, Paterson AM, Lovitch SB, Sharpe AH (2014) The coinhibitory receptor CTLA-4 controls B cell responses by modulating T follicular helper, T follicular regulatory, and T regulatory cells. Immunity 41:1026–1039. https://doi.org/10.1016/j.immuni.2014.12.005
Baumojohann D, Brossart P (2021) T follicular helper cells: linking cancer immunotherapy and immune-related adverse events. J Immunother Cancer 9:e002588. https://doi.org/10.1136/jitc-2021-002588
Iwama S, De Remigis A, Callahan MK, Slovin SF, Wolchok JD, Caturegli P (2014) Pituitary expression of CTLA-4 mediates hypophysitis secondary to administration of CTLA-4 blocking antibody. Sci Transl Med 6:230ra245. https://doi.org/10.1126/scitranslmed.3008002
Tahir SA, Gao J, Miura Y, Blando J, Tidwell RSS, Zhao H, Subudhi SK, Tawbi H, Keung E, Wargo J et al (2019) Autoimmune antibodies correlate with immune checkpoint therapy-induced toxicities. Proc Natl Acad Sci USA 116:22246–22251. https://doi.org/10.1073/pnas.1908079116
Seki M, Kitano S, Suzuki S (2022) Neurological disorders associated with immune checkpoint inhibitors: an association with autoantibodies. Cancer Immunol Immunother 71:769–775. https://doi.org/10.1007/s00262-021-03053-9
Hasan Ali O, Bomze D, Ring SS, Berner F, Fässler M, Diem S, Abdou MT, Hammers C, Emtenani S, Braun A et al (2020) BP180-specific IgG is associated with skin adverse events, therapy response, and overall survival in non-small cell lung cancer patients treated with checkpoint inhibitors. J Am Acad Dermatol 82:854–861. https://doi.org/10.1016/j.jaad.2019.08.045
Acknowledgements
We appreciate the technical assistance provided by the Department of Morphology Core Unit, Kyushu University Hospital, Fukuoka, Japan.
Funding
This study was supported by JSPS KAKENHI Grant Number JP20K08311.
Author information
Authors and Affiliations
Contributions
KU designed the study, performed the experiments, and analyzed all the data. KTa, KY, and HO assisted with the experiments. YM, ST, HS, YS, and RT treated the enrolled patients and collected the clinical data. HY and YO provided pituitary specimens. All authors discussed the data. KU, KTa, KY, and EB wrote the manuscript. All authors participated in revision of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
KTa received honoraria from Ono Pharmaceutical. KY received honoraria from Ono Pharmaceutical and Chugai Pharmaceutical. HO received honoraria from Chugai Pharmaceutical. YM received honoraria from Kyowa Kirin. KTu received Ono Pharmaceutical and Taiho Pharmaceutical. ST received honoraria from Bayer, Ono Pharmaceutical, Chugai Pharmaceutical, Eli Lilly, Merck, Daiichi-Sankyo, LSI Medience, and MSD. HS received honoraria from Ono Pharmaceutical, Eli Lilly, Merck. YS received honoraria from Eli Lilly, Daiichi-Sankyo and Kyowa Kirin. HK received honoraria from Chugai Pharmaceutical. HY received honoraria from AstraZeneca, Bayer, Chugai Pharmaceutical Novartis Pharmaceutical. KA received honoraria from BMS, Ono Pharmaceutical, Janssen Pharmaceutical, Chugai Pharmaceutical, Kyowa Kirin, AbbVie GK, and Eisai; and research grants from Chugai Pharmaceutical, Kyowa Kirin, AbbVie GK, Asahi-Kasei, and Takeda Pharmaceutical. EB received honoraria from Ono Pharmaceutical, Chugai Pharmaceutical, Eli Lilly, Taiho Pharmaceutical, Merck, Sanofi, MSD, Eisai, and Daiichi-Sankyo; and research grants from Taiho Pharmaceutical, Chugai Pharmaceutical, and Eli Lilly. The other authors have no competing interests.
Ethics approval
This study was approved by the Ethical Committee of Kyushu University Hospital (no. 29-348). Written informed consent was obtained from all patients prior to enrollment in the study.
Consent for publication
Not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Uehara, K., Tanoue, K., Yamaguchi, K. et al. Preferential B cell differentiation by combined immune checkpoint blockade for renal cell carcinoma is associated with clinical response and autoimmune reactions. Cancer Immunol Immunother 72, 3543–3558 (2023). https://doi.org/10.1007/s00262-023-03505-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-023-03505-4