Abstract
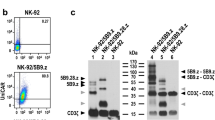
NK cells, especially FDA-approved NK-92 cells, could be used for TCR engineering owing to their specialized cytotoxicity against tumors, safety profile and potential use as an off-the-shelf cellular therapy. The TCR complex requires assembly of TCR- α/ β chains with CD3 molecules (CD3δ, CD3γ, CD3ε, CD3ζ) to be correctly expressed at the cell membrane, and yet NK cells lack expression of these CD3 subunits besides CD3ζ. Since transmembrane regions of TCR α and β chains are involved in TCR complex assembly, transmembrane regions of TCR replaced by CD28 transmembrane domain could result in the expression of TCR independent of its companion CD3 subunits. However, since the absence of CD3 signaling components can influence the transmission of TCR signals to NK cells, it is necessary to add the signaling molecules of NK cells followed by CD28 transmembrane domain. Both CD3ζ and DAP10 play an important role in the activation and cytotoxicity of NK cells; moreover, 2B4 and 4-1BB are the main costimulatory molecules in NK cells. Therefore, we designed a chimeric TCR that consisted of the extracellular domains of the TCR α and β chains specific for NYESO-1 fused to the CD28 transmembrane domain followed by the 41BB and CD3ζ signaling domains as well as the 2B4 and DAP10 signaling domain, respectively. The chimeric TCR genetically engineered NK-92 cells exhibit antigen-specific recognition and lysis of tumor cells both in vitro and in vivo. In addition, TCR-28-2B10/BBζ can be feasibly expressed in primary NK cells and exhibit antigen-reactive recognition and effect function. The overall encouraging data highlight the value of NK-92 cells and primary NK cells engineered to express therapeutic chimeric TCR for adoptive immunotherapies.


Similar content being viewed by others
Data availability
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
References
Sadelain M, Riviere I, Riddell S (2017) Therapeutic T cell engineering. Nature 545(7655):423–431
Daher M, Rezvani K (2020) Outlook for new CAR-based therapies with a focus on CAR NK cells: what lies beyond CAR-engineered t cells in the race against cancer. Cancer discov 1:45–58
Parlar A, Sayitoglu EC, Ozkazanc D, Georgoudaki AM, Pamukcu C, Aras M et al (2019) Engineering antigen-specific NK cell lines against the melanoma-associated antigen tyrosinase via TCR gene transfer. Eur J Immunol 49(8):1278–1290
Mensali N, Dillard P, Hebeisen M, Lorenz S, Theodossiou T, Myhre MR et al (2019) NK cells specifically TCR-dressed to kill cancer cells. EBioMedicine 40:106–117
Liu E, Marin D, Banerjee P, Macapinlac HA, Thompson P, Basar R et al (2020) Use of CAR-Transduced Natural Killer Cells in CD19-Positive Lymphoid Tumors. N Engl J Med 382(6):545–553
Morton LT, Wachsmann TL, Meeuwsen MH, Wouters AK, Remst DF, van Loenen MM, Falkenburg JF, Heemskerk MH (2022) T cell receptor engineering of primary NK cells to therapeutically target tumors and tumor immune evasion. J immunother cancer 10(3):e003715
Shimasaki N, Jain A, Campana D (2020) NK cells for cancer immunotherapy. Nat Rev Drug Discovery 19(3):200–218
Milone MC, Xu J, Chen SJ, Collins MA, Zhou J, Powell DJ Jr et al (2021) Engineering enhanced CAR T-cells for improved cancer therapy. Nat Cancer 2(8):780–793
Imai C, Iwamoto S, Campana D (2005) Genetic modification of primary natural killer cells overcomes inhibitory signals and induces specific killing of leukemic cells. Blood 106(1):376–383
Xu Y, Liu Q, Zhong M, Wang Z, Chen Z, Zhang Y et al (2019) 2B4 costimulatory domain enhancing cytotoxic ability of anti-CD5 chimeric antigen receptor engineered natural killer cells against T cell malignancies. J Hematol Oncol 12(1):49
Tan Q, Zhang C, Yang W, Liu Y, Heyilimu P, Feng D et al (2019) Isolation of T cell receptor specifically reactive with autologous tumour cells from tumour-infiltrating lymphocytes and construction of T cell receptor engineered T cells for esophageal squamous cell carcinoma. J Immunother Cancer 7(1):232
Zhang C, Tan Q, Li S, Shen L, Zhang J, Liu Y et al (2021) Induction of EBV latent membrane protein-2A (LMP2A)-specific T cells and construction of individualized TCR-engineered T cells for EBV-associated malignancies. J Immunother Cancer. 9(7):e002516
Acknowledgements
Not applicable.
Funding
This work was supported by Natural Science Foundation of China [Grant No 81972880–ZL, Grant No 82003246–CZ]; Capital’s Funds for Health Improvement and Research (Grant No 2022-1-1022–ZL, 2020-4-1028–CZ); Open Project funded by Key laboratory of Carcinogenesis and Translational Research, Ministry of Education/Beijing (2022 Open Project-1); Cooperation Fund of Beijing Cancer Hospital and Beijing Institute for Cancer Research; Clinical Medicine Plus X—Young Scholars Project (PKU2022LCXQ036), Peking University, the Fundamental Research Funds for the Central Universities.
Author information
Authors and Affiliations
Contributions
ZML, BTY and CTZ designed the research; SCL, CTZ, LYS, XT and YFX conducted experiments; SCL, CTZ, and ZML analyzed data; and CTZ, ZML and SCL wrote the paper.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval
This project was approved by the Institutional Review Board of the Peking University School of Oncology, China. The patient gave written informed consent for the use of their tumor tissues for research purposes.
Consent for publication
All authors have approved for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, S., Zhang, C., Shen, L. et al. TCR extracellular domain genetically linked to CD28, 2B4/41BB and DAP10/CD3ζ -engineered NK cells mediates antitumor effects. Cancer Immunol Immunother 72, 769–774 (2023). https://doi.org/10.1007/s00262-022-03275-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-022-03275-5