Abstract
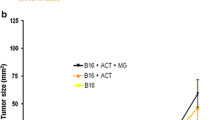
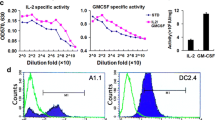
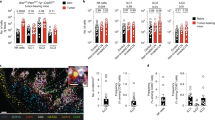
Interleukin-33 (IL-33) regulates innate and acquired immune response to pathogens, self-antigens and tumors. IL-33 effects on tumors depend on the dose and mode of administration along with the type of malignancy. We studied the effects of IL-33 on the development of primary and metastatic melanoma induced by B16-F1 cell line in C57BL/6 mice. Intraperitoneally applied IL-33 restricts primary tumor growth. When administered intranasally 3 days prior to the intravenous injection of the tumor cells, IL-33 promoted growth of B16-F1 melanoma metastases, while B16-F10 gave massive metastases independently of IL-33. To mimic natural dissemination, we next used a limited number (5 × 104) of B16-F1 cells intravenously followed by application of IL-33 intraperitoneally. IL-33 increased the size of metastases (10.96 ± 3.96 mm2) when compared to the control group (0.86 ± 0.39 mm2), without changing incidence and number of metastases. IL-33 increased expression of ST2 on both tumor and immune cells in metastases. Also, IL-33 enhanced eosinophils and anti-tumor NK cells in the lung. The striking finding was reduced cytotoxicity of CD8+ T cells derived from metastatic lung of IL-33 injected mice. IL-33 reduced the percentage of TNF-α+ and IFN-γ+ CD8+ T cells while increasing the frequency of CD8+ T cells that express inhibitory molecules (PD-1, KLRG-1 and CTLA-4). There was a significant accumulation of CD11b+Gr-1+ myeloid suppressor cells and FoxP3+, IL-10+ and CTLA-4+ regulatory T cells in the metastatic lung of IL-33 injected mice. The relevance of IL-33 for melanoma metastases was also documented in a significantly increased level of serum IL-33 in stage III melanoma patients.






Similar content being viewed by others
Abbreviations
- AJCC:
-
American Joint Committee on Cancer
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- KLRG1:
-
Killer cell lectin-like receptor subfamily G member 1
- TAP1:
-
Transporter associated with antigen processing 1
References
Hsu MY, Meier F, Herlyn M (2002) Melanoma development and progression: a conspiracy between tumor and host. Differentiation 70(9–10):522–536
Sandru A, Voinea S, Panaitescu E, Blidaru A (2014) Survival rates of patients with metastatic malignant melanoma. J Med Life 7(4):572–576
Dummer R, Hauschild A, Lindenblatt N, Pentheroudakis G, Keilholz U (2015) Cutaneous melanoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 26(Suppl 5):v126–v132
Rosenberg SA (2001) Progress in human tumour immunology and immunotherapy. Nature 411(6835):380–384
Molofsky AB, Savage AK, Locksley RM (2015) Interleukin-33 in tissue homeostasis, injury, and inflammation. Immunity 42(6):1005–1019
Liew FY, Girard JP, Turnquist HR (2016) Interleukin-33 in health and disease. Nat Rev Immunol 16(11):676–689
Milovanovic M, Volarevic V, Radosavljevic G, Jovanovic I, Pejnovic N, Arsenijevic N, Lukic ML (2012) IL-33/ST2 axis in inflammation and immunopathology. Immunol Res 52(1–2):89–99
Jovanovic I, Radosavljevic G, Mitrovic M, Juranic VL, McKenzie AN, Arsenijevic N, Jonjic S, Lukic ML (2011) ST2 deletion enhances innate and acquired immunity to murine mammary carcinoma. Eur J Immunol 41(7):1902–1912
Jovanovic IP, Pejnovic NN, Radosavljevic GD, Pantic JM, Milovanovic MZ, Arsenijevic NN, Lukic ML (2014) Interleukin-33/ST2 axis promotes breast cancer growth and metastases by facilitating intratumoral accumulation of immunosuppressive and innate lymphoid cells. Int J Cancer 134(7):1669–1682
Milosavljevic MZ, Jovanovic IP, Pejnovic NN, Mitrovic SL, Arsenijevic NN, Simovic Markovic BJ, Lukic ML (2016) Deletion of IL-33R attenuates VEGF expression and enhances necrosis in mammary carcinoma. Oncotarget 7(14):18106–18115
Schmieder A, Multhoff G, Radons J (2012) Interleukin-33 acts as a pro-inflammatory cytokine and modulates its receptor gene expression in highly metastatic human pancreatic carcinoma cells. Cytokine 60(2):514–521
Sun P, Ben Q, Tu S, Dong W, Qi X, Wu Y (2011) Serum interleukin-33 levels in patients with gastric cancer. Dig Dis Sci 56(12):3596–3601
Cui G, Qi H, Gundersen MD, Yang H, Christiansen I, Sorbye SW, Goll R, Florholmen J (2015) Dynamics of the IL-33/ST2 network in the progression of human colorectal adenoma to sporadic colorectal cancer. Cancer Immunol Immunother 64(2):181–190
Liu X, Zhu L, Lu X, Bian H, Wu X, Yang W, Qin Q (2014) IL-33/ST2 pathway contributes to metastasis of human colorectal cancer. Biochem Biophys Res Commun 453(3):486–492
Wang C, Chen Z, Bu X, Han Y, Shan S, Ren T, Song W (2016) IL-33 signaling fuels outgrowth and metastasis of human lung cancer. Biochem Biophys Res Commun 479(3):461–468
Wang K, Shan S, Yang Z, Gu X, Wang Y, Wang C, Ren T (2017) IL-33 blockade suppresses tumor growth of human lung cancer through direct and indirect pathways in a preclinical model. Oncotarget 8(40):68571–68582
Brunner SM, Rubner C, Kesselring R, Martin M, Griesshammer E, Ruemmele P, Stempfl T, Teufel A, Schlitt HJ, Fichtner-Feigl S (2015) Tumor-infiltrating, interleukin-33-producing effector-memory CD8(+) T cells in resected hepatocellular carcinoma prolong patient survival. Hepatology 61(6):1957–1967
Yang M, Feng Y, Yue C, Xu B, Chen L, Jiang J, Lu B, Zhu Y (2018) Lower expression level of IL-33 is associated with poor prognosis of pulmonary adenocarcinoma. PLoS ONE 13(3):e0193428
Saranchova I, Han J, Huang H, Fenninger F, Choi KB, Munro L, Pfeifer C, Welch I, Wyatt AW, Fazli L, Gleave ME, Jefferies WA (2016) Discovery of a metastatic immune escape mechanism initiated by the loss of expression of the tumour biomarker interleukin-33. Sci Rep 6:30555
Kim J, Kim W, Moon UJ, Kim HJ, Choi HJ, Sin JI, Park NH, Cho HR, Kwon B (2016) Intratumorally establishing type 2 innate lymphoid cells blocks tumor growth. J Immunol 196(5):2410–2423
Gao X, Wang X, Yang Q, Zhao X, Wen W, Li G, Lu J, Qin W, Qi Y, Xie F, Jiang J, Wu C, Zhang X, Chen X, Turnquist H, Zhu Y, Lu B (2015) Tumoral expression of IL-33 inhibits tumor growth and modifies the tumor microenvironment through CD8+ T and NK cells. J Immunol 194(1):438–445
Dominguez D, Ye C, Geng Z, Chen S, Fan J, Qin L, Long A, Wang L, Zhang Z, Zhang Y, Fang D, Kuzel TM, Zhang B (2017) Exogenous IL-33 restores dendritic cell activation and maturation in established cancer. J Immunol 198(3):1365–1375
Li X, Lv Q, Feng Y, Gu Y, Xia R, Ma J, He H, Zhu Y (2018) Interleukin-33, a potential cytokine expressed in tumor microenvironment involves in antitumor immunotherapy through facilitates CD8+ T Cells. J Interferon Cytokine Res 38(11):491–499
Gao K, Li X, Zhang L, Bai L, Dong W, Gao K, Shi G, Xia X, Wu L, Zhang L (2013) Transgenic expression of IL-33 activates CD8(+) T cells and NK cells and inhibits tumor growth and metastasis in mice. Cancer Lett 335(2):463–471
Lucarini V, Ziccheddu G, Macchia I, La Sorsa V, Peschiaroli F, Buccione C, Sistigu A, Sanchez M, Andreone S, D’Urso MT, Spada M, Macchia D, Afferni C, Mattei F, Schiavoni G (2017) IL-33 restricts tumor growth and inhibits pulmonary metastasis in melanoma-bearing mice through eosinophils. Oncoimmunology 6(6):e1317420
Radosavljevic G, Jovanovic I, Majstorovic I, Mitrovic M, Lisnic VJ, Arsenijevic N, Jonjic S, Lukic ML (2011) Deletion of galectin-3 in the host attenuates metastasis of murine melanoma by modulating tumor adhesion and NK cell activity. Clin Exp Metastasis 28(5):451–462
Fidler IJ (1973) Selection of successive tumor lines for metastasis. Nature New Biol 242(118):148–149
Petersen RP, Hanish S, Haney JC, Miller CC 3rd, Burfeind WR Jr, Tyler DS, Seigler HF, Wolfe W, D’Amico TA, Harpole DH Jr (2007) Improved survival with pulmonary metastasectomy: an analysis of 1720 patients with pulmonary metastatic melanoma. J Thorac Cardiovasc Surg 133(1):104–110
Dao DT, Vuong JT, Anez-Bustillos L, Pan A, Mitchell PD, Fell GL, Baker MA, Bielenberg DR, Puder M (2018) Intranasal delivery of VEGF enhances compensatory lung growth in mice. PLoS ONE 13(6):e0198700
Martin NT, Martin MU (2016) Interleukin 33 is a guardian of barriers and a local alarmin. Nat Immunol 17(2):122–131
Liew FY (2012) IL-33: a Janus cytokine. Ann Rheum Dis 71(Suppl 2):i101–i104
Oboki K, Nakae S, Matsumoto K, Saito H (2011) IL-33 and airway inflammation. Allergy Asthma Immunol Res 3(2):81–88
Zhiguang X, Wei C, Steven R, Wei D, Wei Z, Rong M, Zhanguo L, Lianfeng Z (2010) Over-expression of IL-33 leads to spontaneous pulmonary inflammation in mIL-33 transgenic mice. Immunol Lett 131(2):159–165
Ibegbu CC, Xu YX, Harris W, Maggio D, Miller JD, Kourtis AP (2005) Expression of killer cell lectin-like receptor G1 on antigen-specific human CD8+ T lymphocytes during active, latent, and resolved infection and its relation with CD57. J Immunol 174(10):6088–6094
Li L, Wan S, Tao K, Wang G, Zhao E (2016) KLRG1 restricts memory T cell antitumor immunity. Oncotarget 7(38):61670–61678
Furuta J, Inozume T, Harada K, Shimada S (2014) CD271 on melanoma cell is an IFN-γ-inducible immunosuppressive factor that mediates downregulation of melanoma antigens. J Invest Dermatol 134(5):1369–1377
Lin L, Rayman P, Pavicic PG Jr, Tannenbaum C, Hamilton T, Montero A, Ko J, Gastman B, Finke J, Ernstoff M, Diaz-Montero CM (2019) Ex vivo conditioning with IL-12 protects tumor-infiltrating CD8+ T cells from negative regulation by local IFN-γ. Cancer Immunol Immunother 68(3):395–405
Wang JM, Cheng YQ, Shi L, Ying RS, Wu XY, Li GY, Moorman JP, Yao ZQ (2013) KLRG1 negatively regulates natural killer cell functions through the Akt pathway in individuals with chronic hepatitis C virus infection. J Virol 87(21):11626–11636
Paczesny S, Banchereau J, Wittkowski KM, Saracino G, Fay J, Palucka AK (2004) Expansion of melanoma-specific cytolytic CD8+ T cell precursors in patients with metastatic melanoma vaccinated with CD34+ progenitor-derived dendritic cells. J Exp Med 199(11):1503–1511
Le Gros G, Erard F (1994) Non-cytotoxic, IL-4, IL-5, IL-10 producing CD8+ T cells: their activation and effector functions. Curr Opin Immunol 6(3):453–457
Seidel JA, Otsuka A, Kabashima K (2018) Anti-PD-1 and anti-CTLA-4 therapies in cancer: mechanisms of action, efficacy, and limitations. Front Oncol 8:86
Liu H, Weber A, Morse J, Kodumudi K, Scott E, Mullinax J, Sarnaik AA, Pilon-Thomas S (2018) Cell mediated immunity after combination therapy with intralesional PV-10 and blockade of the PD-1/PD-L1 pathway in a murine melanoma model. PLoS ONE 13(4):e0196033
Wen YH, Lin HQ, Li H, Zhao Y, Lui VWY, Chen L, Wu XM, Sun W, Wen WP (2019) Stromal interleukin-33 promotes regulatory T cell-mediated immunosuppression in head and neck squamous cell carcinoma and correlates with poor prognosis. Cancer Immunol Immunother 68(2):221–232
Xiao P, Wan X, Cui B, Liu Y, Qiu C, Rong J, Zheng M, Song Y, Chen L, He J, Tan Q, Wang X, Shao X, Liu Y, Cao X, Wang Q (2015) Interleukin 33 in tumor microenvironment is crucial for the accumulation and function of myeloid-derived suppressor cells. Oncoimmunology 5(1):e1063772
Dennis KL, Blatner NR, Gounari F, Khazaie K (2013) Current status of interleukin-10 and regulatory T cells in cancer. Curr Opin Oncol 25(6):637–645
Acknowledgements
The authors thank Professor Milan Knezevic for great help in pathohistological analyses and Aleksandar Arsenijevic, Aleksandar Ilic and Dusan Tomasevic for excellent technical assistance.
Funding
This work was funded by grants from the Ministry of Education, Science and Technological Development, Serbia (ON 175071, ON 175069 and ON 175103), a bilateral project with People’s Republic of China (06/2018) and by the Faculty of Medical Sciences of the University of Kragujevac, Serbia (MP 02/14).
Author information
Authors and Affiliations
Contributions
AJ performed most of the experiments, analyzed the data and drafted the manuscript. GDR contributed to the design and interpretation of the experiments, analyzed the data, generated figures and revised the manuscript. JP contributed to the design and interpretation of the experiments, assisted and/or performed some of the experiments, analyzed the data and revised the manuscript. IJ, MM and DV assisted and/or performed some of the experiments and analyzed the data. IS oversaw the consent and contributed to the collection of human samples. NA and MLL coordinated the design and execution of the experiments, data analysis, and manuscript drafting and revision. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Cell line authentication
The murine melanoma B16-F1 and B16-F10 cell lines, purchased from American Type Culture Collection (ATCC, Manassas, USA), were routinely cultured. Cell culture was performed under standardized protocols to ensure that phenotypically similar cells are implanted during each experiment. Viability of cells was determined by trypan blue exclusion and only cell suspensions with > 95% viable cells were used.
Ethical approval and ethical standards
All animal care procedures and experimental protocols were performed in accordance with institutional and the official guidelines of EU Directive 2010/63/EU, at the Faculty of Medical Sciences, University of Kragujevac, Serbia. The experiments were approved by the Animal Ethics Board of the Faculty of Medical Sciences, University of Kragujevac, Serbia (01-2588, 17/03/2014). Consented melanoma patients were recruited from the Clinics of Dermatovenerology and Plastic Surgery, Military Medical Academy, Belgrade, Serbia, while healthy donors (with no prior history of cancer) were recruited on periodical systematic examinations. The study was approved by the local Research Ethics Committee, Military Medical Academy (11-03/2014).
Animal source
C57BL/6 mice were bred in animal breeding facilities of the Faculty of Medical Sciences, University of Kragujevac, Serbia.
Informed consent
Written informed consent was obtained prior to sample collection from all individual participants included in the study in accordance with the Declaration of Helsinki.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jevtovic, A., Pantic, J., Jovanovic, I. et al. Interleukin-33 pretreatment promotes metastatic growth of murine melanoma by reducing the cytotoxic capacity of CD8+ T cells and enhancing regulatory T cells. Cancer Immunol Immunother 69, 1461–1475 (2020). https://doi.org/10.1007/s00262-020-02522-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-020-02522-x