Abstract
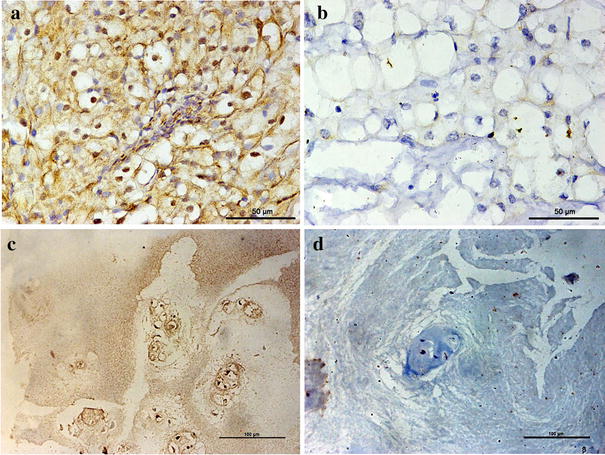
Chordoma is difficult to eradicate due to high local recurrence rates. The immune microenvironment is closely associated with tumor prognosis; however, its role in skull base chordoma is unknown. The expression of Galectin-9 (Gal9) and tumor-infiltrating lymphocyte (TIL) markers was assessed by immunohistochemistry. Kaplan–Meier and multivariate Cox analyses were used to assessing local recurrence-free survival (LRFS) and overall survival (OS) of patients. MiR-455-5p was identified as a regulator of Gal9 expression. Immunopositivity for Gal9 was associated with tumor invasion (p = 0.019), Karnofsky performance status (KPS) score (p = 0.017), and total TIL count (p < 0.001); downregulation of miR-455-5p was correlated with tumor invasion (p = 0.017) and poor prognosis; and the T-cell immunoglobulin and mucin-domain 3 (TIM3)+ TIL count was associated with chordoma invasion (p = 0.010) and KPS score (p = 0.037). Furthermore, multivariate analysis indicated that only TIM3+ TIL density was an independent prognostic factor for LRFS (p = 0.010) and OS (p = 0.016). These results can be used to predict clinical outcome and provide a basis for immune therapy in skull base chordoma patients.



Similar content being viewed by others
Abbreviations
- ATCC:
-
American Type Culture Collection
- C-Cbl:
-
C-Casitas B lineage lymphoma
- CD8:
-
Cluster of differentiation 8
- CT:
-
Computed tomography
- CTLA-4:
-
Cytolytic T lymphocyte-associated Ag-4
- Gal9:
-
Galectin-9
- HE:
-
Hematoxylin and eosin
- KPS:
-
Karnofsky performance status
- LRFS:
-
Local recurrence-free survival
- MiRNAs:
-
MicroRNAs
- NC:
-
Negative control
- qPCR:
-
Quantitative real-time polymerase chain reaction
- SOX9:
-
Sex-determining region Y (SRY)-box 9
- TIM3:
-
T-cell immunoglobulin and mucin-domain 3
References
McMaster ML, Goldstein AM, Bromley CM, Ishibe N, Parry DM (2001) Chordoma: incidence and survival patterns in the United States, 1973–1995. Cancer Causes Control 12(1):1–11
Heffelfinger MJ, Dahlin DC, MacCarty CS, Beabout JW (1973) Chordomas and cartilaginous tumors at the skull base. Cancer 32(2):410–420
Salisbury JR (1993) The pathology of the human notochord. J Pathol 171(4):253–255. https://doi.org/10.1002/path.1711710404
Stacchiotti S, Sommer J (2015) Building a global consensus approach to chordoma: a position paper from the medical and patient community. Lancet Oncol 16(2):e71–e83. https://doi.org/10.1016/s1470-2045(14)71190-8
Chibbaro S, Cornelius JF, Froelich S, Tigan L, Kehrli P, Debry C, Romano A, Herman P, George B, Bresson D (2014) Endoscopic endonasal approach in the management of skull base chordomas—clinical experience on a large series, technique, outcome, and pitfalls. Neurosurg Rev 37(2):217–224. https://doi.org/10.1007/s10143-013-0503-9 (discussion 224–215)
Di Maio S, Rostomily R, Sekhar LN (2012) Current surgical outcomes for cranial base chordomas: cohort study of 95 patients. Neurosurgery 70(6):1355–1360. https://doi.org/10.1227/neu.0b013e3182446783 (discussion 1360)
Hines JP, Ashmead MG, Stringer SP (2014) Clival chordoma of the nasal septum secondary to surgical pathway seeding. Am J Otolaryngol 35(3):431–434. https://doi.org/10.1016/j.amjoto.2013.12.018
Koutourousiou M, Gardner PA, Tormenti MJ, Henry SL, Stefko ST, Kassam AB, Fernandez-Miranda JC, Snyderman CH (2012) Endoscopic endonasal approach for resection of cranial base chordomas: outcomes and learning curve. Neurosurgery 71(3):614–624. https://doi.org/10.1227/neu.0b013e31825ea3e0 (discussion 624–615)
Jackson CM, Lim M, Drake CG (2014) Immunotherapy for brain cancer: recent progress and future promise. Clin Cancer Res 20(14):3651–3659. https://doi.org/10.1158/1078-0432.CCR-13-2057
Dougan M, Dranoff G (2009) Immune therapy for cancer. Annu Rev Immunol 27:83–117. https://doi.org/10.1146/annurev.immunol.021908.132544
Sharma P, Allison JP (2015) Immune checkpoint targeting in cancer therapy: toward combination strategies with curative potential. Cell 161(2):205–214. https://doi.org/10.1016/j.cell.2015.03.030
Garber ST, Hashimoto Y, Weathers SP, Xiu J, Gatalica Z, Verhaak RG, Zhou S, Fuller GN, Khasraw M, de Groot J, Reddy SK, Spetzler D, Heimberger AB (2016) Immune checkpoint blockade as a potential therapeutic target: surveying CNS malignancies. Neurooncology 18(10):1357–1366. https://doi.org/10.1093/neuonc/now132
Lim M, Xia Y, Bettegowda C, Weller M (2018) Current state of immunotherapy for glioblastoma. Nat Rev Clin Oncol. https://doi.org/10.1038/s41571-018-0003-5
Sounni NE, Noel A (2013) Targeting the tumor microenvironment for cancer therapy. Clin Chem 59(1):85–93. https://doi.org/10.1373/clinchem.2012.185363
Nirschl CJ, Drake CG (2013) Molecular pathways: coexpression of immune checkpoint molecules: signaling pathways and implications for cancer immunotherapy. Clin Cancer Res 19(18):4917–4924. https://doi.org/10.1158/1078-0432.CCR-12-1972
Hiraoka K, Miyamoto M, Cho Y, Suzuoki M, Oshikiri T, Nakakubo Y, Itoh T, Ohbuchi T, Kondo S, Katoh H (2006) Concurrent infiltration by CD8+ T cells and CD4+ T cells is a favourable prognostic factor in non-small-cell lung carcinoma. Br J Cancer 94(2):275–280. https://doi.org/10.1038/sj.bjc.6602934
Shang B, Liu Y, Jiang SJ, Liu Y (2015) Prognostic value of tumor-infiltrating FOXp3+ regulatory T cells in cancers: a systematic review and meta-analysis. Sci Rep 5:15179. https://doi.org/10.1038/srep15179
Seo AN, Lee HJ, Kim EJ, Kim HJ, Jang MH, Lee HE, Kim YJ, Kim JH, Park SY (2013) Tumour-infiltrating CD8+ lymphocytes as an independent predictive factor for pathological complete response to primary systemic therapy in breast cancer. Br J Cancer 109(10):2705–2713. https://doi.org/10.1038/bjc.2013.634
Li G, Wang Z, Zhang C, Liu X, Cai J, Wang Z, Hu H, Wu F, Bao Z, Liu Y, Zhao L, Liang T, Yang F, Huang R, Zhang W, Jiang T (2017) Molecular and clinical characterization of TIM-3 in glioma through 1,024 samples. Oncoimmunology 6(8):e1328339. https://doi.org/10.1080/2162402X.2017.1328339
Zhu S, Lin J, Qiao G, Wang X, Xu Y (2016) Tim-3 identifies exhausted follicular helper T cells in breast cancer patients. Immunobiology 221(9):986–993. https://doi.org/10.1016/j.imbio.2016.04.005
Tan Y, Trent JC, Wilky BA, Kerr DA, Rosenberg AE (2017) Current status of immunotherapy for gastrointestinal stromal tumor. Cancer Gene Ther 24(3):130–133. https://doi.org/10.1038/cgt.2016.58
Monney L, Sabatos CA, Gaglia JL, Ryu A, Waldner H, Chernova T, Manning S, Greenfield EA, Coyle AJ, Sobel RA, Freeman GJ, Kuchroo VK (2002) Th1-specific cell surface protein Tim-3 regulates macrophage activation and severity of an autoimmune disease. Nature 415(6871):536–541. https://doi.org/10.1038/415536a
Zhu C, Anderson AC, Schubart A, Xiong H, Imitola J, Khoury SJ, Zheng XX, Strom TB, Kuchroo VK (2005) The Tim-3 ligand galectin-9 negatively regulates T helper type 1 immunity. Nat Immunol 6(12):1245–1252. https://doi.org/10.1038/ni1271
Sharma P, Allison JP (2015) Immune checkpoint targeting in cancer therapy: toward combination strategies with curative potential. Cell 161(2):205–214. https://doi.org/10.1016/j.cell.2015.03.030
Ngiow SF, Teng MW, Smyth MJ (2011) Prospects for TIM3-targeted antitumor immunotherapy. Can Res 71(21):6567–6571. https://doi.org/10.1158/0008-5472.CAN-11-1
Ferris RL, Lu B, Kane LP (2014) Too much of a good thing? Tim-3 and TCR signaling in T cell exhaustion. J Immunol 193(4):1525–1530. https://doi.org/10.4049/jimmunol.1400557
Liu Z, Han H, He X, Li S, Wu C, Yu C, Wang S (2016) Expression of the galectin-9-Tim-3 pathway in glioma tissues is associated with the clinical manifestations of glioma. Oncol Lett 11(3):1829–1834. https://doi.org/10.3892/ol.2016.4142
Li X, Chen Y, Liu X, Zhang J, He X, Teng G, Yu D (2017) Tim3/Gal9 interactions between T cells and monocytes result in an immunosuppressive feedback loop that inhibits Th1 responses in osteosarcoma patients. Int Immunopharmacol 44:153–159. https://doi.org/10.1016/j.intimp.2017.01.006
Komita H, Koido S, Hayashi K, Kan S, Ito M, Kamata Y, Suzuki M, Homma S (2015) Expression of immune checkpoint molecules of T cell immunoglobulin and mucin protein 3/galectin-9 for NK cell suppression in human gastrointestinal stromal tumors. Oncol Rep 34(4):2099–2105. https://doi.org/10.3892/or.2015.4149
Farazi TA, Spitzer JI, Morozov P, Tuschl T (2011) miRNAs in human cancer. J Pathol 223(2):102–115. https://doi.org/10.1002/path.2806
Le XF, Merchant O, Bast RC, Calin GA (2010) The roles of MicroRNAs in the cancer invasion-metastasis cascade. Cancer Microenviron 3(1):137–147. https://doi.org/10.1007/s12307-010-0037-4
Wang Z, Han J, Cui Y, Zhou X, Fan K (2013) miRNA-21 inhibition enhances RANTES and IP-10 release in MCF-7 via PIAS3 and STAT3 signalling and causes increased lymphocyte migration. Biochem Biophys Res Commun 439(3):384–389. https://doi.org/10.1016/j.bbrc.2013.08.072
Jiang S, Zhang HW, Lu MH, He XH, Li Y, Gu H, Liu MF, Wang ED (2010) MicroRNA-155 functions as an OncomiR in breast cancer by targeting the suppressor of cytokine signaling 1 gene. Can Res 70(8):3119–3127. https://doi.org/10.1158/0008-5472.CAN-09-4250
Yang Q, Jiang W, Zhuang C, Geng Z, Hou C, Huang D, Hu L, Wang X (2015) MicroRNA-22 downregulation of galectin-9 influences lymphocyte apoptosis and tumor cell proliferation in liver cancer. Oncol Rep 34(4):1771–1778. https://doi.org/10.3892/or.2015.4167
Luo P, Wang X, Zhou J, Li L, Jing Z (2018) C-Cbl and Cbl-b expression in skull base chordomas is associated with tumor progression and poor prognosis. Hum Pathol 74:129–134. https://doi.org/10.1016/j.humpath.2017.12.019
Wang L, Wu Z, Tian K, Wang K, Li D, Ma J, Jia G, Zhang L, Zhang J (2017) Clinical features and surgical outcomes of patients with skull base chordoma: a retrospective analysis of 238 patients. J Neurosurg 127(6):1257–1267. https://doi.org/10.3171/2016.9.JNS16559
Fernandez-Miranda JC, Gardner PA, Snyderman CH, Devaney KO, Mendenhall WM, Suarez C, Rinaldo A, Ferlito A (2014) Clival chordomas: A pathological, surgical, and radiotherapeutic review. Head Neck 36(6):892–906. https://doi.org/10.1002/hed.23415
Jiang Y, Han S, Cheng W, Wang Z, Wu A (2017) NFAT1-regulated IL6 signalling contributes to aggressive phenotypes of glioma. Cell Commun Signal 15(1):54. https://doi.org/10.1186/s12964-017-0210-1
Zou MX, Peng AB, Lv GH, Wang XB, Li J, She XL, Jiang Y (2016) Expression of programmed death-1 ligand (PD-L1) in tumor-infiltrating lymphocytes is associated with favorable spinal chordoma prognosis. Am J Transl Res 8(7):3274–3287
Bellmunt J, Mullane SA, Werner L, Fay AP, Callea M, Leow JJ, Taplin ME, Choueiri TK, Hodi FS, Freeman GJ, Signoretti S (2015) Association of PD-L1 expression on tumor-infiltrating mononuclear cells and overall survival in patients with urothelial carcinoma. Ann Oncol 26(4):812–817. https://doi.org/10.1093/annonc/mdv009
Hutterer M, Knyazev P, Abate A, Reschke M, Maier H, Stefanova N, Knyazeva T, Barbieri V, Reindl M, Muigg A, Kostron H, Stockhammer G, Ullrich A (2008) Axl and growth arrest specific gene 6 are frequently overexpressed in human gliomas and predict poor prognosis in patients with glioblastoma multiforme. Clin Cancer Res 14(1):130–138. https://doi.org/10.1158/1078-0432.ccr-07-0862
Remmele W, Schicketanz KH (1993) Immunohistochemical determination of estrogen and progesterone receptor content in human breast cancer. Pathol Res Pract 189(8):862–866. https://doi.org/10.1016/s0344-0338(11)81095-2
Sato E, Olson SH, Ahn J, Bundy B, Nishikawa H, Qian F, Jungbluth AA, Frosina D, Gnjatic S, Ambrosone C, Kepner J, Odunsi T, Ritter G, Lele S, Chen YT, Ohtani H, Old LJ, Odunsi K (2005) Intraepithelial CD8+ tumor-infiltrating lymphocytes and a high CD8+/regulatory T cell ratio are associated with favorable prognosis in ovarian cancer. Proc Natl Acad Sci USA 102(51):18538–18543. https://doi.org/10.1073/pnas.0509182102
Betel D, Koppal A, Agius P, Sander C, Leslie C (2010) Comprehensive modeling of microRNA targets predicts functional non-conserved and non-canonical sites. Genome Biol 11(8):R90. https://doi.org/10.1186/gb-2010-11-8-r90
Wong N, Wang X (2015) miRDB: an online resource for microRNA target prediction and functional annotations. Nucleic Acids Res 43(Database issue):D146–D152. https://doi.org/10.1093/nar/gku1104
Lewis BP, Burge CB, Bartel DP (2005) Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 120(1):15–20. https://doi.org/10.1016/j.cell.2004.12.035
Budczies J, Klauschen F, Sinn BV, Gyorffy B, Schmitt WD, Darb-Esfahani S, Denkert C (2012) Cutoff Finder: a comprehensive and straightforward Web application enabling rapid biomarker cutoff optimization. PLoS One 7(12):e51862. https://doi.org/10.1371/journal.pone.0051862
Han G, Chen G, Shen B, Li Y (2013) Tim-3: an activation marker and activation limiter of innate immune cells. Front Immunol 4:449. https://doi.org/10.3389/fimmu.2013.00449
Ocana-Guzman R, Torre-Bouscoulet L, Sada-Ovalle I (2016) TIM-3 regulates distinct functions in macrophages. Front Immunol 7:229. https://doi.org/10.3389/fimmu.2016.00229
Imaizumi T, Kumagai M, Sasaki N, Kurotaki H, Mori F, Seki M, Nishi N, Fujimoto K, Tanji K, Shibata T, Tamo W, Matsumiya T, Yoshida H, Cui XF, Takanashi S, Hanada K, Okumura K, Yagihashi S, Wakabayashi K, Nakamura T, Hirashima M, Satoh K (2002) Interferon-gamma stimulates the expression of galectin-9 in cultured human endothelial cells. J Leukoc Biol 72(3):486–491
Yoshida H, Imaizumi T, Kumagai M, Kimura K, Satoh C, Hanada N, Fujimoto K, Nishi N, Tanji K, Matsumiya T, Mori F, Cui XF, Tamo W, Shibata T, Takanashi S, Okumura K, Nakamura T, Wakabayashi K, Hirashima M, Sato Y, Satoh K (2001) Interleukin-1beta stimulates galectin-9 expression in human astrocytes. Neuroreport 12(17):3755–3758
Sabatos CA, Chakravarti S, Cha E, Schubart A, Sanchez-Fueyo A, Zheng XX, Coyle AJ, Strom TB, Freeman GJ, Kuchroo VK (2003) Interaction of Tim-3 and Tim-3 ligand regulates T helper type 1 responses and induction of peripheral tolerance. Nat Immunol 4(11):1102–1110. https://doi.org/10.1038/ni988
Sanchez-Fueyo A, Tian J, Picarella D, Domenig C, Zheng XX, Sabatos CA, Manlongat N, Bender O, Kamradt T, Kuchroo VK, Gutierrez-Ramos JC, Coyle AJ, Strom TB (2003) Tim-3 inhibits T helper type 1-mediated auto- and alloimmune responses and promotes immunological tolerance. Nat Immunol 4(11):1093–1101. https://doi.org/10.1038/ni987
Cheng G, Li M, Wu J, Ji M, Fang C, Shi H, Zhu D, Chen L, Zhao J, Shi L, Xu B, Zheng X, Wu C, Jiang J (2015) Expression of Tim-3 in gastric cancer tissue and its relationship with prognosis. Int J Clin Exp Pathol 8(8):9452–9457
Li H, Wu K, Tao K, Chen L, Zheng Q, Lu X, Liu J, Shi L, Liu C, Wang G, Zou W (2012) Tim-3/galectin-9 signaling pathway mediates T-cell dysfunction and predicts poor prognosis in patients with hepatitis B virus-associated hepatocellular carcinoma. Hepatology 56(4):1342–1351. https://doi.org/10.1002/hep.25777
Duan Z, Shen J, Yang X, Yang P, Osaka E, Choy E, Cote G, Harmon D, Zhang Y, Nielsen GP, Spentzos D, Mankin H, Hornicek F (2014) Prognostic significance of miRNA-1 (miR-1) expression in patients with chordoma. J Orthop Res 32(5):695–701. https://doi.org/10.1002/jor.22589
Zhang H, Yang K, Ren T, Huang Y, Tang X, Guo W (2018) miR-16-5p inhibits chordoma cell proliferation, invasion and metastasis by targeting Smad3. Cell Death Dis 9(6):680. https://doi.org/10.1038/s41419-018-0738-z
Wei W, Zhang Q, Wang Z, Yan B, Feng Y, Li P (2016) miR-219-5p inhibits proliferation and clonogenicity in chordoma cells and is associated with tumor recurrence. Oncol Lett 12(6):4568–4576. https://doi.org/10.3892/ol.2016.5222
Zou MX, Guo KM, Lv GH, Huang W, Li J, Wang XB, Jiang Y, She XL (2018) Clinicopathologic implications of CD8(+)/FOXp3(+) ratio and miR-574-3p/PD-L1 axis in spinal chordoma patients. Cancer Immunol Immunother 67(2):209–224. https://doi.org/10.1007/s00262-017-2080-1
Zou MX, Huang W, Wang XB, Li J, Lv GH, Wang B, Deng YW (2015) Reduced expression of miRNA-1237-3p associated with poor survival of spinal chordoma patients. Eur Spine J 24(8):1738–1746. https://doi.org/10.1007/s00586-015-3927-9
Osaka E, Yang X, Shen JK, Yang P, Feng Y, Mankin HJ, Hornicek FJ, Duan Z (2014) MicroRNA-1 (miR-1) inhibits chordoma cell migration and invasion by targeting slug. J Orthop Res 32(8):1075–1082. https://doi.org/10.1002/jor.22632
Liu J, Zhang J, Li Y, Wang L, Sui B, Dai D (2016) MiR-455-5p acts as a novel tumor suppressor in gastric cancer by down-regulating RAB18. Gene 592(2):308–315. https://doi.org/10.1016/j.gene.2016.07.034
Yang Q, Hou C, Huang D, Zhuang C, Jiang W, Geng Z, Wang X, Hu L (2017) miR-455-5p functions as a potential oncogene by targeting galectin-9 in colon cancer. Oncol Lett 13(3):1958–1964. https://doi.org/10.3892/ol.2017.5608
Chen H, Garbutt CC, Spentzos D, Choy E, Hornicek FJ, Duan Z (2017) Expression and therapeutic potential of SOX9 in chordoma. Clin Cancer Res 23(17):5176–5186. https://doi.org/10.1158/1078-0432.CCR-17-0177
Fritzsching B, Fellenberg J, Moskovszky L, Sapi Z, Krenacs T, Machado I, Poeschl J, Lehner B, Szendroi M, Bosch AL, Bernd L, Csoka M, Mechtersheimer G, Ewerbeck V, Kinscherf R, Kunz P (2015) CD8(+)/FOXP3(+)-ratio in osteosarcoma microenvironment separates survivors from non-survivors: a multicenter validated retrospective study. Oncoimmunology 4(3):e990800. https://doi.org/10.4161/2162402X.2014.990800
Funding
This work was supported by the National Natural Science Foundation of China (Nos. 81101917, 81270036, 30901736), the Liaoning Province Natural Science Foundation (No. 20170541022), the Plan to Focus on Research and Development from Science and Technology project of Liaoning Province (No. 2017225029), the Science and Technology Plan Project of Shenyang City (No. 18-014-4-11), and the Fund for Scientific Research of The First Hospital of China Medical University (No. FHCMU-FSR).
Author information
Authors and Affiliations
Contributions
ZJ and YZ conceived and designed the study; JZ and YJ performed the experiments and collected the data; HZ, JZ, LC, PL, LL, JZ, and YJ produced the figures and tables; all authors performed the analysis and analyzed the data. JZ, YJ, YZ, and ZJ interpreted results and wrote the manuscript. FL, DZ, and HZ modified the manuscript. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Ethical approval
The study was approved by the Research Ethics Committee of the First Hospital of China Medical University and was in accordance with the ethical standards of the institutional committees and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study approval number is AF-SOP-07-01.
Informed consent
Informed consent was obtained from all individual participants included in the study. With approval from the National Science Foundation of China (81101917), we designed informed consent forms that were signed by eligible patients before recruitment into the study and admission to the hospital. The patients agreed to the use of their specimens and clinical data for research purposes only.
Cell line authentication
The human chordoma cell line UM-Chor1 was obtained as a gift from Professor Yazhuo Zhang, Department of Neurosurgery, Beijing Tiantan Hospital. The origin of UM-Chor1 was human clivus chordoma tissue. The American Type Culture Collection (ATCC) performed authentication of cell line UM-Chor1 via cytochrome C oxidase I assay and short tandem repeat analysis. We obtained a cell line authentication certificate from the ATCC.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhou, J., Jiang, Y., Zhang, H. et al. Clinicopathological implications of TIM3+ tumor-infiltrating lymphocytes and the miR-455-5p/Galectin-9 axis in skull base chordoma patients. Cancer Immunol Immunother 68, 1157–1169 (2019). https://doi.org/10.1007/s00262-019-02349-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-019-02349-1