Abstract
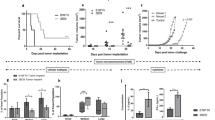
Regulatory T cells (Treg) and myeloid-derived suppressor cells (MDSC) are the two important and interactive immunosuppressive components of the tumor microenvironment that hamper anti-tumor immune responses. Therefore, targeting these two populations together might be beneficial for overcoming immune suppression in the tumor microenvironment. We have recently shown that prophylactic Foxp3 DNA/recombinant protein vaccine (Foxp3 vaccine) promotes immunity against Treg in tumor-free conditions. In the present study, we investigated the immune modulatory effects of a prophylactic regimen of the redesigned Foxp3 vaccine in the B16F10 melanoma model. Our results indicate that Foxp3 vaccination continuously reduces Treg population in both the tumor site and the spleen. Surprisingly, Treg reduction was associated with a significant decrease in the frequency of MDSC, both in the spleen and in the tumor environment. Furthermore, Foxp3 vaccination resulted in a significant reduction of arginase-1(Arg-1)-induced nitric oxide synthase (iNOS), reactive oxygen species (ROS) and suppressed MDSC activity. Moreover, this concurrent depletion restored production of inflammatory cytokine IFN-γ and enhanced tumor-specific CTL response, which subsequently resulted in the reduction of tumor growth and the improved survival rate of vaccinated mice. In conclusion, our results revealed that Foxp3 vaccine promotes an immune response against tumor by targeting both Treg and MDSC, which could be exploited as a potential immunotherapy approach.






Similar content being viewed by others
Abbreviations
- Arg-1:
-
Arginase-1
- BCA:
-
Bicinchoninic acid
- CFSE:
-
Carboxy fluorescein succinimidyl ester
- DCFDA:
-
2′,7′-Dichlorodihydrofluorescein diacetate
- E. coli :
-
Escherichia coli
- Gr-1:
-
Granulocyte differentiation marker 1
- His-tag:
-
(Poly)-histidine-tagged
- IFA:
-
Incomplete Freund’s adjuvant
- iNOS:
-
Induced nitric oxide synthase
- MDSC:
-
Myeloid-derived suppressor cell(s)
- Ni–NTA:
-
Nickel nitrilotriacetic acid
- ROS:
-
Reactive oxygen species
- Treg:
-
Regulatory T cell(s)
References
Sakaguchi S, Sakaguchi N, Asano M, Itoh M, Toda M (1995) Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J Immunol 155(3):1151–1164
Fontenot JD, Gavin MA, Rudensky AY (2003) Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat Immunol 4(4):330–336. https://doi.org/10.1038/ni904
Sakaguchi S (2005) Naturally arising Foxp3-expressing CD25+CD4+ regulatory T cells in immunological tolerance to self and non-self. Nat Immunol 6(4):345–352. https://doi.org/10.1038/ni1178
Curiel TJ, Coukos G, Zou L, Alvarez X, Cheng P, Mottram P et al (2004) Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med 10(9):942–949. https://doi.org/10.1038/nm1093
Wolf AM, Wolf D, Steurer M, Gastl G, Gunsilius E, Grubeck-Loebenstein B (2003) Increase of regulatory T cells in the peripheral blood of cancer patients. Clin Cancer Res 9(2):606–612
Viehl CT, Moore TT, Liyanage UK, Frey DM, Ehlers JP, Eberlein TJ et al (2006) Depletion of CD4+CD25+ regulatory T cells promotes a tumor-specific immune response in pancreas cancer-bearing mice. Ann Surg Oncol 13(9):1252–1258. https://doi.org/10.1245/s10434-006-9015-y
Gabrilovich DI, Nagaraj S (2009) Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol 9(3):162–174. https://doi.org/10.1038/nri2506
Gabrilovich DI, Ostrand-Rosenberg S, Bronte V (2012) Coordinated regulation of myeloid cells by tumours. Nat Rev Immunol 12(4):253–268. https://doi.org/10.1038/nri3175
Lindau D, Gielen P, Kroesen M, Wesseling P, Adema GJ (2013) The immunosuppressive tumour network: myeloid-derived suppressor cells, regulatory T cells and natural killer T cells. Immunology 138(2):105–115. https://doi.org/10.1111/imm.12036
Huang B, Pan PY, Li Q, Sato AI, Levy DE, Bromberg J et al (2006) Gr-1+CD115+ immature myeloid suppressor cells mediate the development of tumor-induced T regulatory cells and T-cell anergy in tumor-bearing host. Cancer Res 66(2):1123–1131. https://doi.org/10.1158/0008-5472.CAN-05-1299
Fujimura T, Ring S, Umansky V, Mahnke K, Enk AH (2012) Regulatory T cells stimulate B7-H1 expression in myeloid-derived suppressor cells in ret melanomas. J Investig Dermatol 132(4):1239–1246. https://doi.org/10.1038/jid.2011.416
Lutsiak ME, Semnani RT, De Pascalis R, Kashmiri SV, Schlom J, Sabzevari H (2005) Inhibition of CD4(+)25+ T regulatory cell function implicated in enhanced immune response by low-dose cyclophosphamide. Blood 105(7):2862–2868. https://doi.org/10.1182/blood-2004-06-2410
Onizuka S, Tawara I, Shimizu J, Sakaguchi S, Fujita T, Nakayama E (1999) Tumor rejection by in vivo administration of anti-CD25 (interleukin-2 receptor alpha) monoclonal antibody. Cancer Res 59(13):3128–3133
Foss FM (2000) DAB(389)IL-2 (ONTAK): a novel fusion toxin therapy for lymphoma. Clin Lymphoma 1(2):110–116 (discussion 7)
Zhang H, Chua KS, Guimond M, Kapoor V, Brown MV, Fleisher TA et al (2005) Lymphopenia and interleukin-2 therapy alter homeostasis of CD4+CD25+ regulatory T cells. Nat Med 11(11):1238–1243. https://doi.org/10.1038/nm1312
Nair S, Boczkowski D, Fassnacht M, Pisetsky D, Gilboa E (2007) Vaccination against the forkhead family transcription factor Foxp3 enhances tumor immunity. Cancer Res 67(1):371–380. https://doi.org/10.1158/0008-5472.CAN-06-2903
Stevenson FK (1999) DNA vaccines against cancer: from genes to therapy. Ann Oncol 10(12):1413–1418
Stevenson FK, Ottensmeier CH, Rice J (2010) DNA vaccines against cancer come of age. Curr Opin Immunol 22(2):264–270. https://doi.org/10.1016/j.coi.2010.01.019
Kyutoku M, Nakagami H, Koriyama H, Tomioka H, Nakagami F, Shimamura M et al (2013) Development of novel DNA vaccine for VEGF in murine cancer model. Sci Rep 3:3380. https://doi.org/10.1038/srep03380
Liu S, Shi D, Wang HC, Yu YZ, Xu Q, Sun ZW (2015) Co-immunization with DNA and protein mixture: a safe and efficacious immunotherapeutic strategy for Alzheimer’s disease in PDAPP mice. Sci Rep 5:7771. https://doi.org/10.1038/srep07771
Mousavi Niri N, Memarnejadian A, Pilehvar-Soltanahmadi Y, Agha Sadeghi M, Mahdavi M, Kheshtchin N et al (2016) Improved anti-Treg vaccination targeting Foxp3 efficiently decreases regulatory T cells in mice. J Immunother 39(7):269–275. https://doi.org/10.1097/CJI.0000000000000133
Niri NM, Hadjati J, Sadat M, Memarnejadian A, Aghasadeghi M, Akbarzadeh A et al (2015) Inducing humoral immune responses against regulatory T cells by Foxp3-Fc(IgG) fusion protein. Monoclon Antib Immunodiagn Immunother 34(6):381–385. https://doi.org/10.1089/mab.2015.0048
Namdar A, Mirzaei HR, Jadidi-Niaragh F, Ashourpour M, Ajami M, Hadjati J et al (2015) Multiple low doses of 5-fluorouracil diminishes immunosuppression by myeloid derived suppressor cells in murine melanoma model. Iran J Immunol 12(3):176–187
Liu Y, Van Ginderachter JA, Brys L, De Baetselier P, Raes G, Geldhof AB (2003) Nitric oxide-independent CTL suppression during tumor progression: association with arginase-producing (M2) myeloid cells. J Immunol 170(10):5064–5074
Zou W (2005) Immunosuppressive networks in the tumour environment and their therapeutic relevance. Nat Rev Cancer 5(4):263–274. https://doi.org/10.1038/nrc1586
Sutmuller RP, van Duivenvoorde LM, van Elsas A, Schumacher TN, Wildenberg ME, Allison JP et al (2001) Synergism of cytotoxic T lymphocyte-associated antigen 4 blockade and depletion of CD25(+) regulatory T cells in antitumor therapy reveals alternative pathways for suppression of autoreactive cytotoxic T lymphocyte responses. J Exp Med 194(6):823–832
Coe D, Begom S, Addey C, White M, Dyson J, Chai JG (2010) Depletion of regulatory T cells by anti-GITR mAb as a novel mechanism for cancer immunotherapy. Cancer Immunol Immunother 59(9):1367–1377. https://doi.org/10.1007/s00262-010-0866-5
Hori S, Sakaguchi S (2004) Foxp3: a critical regulator of the development and function of regulatory T cells. Microbes Infect 6(8):745–751. https://doi.org/10.1016/j.micinf.2004.02.020
Andersen MH (2013) FOXP3-specific immunity. Oncoimmunology 2(10):e26247. https://doi.org/10.4161/onci.26247
Ebert LM, Tan BS, Browning J, Svobodova S, Russell SE, Kirkpatrick N et al (2008) The regulatory T cell-associated transcription factor FoxP3 is expressed by tumor cells. Cancer Res 68(8):3001–3009. https://doi.org/10.1158/0008-5472.CAN-07-5664
Ahmadzadeh M, Felipe-Silva A, Heemskerk B, Powell DJ Jr, Wunderlich JR, Merino MJ et al (2008) FOXP3 expression accurately defines the population of intratumoral regulatory T cells that selectively accumulate in metastatic melanoma lesions. Blood 112(13):4953–4960. https://doi.org/10.1182/blood-2008-06-163048
Marabelle A, Kohrt H, Sagiv-Barfi I, Ajami B, Axtell RC, Zhou G et al (2013) Depleting tumor-specific Tregs at a single site eradicates disseminated tumors. J Clin Investig 123(6):2447–2463. https://doi.org/10.1172/JCI64859
Klages K, Mayer CT, Lahl K, Loddenkemper C, Teng MW, Ngiow SF et al (2010) Selective depletion of Foxp3+ regulatory T cells improves effective therapeutic vaccination against established melanoma. Cancer Res 70(20):7788–7799. https://doi.org/10.1158/0008-5472.CAN-10-1736
Larsen SK, Munir S, Woetmann A, Frosig TM, Odum N, Svane IM et al (2013) Functional characterization of Foxp3-specific spontaneous immune responses. Leukemia 27(12):2332–2340. https://doi.org/10.1038/leu.2013.196
Franco-Molina MA, Miranda-Hernandez DF, Mendoza-Gamboa E, Zapata-Benavides P, Coronado-Cerda EE, Sierra-Rivera CA et al (2016) Silencing of Foxp3 delays the growth of murine melanomas and modifies the tumor immunosuppressive environment. Onco Targets Ther 9:243–253. https://doi.org/10.2147/OTT.S90476
Miguel A, Sendra L, Noe V, Ciudad CJ, Dasi F, Hervas D et al (2017) Silencing of Foxp3 enhances the antitumor efficacy of GM-CSF genetically modified tumor cell vaccine against B16 melanoma. Onco Targets Ther 10:503–514. https://doi.org/10.2147/OTT.S104393
Serafini P, Mgebroff S, Noonan K, Borrello I (2008) Myeloid-derived suppressor cells promote cross-tolerance in B-cell lymphoma by expanding regulatory T cells. Cancer Res 68(13):5439–5449. https://doi.org/10.1158/0008-5472.CAN-07-6621
Dolcetti L, Marigo I, Mantelli B, Peranzoni E, Zanovello P, Bronte V (2008) Myeloid-derived suppressor cell role in tumor-related inflammation. Cancer Lett 267(2):216–225. https://doi.org/10.1016/j.canlet.2008.03.012
Ostrand-Rosenberg S, Sinha P (2009) Myeloid-derived suppressor cells: linking inflammation and cancer. J Immunol 182(8):4499–4506. https://doi.org/10.4049/jimmunol.0802740
Ostrand-Rosenberg S (2010) Myeloid-derived suppressor cells: more mechanisms for inhibiting antitumor immunity. Cancer Immunol Immunother 59(10):1593–1600. https://doi.org/10.1007/s00262-010-0855-8
Ko HJ, Lee JM, Kim YJ, Kim YS, Lee KA, Kang CY (2009) Immunosuppressive myeloid-derived suppressor cells can be converted into immunogenic APCs with the help of activated NKT cells: an alternative cell-based antitumor vaccine. J Immunol 182(4):1818–1828. https://doi.org/10.4049/jimmunol.0802430
Zou W (2006) Regulatory T cells, tumour immunity and immunotherapy. Nat Rev Immunol 6(4):295–307. https://doi.org/10.1038/nri1806
Betts MR, Koup RA (2004) Detection of T-cell degranulation: CD107a and b. Methods Cell Biol 75:497–512
MacKie RM, Hauschild A, Eggermont AM (2009) Epidemiology of invasive cutaneous melanoma. Ann Oncol. 20(Suppl 6):vi1–vi7. https://doi.org/10.1093/annonc/mdp252
Garbe C, Peris K, Hauschild A, Saiag P, Middleton M, Spatz A et al (2010) Diagnosis and treatment of melanoma: European consensus-based interdisciplinary guideline. Eur J Cancer 46(2):270–283. https://doi.org/10.1016/j.ejca.2009.10.032
Author information
Authors and Affiliations
Contributions
AN performed plasmid and protein preparation, animal studies, flow cytometry analysis, real-time PCR, suppression assay, and nitrite assay, analyzed the data, and wrote the manuscript. RM contributed in flow cytometry analysis and animal studies. AM advised in plasmid design. RB performed intracellular cytokine staining. MS assisted in plasmid and protein preparation. HRM performed Treg staining and CD107a cytotoxic assay. HF performed RNA extraction. MZ performed MDSC staining. KA provided the main construct and co-supervised the designing of plasmids. SE contributed to writing and editing the article. FN advised on plasmid design and data analysis. AR co-supervised the study. JH supervised the study.
Corresponding author
Ethics declarations
Funding
The main part of this study was supported by the Iran National Science Foundation (INSF, Grant Number 90007957) and partially by the Isfahan University of Medical Sciences (Grant Number 391146).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Mouse experiments were approved by the animal ethics committee of Tehran University of Medical Sciences (Tehran, Iran).
Rights and permissions
About this article
Cite this article
Namdar, A., Mirzaei, R., Memarnejadian, A. et al. Prophylactic DNA vaccine targeting Foxp3+ regulatory T cells depletes myeloid-derived suppressor cells and improves anti-melanoma immune responses in a murine model. Cancer Immunol Immunother 67, 367–379 (2018). https://doi.org/10.1007/s00262-017-2088-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-017-2088-6