Abstract
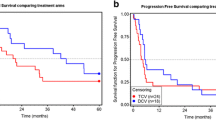
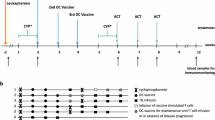
Dendritic cells (DC) are the most potent antigen presenting cells and have proven effective in stimulation of specific immune responses in vivo. Competing immune inhibition could limit the clinical efficacy of DC vaccination. In this phase II trial, metronomic Cyclophosphamide and a Cox-2 inhibitor have been added to a DC vaccine with the intend to dampen immunosuppressive mechanisms. Twenty-eight patients with progressive metastatic melanoma were treated with autologous DCs pulsed with survivin, hTERT, and p53-derived peptides (HLA-A2+) or tumor lysate (HLA-A2−). Concomitantly the patients were treated with IL-2, Cyclophosphamide, and Celecoxib. The treatment was safe and tolerable. Sixteen patients (57 %) achieved stable disease (SD) at 1st evaluation and 8 patients had prolonged SD (7–13.7 months). The median OS was 9.4 months. Patients with SD had an OS of 10.5 months while patients with progressive disease (PD) had an OS of 6.0 months (p = 0.048) even though there were no differences in prognostic factors between the two groups. Despite the use of metronomic Cyclophosphamide, regulatory T cells did not decrease during treatment. Indirect IFN-γ ELISPOT assays showed a general increase in immune responses from baseline to the time of 4th vaccination. Induction of antigen-specific immune responses was seen in 9 out of 15 screened HLA-A2+ patients. In conclusion, the number of patients obtaining SD more than doubled and 6-month survival significantly increased compared to a previous trial without Cyclophosphamide and Celecoxib. A general increase in immune responses against the tested peptides was observed.





Similar content being viewed by others
References
Lee ML, Tomsu K, Von Eschen KB (2000) Duration of survival for disseminated malignant melanoma: results of a meta-analysis. Melanoma Res 10:81–92
Agarwala SS (2009) Current systemic therapy for metastatic melanoma. Expert Rev Anticancer Ther 9:587–595
Quirt I, Verma S, Petrella T, Bak K, Charette M (2007) Temozolomide for the treatment of metastatic melanoma: a systematic review. Oncologist 12:1114–1123
Anderson CM, Buzaid AC, Legha SS (1995) Systemic treatments for advanced cutaneous melanoma. Oncology (Williston Park) 9:1149–1158
Dutcher J (2002) Current status of interleukin-2 therapy for metastatic renal cell carcinoma and metastatic melanoma. Oncology (Williston Park) 16:4–10
Gajewski TF (2010) Improved melanoma survival at last! Ipilimumab and a paradigm shift for immunotherapy. Pigment Cell Melanoma Res 23:580–581
Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB et al (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363:711–723
Gilboa E (2007) DC-based cancer vaccines. J Clin Invest 117:1195–1203
Osada T, Clay TM, Woo CY, Morse MA, Lyerly HK (2006) Dendritic cell-based immunotherapy. Int Rev Immunol 25:377–413
Zou W (2006) Regulatory T cells, tumour immunity and immunotherapy. Nat Rev Immunol 6:295–307
Gabrilovich DI, Nagaraj S (2009) Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol 9:162–174
Ahmadzadeh M, Rosenberg SA (2006) IL-2 administration increases CD4+ CD25(hi) Foxp3+ regulatory T cells in cancer patients. Blood 107:2409–2414
Berntsen A, Trepiakas R, Wenandy L, Geertsen PF, Thor SP, Andersen MH et al (2008) Therapeutic dendritic cell vaccination of patients with metastatic renal cell carcinoma: a clinical phase 1/2 trial. J Immunother 31:771–780
Trepiakas R, Berntsen A, Hadrup SR, Bjorn J, Geertsen PF, Straten PT et al (2010) Vaccination with autologous dendritic cells pulsed with multiple tumor antigens for treatment of patients with malignant melanoma: results from a phase I/II trial. Cytotherapy 12:721–734
Curiel TJ (2008) Regulatory T cells and treatment of cancer. Curr Opin Immunol 20:241–246
Jacobs JF, Punt CJ, Lesterhuis WJ, Sutmuller RP, Brouwer HM, Scharenborg NM et al (2010) Dendritic cell vaccination in combination with anti-CD25 monoclonal antibody treatment: a phase I/II study in metastatic melanoma patients. Clin Cancer Res 16:5067–5078
Nishikawa H, Sakaguchi S (2010) Regulatory T cells in tumor immunity. Int J Cancer 127:759–767
Ghiringhelli F, Menard C, Puig PE, Ladoire S, Roux S, Martin F et al (2007) Metronomic cyclophosphamide regimen selectively depletes CD4+ CD25+ regulatory T cells and restores T and NK effector functions in end stage cancer patients. Cancer Immunol Immunother 56:641–648
Liu JY, Wu Y, Zhang XS, Yang JL, Li HL, Mao YQ et al (2007) Single administration of low dose cyclophosphamide augments the antitumor effect of dendritic cell vaccine. Cancer Immunol Immunother 56:1597–1604
Lutsiak ME, Semnani RT, De PR, Kashmiri SV, Schlom J, Sabzevari H (2005) Inhibition of CD4(+)25+ T regulatory cell function implicated in enhanced immune response by low-dose cyclophosphamide. Blood 105:2862–2868
Denkert C, Kobel M, Berger S, Siegert A, Leclere A, Trefzer U et al (2001) Expression of cyclooxygenase 2 in human malignant melanoma. Cancer Res 61:303–308
Goulet AC, Einsphar JG, Alberts DS, Beas A, Burk C, Bhattacharyya A et al (2003) Analysis of cyclooxygenase 2 (COX-2) expression during malignant melanoma progression. Cancer Biol Ther 2:713–718
Becker MR, Siegelin MD, Rompel R, Enk AH, Gaiser T (2009) COX-2 expression in malignant melanoma: a novel prognostic marker? Melanoma Res 19:8–16
Bergmann C, Strauss L, Zeidler R, Lang S, Whiteside TL (2007) Expansion of human T regulatory type 1 cells in the microenvironment of cyclooxygenase 2 overexpressing head and neck squamous cell carcinoma. Cancer Res 67:8865–8873
Mahic M, Yaqub S, Johansson CC, Tasken K, Aandahl EM (2006) FOXP3+ CD4+ CD25+ adaptive regulatory T cells express cyclooxygenase-2 and suppress effector T cells by a prostaglandin E2-dependent mechanism. J Immunol 177:246–254
Sharma S, Yang SC, Zhu L, Reckamp K, Gardner B, Baratelli F et al (2005) Tumor cyclooxygenase-2/prostaglandin E2-dependent promotion of FOXP3 expression and CD4+ CD25+ T regulatory cell activities in lung cancer. Cancer Res 65:5211–5220
Sinha P, Clements VK, Fulton AM, Ostrand-Rosenberg S (2007) Prostaglandin E2 promotes tumor progression by inducing myeloid-derived suppressor cells. Cancer Res 67:4507–4513
Toebes M, Coccoris M, Bins A, Rodenko B, Gomez R, Nieuwkoop NJ et al (2006) Design and use of conditional MHC class I ligands. Nat Med 12:246–251
Hadrup SR, Bakker AH, Shu CJ, Andersen RS, van VJ, Hombrink P et al (2009) Parallel detection of antigen-specific T-cell responses by multidimensional encoding of MHC multimers. Nat Methods 6:520–526
Andersen MH, Pedersen LO, Becker JC, Straten PT (2001) Identification of a cytotoxic T lymphocyte response to the apoptosis inhibitor protein survivin in cancer patients. Cancer Res 61:869–872
McCutcheon M, Wehner N, Wensky A, Kushner M, Doan S, Hsiao L et al (1997) A sensitive ELISPOT assay to detect low-frequency human T lymphocytes. J Immunol Methods 210:149–166
Poschke I, Mougiakakos D, Hansson J, Masucci GV, Kiessling R (2010) Immature immunosuppressive CD14 + HLA-DR-/low cells in melanoma patients are Stat3hi and overexpress CD80, CD83, and DC-sign. Cancer Res 70:4335–4345
Figdor CG, de Vries IJ, Lesterhuis WJ, Melief CJ (2004) Dendritic cell immunotherapy: mapping the way. Nat Med 10:475–480
Manola J, Atkins M, Ibrahim J, Kirkwood J (2000) Prognostic factors in metastatic melanoma: a pooled analysis of Eastern cooperative oncology group trials. J Clin Oncol 18:3782–3793
Andersen RS, Thrue CA, Junker N, Lyngaa R, Donia M, Ellebaek E et al (2012) Dissection of T cell antigen specificity in human melanoma. Cancer Res. doi:10.1158/0008-5472.CAN-11-2614
Svane IM, Pedersen AE, Johnsen HE, Nielsen D, Kamby C, Gaarsdal E et al (2004) Vaccination with p53-peptide-pulsed dendritic cells, of patients with advanced breast cancer: report from a phase I study. Cancer Immunol Immunother 53:633–641
Schrama D, Pedersen LO, Keikavoussi P, Andersen MH, Straten Pt, Brocker EB et al (2002) Aggregation of antigen-specific T cells at the inoculation site of mature dendritic cells. J Invest Dermatol 119:1443–1448
Lopez MN, Pereda C, Segal G, Munoz L, Aguilera R, Gonzalez FE et al (2009) Prolonged survival of dendritic cell-vaccinated melanoma patients correlates with tumor-specific delayed type IV hypersensitivity response and reduction of tumor growth factor beta-expressing T cells. J Clin Oncol 27:945–952
Ridolfi L, Petrini M, Fiammenghi L, Granato AM, Ancarani V, Pancisi E et al (2010) Unexpected high response rate to traditional therapy after dendritic cell-based vaccine in advanced melanoma: update of clinical outcome and subgroup analysis. Clin Dev Immunol 2010:504979
Ridolfi L, Petrini M, Fiammenghi L, Granato AM, Ancarani V, Pancisi E et al (2011) Dendritic cell-based vaccine in advanced melanoma: update of clinical outcome. Melanoma Res 21:524–529
Engell-Noerregaard L, Hansen TH, Andersen MH, Thor SP, Svane IM (2009) Review of clinical studies on dendritic cell-based vaccination of patients with malignant melanoma: assessment of correlation between clinical response and vaccine parameters. Cancer Immunol Immunother 58:1–14
Hersey P, Halliday G, Farrelly M, DeSilva C, Lett M, Menzies S (2008) Phase I/II study of treatment with matured dendritic cells with or without low dose IL-2 in patients with disseminated melanoma. Cancer Immunol Immunother 57:1039–1051
Andersen MH, Gehl J, Reker S, Pedersen LO, Becker JC, Geertsen P et al (2003) Dynamic changes of specific T cell responses to melanoma correlate with IL-2 administration. Semin Cancer Biol 13:449–459
Iniguez MA, Punzon C, Fresno M (1999) Induction of cyclooxygenase-2 on activated T lymphocytes: regulation of T cell activation by cyclooxygenase-2 inhibitors. J Immunol 163:111–119
Paccani SR, Boncristiano M, Ulivieri C, D’Elios MM, Del PG, Baldari CT (2002) Nonsteroidal anti-inflammatory drugs suppress T-cell activation by inhibiting p38 MAPK induction. J Biol Chem 277:1509–1513
Pettersen FO, Torheim EA, Dahm AE, Aaberge IS, Lind A, Holm M et al (2011) An exploratory trial of cyclooxygenase type 2 inhibitor in HIV-1 infection: downregulated immune activation and improved T cell-dependent vaccine responses. J Virol 85:6557–6566
Kleinewietfeld M, Starke M, Di MD, Borsellino G, Battistini L, Rotzschke O et al (2009) CD49d provides access to “untouched” human Foxp3 + Treg free of contaminating effector cells. Blood 113:827–836
Solito S, Falisi E, Diaz-Montero CM, Doni A, Pinton L, Rosato A et al (2011) A human promyelocytic-like population is responsible for the immune suppression mediated by myeloid-derived suppressor cells. Blood 118:2254–2265
Diaz-Montero CM, Salem ML, Nishimura MI, Garrett-Mayer E, Cole DJ, Montero AJ (2009) Increased circulating myeloid-derived suppressor cells correlate with clinical cancer stage, metastatic tumor burden, and doxorubicin-cyclophosphamide chemotherapy. Cancer Immunol Immunother 58:49–59
Emens LA (2010) Chemoimmunotherapy. Cancer J 16:295–303
Finkelstein SE, Carey T, Fricke I, Yu D, Goetz D, Gratz M et al (2010) Changes in dendritic cell phenotype after a new high-dose weekly schedule of interleukin-2 therapy for kidney cancer and melanoma. J Immunother 33:817–827
Mirza N, Fishman M, Fricke I, Dunn M, Neuger AM, Frost TJ et al (2006) All-trans-retinoic acid improves differentiation of myeloid cells and immune response in cancer patients. Cancer Res 66:9299–9307
de Vries IJ, Lesterhuis WJ, Scharenborg NM, Engelen LP, Ruiter DJ, Gerritsen MJ et al (2003) Maturation of dendritic cells is a prerequisite for inducing immune responses in advanced melanoma patients. Clin Cancer Res 9:5091–5100
Wadler S, Einzig AI, Dutcher JP, Ciobanu N, Landau L, Wiernik PH (1988) Phase II trial of recombinant alpha-2b-interferon and low-dose cyclophosphamide in advanced melanoma and renal cell carcinoma. Am J Clin Oncol 11:55–59
Lindemann A, Hoffken K, Schmidt RE, Diehl V, Kloke O, Gamm H et al (1989) A phase-II study of low-dose cyclophosphamide and recombinant human interleukin-2 in metastatic renal cell carcinoma and malignant melanoma. Cancer Immunol Immunother 28:275–281
Eggermont AM, Kirkwood JM (2004) Re-evaluating the role of dacarbazine in metastatic melanoma: what have we learned in 30 years? Eur J Cancer 40:1825–1836
Jilaveanu LB, Aziz SA, Kluger HM (2009) Chemotherapy and biologic therapies for melanoma: do they work? Clin Dermatol 27:614–625
Powell DJ Jr, de Vries CR, Allen T, Ahmadzadeh M, Rosenberg SA (2007) Inability to mediate prolonged reduction of regulatory T Cells after transfer of autologous CD25-depleted PBMC and interleukin-2 after lymphodepleting chemotherapy. J Immunother 30:438–447
Gasparini G (2001) Metronomic scheduling: the future of chemotherapy? Lancet Oncol 2:733–740
Shaked Y, Emmenegger U, Man S, Cervi D, Bertolini F, Ben-David Y et al (2005) Optimal biologic dose of metronomic chemotherapy regimens is associated with maximum antiangiogenic activity. Blood 106:3058–3061
Browder T, Butterfield CE, Kräling BM, Shi B, Marshall B, O’Reilly MS et al (2000) Antiangiogenic scheduling of chemotherapy improves efficacy against experimental drug-resistant cancer. Cancer Res 60:1878–1886
Acknowledgments
This work was supported by grants from Aase and Einer Danielsens foundation. We thank Kirsten Nicholaisen, Eva Gaardsdal, Charlotte Vajhøj, and Tina Seremet for excellent technical assistance in the laboratory, the study nurses Dorte Carlsen and Susanne Wehmeyer for helping with practical and monitoring issues, and Tobias W. Clausen for performing statistical analyses.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ellebaek, E., Engell-Noerregaard, L., Iversen, T.Z. et al. Metastatic melanoma patients treated with dendritic cell vaccination, Interleukin-2 and metronomic cyclophosphamide: results from a phase II trial. Cancer Immunol Immunother 61, 1791–1804 (2012). https://doi.org/10.1007/s00262-012-1242-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-012-1242-4