Abstract
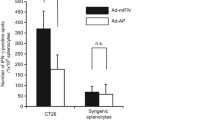
One of the major challenges in the treatment of solid cancers by allogenic hematopoietic stem cell transfer (alloHSCT) is the specific enhancement of antitumor immunity. Interferon (IFN) is a cytokine with pleiotropic biological functions including an immunomoduration, and our preclinical studies have shown that an intratumoral IFN-α gene transfer induced strong local tumor control and systemic tumor-specific immunity. In the present study, we examined whether the IFN-α gene transfer could enhance recognition of tumor-associated antigens by donor T cells and augment the antitumor activity of alloHSCT. First, when a mouse IFN-α adenovirus vector (Ad-mIFN) was injected into subcutaneous xenografts of syngeneic renal and colon cancer cells, tumor growth was significantly suppressed in a dose-dependent manner. A significant tumor cell death and infiltration of immune cells was recognized in the Ad-mIFN-injected tumors, and the dendrtic cells isolated from the tumors showed a strong Th1-oriented response. The antitumor effect of Ad-mIFN was then examined in a murine model of minor histocompatibility antigen-mismatched alloHSCT. The intratumoral IFN-α gene transfer caused significant tumor suppression in the alloHSCT recipients, and this suppression was evident not only in the gene-transduced tumors but also in simultaneously inoculated distant tumors which did not receive the vector injection. A cytotoxicity assay showed specific tumor cell lysis by donor T cells responding to IFN-α. Graft-versus-host disease was not exacerbated serologically or clinically in the mice treated with IFN-α. This combination strategy deserves evaluation in future clinical trials for human solid cancers.









Similar content being viewed by others
References
Sandmaier BM, Mackinnon S, Childs RW (2007) Reduced intensity conditioning for allogeneic hematopoietic cell transplantation: current perspectives. Biol Blood Marrow Transplant 13:87–97
Welniak LA, Blazar BR, Murphy WJ (2007) Immunobiology of allogeneic hematopoietic stem cell transplantation. Annu Rev Immunol 25:139–170
Akatsuka Y, Morishima Y, Kuzushima K, Kodera Y, Takahashi T (2007) Minor histocompatibility antigens as targets for immunotherapy using allogeneic immune reactions. Cancer Sci 98:1139–1146
Gutterman JU (1994) Cytokine therapeutics: lessons from interferon alpha. Proc Natl Acad Sci USA 91:1198–1205
Belardelli F, Ferrantini M, Proietti E, Kirkwood JM (2002) Interferon-alpha in tumor immunity and immunotherapy. Cytokine Growth Factor Rev 13:119–134
Einhorn S, Grander D (1996) Why do so many cancer patients fail to respond to interferon therapy? J Interferon Cytokine Res 16:275–281
Salmon P, Le Cotonnec JY, Galazka A, Abdul-Ahad A, Darragh A (1996) Pharmacokinetics and pharmacodynamics of recombinant human interferon-beta in healthy male volunteers. J Interferon Cytokine Res 16:759–764
Suzuki K, Aoki K, Ohnami S et al (2003) Adenovirus-mediated gene transfer of interferon alpha improves dimethylnitrosamine-induced liver cirrhosis in rat model. Gene Ther 10:765–773
Ohashi M, Yoshida K, Kushida M et al (2005) Adenovirus-mediated interferon alpha gene transfer induces regional direct cytotoxicity and possible systemic immunity against pancreatic cancer. Br J Cancer 93:441–449
Zhang JF, Hu C, Geng Y et al (1996) Treatment of a human breast cancer xenograft with an adenovirus vector containing an interferon gene results in rapid regression due to viral oncolysis and gene therapy. Proc Natl Acad Sci USA 93:4513–4518
Iqbal Ahmed CM, Johnson DE, Demers GW et al (2001) Interferon alpha2b gene delivery using adenoviral vector causes inhibition of tumor growth in xenograft models from a variety of cancers. Cancer Gene Ther 8:788–795
Hottiger MO, Dam TN, Nickoloff BJ, Johnson TM, Nabel GJ (1999) Liposome-mediated gene transfer into human basal cell carcinoma. Gene Ther 6:1929–1935
Horton HM, Anderson D, Hernandez P, Barnhart KM, Norman JA, Parker SE (1999) A gene therapy for cancer using intramuscular injection of plasmid DNA encoding interferon alpha. Proc Natl Acad Sci USA 96:1553–1558
Hatanaka K, Suzuki K, Miura Y et al (2004) Interferon-alpha and antisense K-ras RNA combination gene therapy against pancreatic cancer. J Gene Med 6:1139
Hara H, Kobayashi A, Yoshida K et al (2007) Local interferon-alpha gene therapy elicits systemic immunity in a syngeneic pancreatic cancer model in hamster. Cancer Sci 98:455–463
Aoki K, Barker C, Danthinne X, Imperiale MJ, Nabel GJ (1999) Efficient generation of recombinant adenoviral vectors by Cre-lox recombination in vitro. Mol Med 5:224–231
Niwa H, Yamamura K, Miyazaki J (1991) Efficient selection for high-expression transfectants with a novel eukaryotic vector. Gene 108:193–199
Zhang WW, Koch PE, Roth JA (1995) Detection of wild-type contamination in a recombinant adenoviral preparation by PCR. Biotechniques 18:444–447
Nakayama E, Uenaka A (1985) Effect of in vivo administration of Lyt antibodies. Lyt phenotype of T cells in lymphoid tissues and blocking of tumor rejection. J Exp Med 161:345–355
Ohashi M, Kobayashi A, Hara H et al (2006) Allogeneic MHC gene transfer enhances antitumor activity of allogeneic hematopoietic stem cell transplantation without exacerbating graft-versus-host disease. Clin Cancer Res 12:2208–2215
Taguchi T (1986) Clinical studies of recombinant interferon alfa-2a (Roferon-A) in cancer patients. Cancer 57:1705–1708
Teshima T, Hill GR, Pan L et al (1999) IL-11 separates graft-versus-leukemia effects from graft-versus-host disease after bone marrow transplantation. J Clin Invest 104:317–325
Edinger M, Hoffmann P, Ermann J et al (2003) CD4 + CD25 + regulatory T cells preserve graft-versus-tumor activity while inhibiting graft-versus-host disease after bone marrow transplantation. Nat Med 9:1144–1150
Pillai AB, George TI, Dutt S, Teo P, Strober S (2007) Host NKT cells can prevent graft-versus-host disease and permit graft antitumor activity after bone marrow transplantation. J Immunol 178:6242–6251
Chorny A, Gonzalez-Rey E, Fernandez-Martin A, Ganea D, Delgado M (2006) Vasoactive intestinal peptide induces regulatory dendritic cells that prevent acute graft-versus-host disease while maintaining the graft-versus-tumor response. Blood 107:3787–3794
Zhang C, Lou J, Li N et al (2007) Donor CD8 + T cells mediate graft-versus-leukemia activity without clinical signs of graft-versus-host disease in recipients conditioned with anti-CD3 monoclonal antibody. J Immunol 178:838–850
Schmaltz C, Alpdogan O, Horndasch KJ et al (2001) Differential use of Fas ligand and perforin cytotoxic pathways by donor T cells in graft-versus-host disease and graft-versus-leukemia effect. Blood 97:2886–2895
Mapara MY, Kim YM, Wang SP, Bronson R, Sachs DH, Sykes M (2002) Donor lymphocyte infusions mediate superior graft-versus-leukemia effects in mixed compared to fully allogeneic chimeras: a critical role for host antigen-presenting cells. Blood 100:1903–1909
Billiau AD, Fevery S, Rutgeerts O, Landuyt W, Waer M (2002) Crucial role of timing of donor lymphocyte infusion in generating dissociated graft-versus-host and graft-versus-leukemia responses in mice receiving allogeneic bone marrow transplants. Blood 100:1894–1902
Michalek J, Collins RH, Durrani HP et al (2003) Definitive separation of graft-versus-leukemia- and graft-versus-host-specific CD4 + T cells by virtue of their receptor beta loci sequences. Proc Natl Acad Sci USA 100:1180–1184
Zhang Y, Joe G, Zhu J et al (2004) Dendritic cell-activated CD44hiCD8 + T cells are defective in mediating acute graft-versus-host disease but retain graft-versus-leukemia activity. Blood 103:3970–3978
Anderson LD Jr, Mori S, Mann S, Savary CA, Mullen CA (2000) Pretransplant tumor antigen-specific immunization of allogeneic bone marrow transplant donors enhances graft-versus-tumor activity without exacerbation of graft-versus-host disease. Cancer Res 60:5797–5802
Luznik L, Slansky JE, Jalla S et al (2003) Successful therapy of metastatic cancer using tumor vaccines in mixed allogeneic bone marrow chimeras. Blood 101:1645–1652
Perales MA, Diab A, Cohen AD et al (2006) DNA immunization against tissue-restricted antigens enhances tumor immunity after allogeneic hemopoietic stem cell transplantation. J Immunol 177:4159–4167
Teshima T, Mach N, Hill GR et al (2001) Tumor cell vaccine elicits potent antitumor immunity after allogeneic T-cell-depleted bone marrow transplantation. Cancer Res 61:162–171
Galanis E (2002) Technology evaluation: Allovectin-7, Vical. Curr Opin Mol Ther 4:80–87
Kayagaki N, Yamaguchi N, Nakayama M, Eto H, Okumura K, Yagita H (1999) Type I interferons (IFNs) regulate tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) expression on human T cells: a novel mechanism for the antitumor effects of type I IFNs. J Exp Med 189:1451–1460
Sato K, Hida S, Takayanagi H et al (2001) Antiviral response by natural killer cells through TRAIL gene induction by IFN-alpha/beta. Eur J Immunol 31:3138–3146
Tough DF, Borrow P, Sprent J (1996) Induction of bystander T cell proliferation by viruses and type I interferon in vivo. Science 272:1947–1950
Santini SM, Lapenta C, Logozzi M et al (2000) Type I interferon as a powerful adjuvant for monocyte-derived dendritic cell development and activity in vitro and in Hu-PBL-SCID mice. J Exp Med 191:1777–1788
Kobayashi A, Hara H, Ohashi M et al (2007) Allogeneic major histocompatibility complex gene transfer enhances an effective antitumor immunity in the early period of autologous hematopoietic stem cell transplantation. Clin Cancer Res (in press)
Anderson LD Jr, Savary CA, Mullen CA (2000) Immunization of allogeneic bone marrow transplant recipients with tumor cell vaccines enhances graft-versus-tumor activity without exacerbating graft-versus-host disease. Blood 95:2426–2433
Kurooka M, Kaneda Y (2007) Inactivated Sendai virus particles eradicate tumors by inducing immune responses through blocking regulatory T cells. Cancer Res 67:227–236
Acknowledgments
This work was supported in part by a grant-in-aid for the 3rd Term Comprehensive 10-year Strategy for Cancer Control from the Ministry of Health, Labour and Welfare of Japan, by grants-in-aid for Cancer Research from the Ministry of Health, Labour and Welfare of Japan and by the program for promotion of Fundamental Studies in Health Sciences of the National Institute of Biomedical Innovation (NiBio). H. Hara, T. Nishimoto and M. Ohashi are awardees of a Research Resident Fellowship from the Foundation for Promotion of Cancer Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hara, H., Kobayashi, A., Narumi, K. et al. Intratumoral interferon-α gene transfer enhances tumor immunity after allogeneic hematopoietic stem cell transplantation. Cancer Immunol Immunother 58, 1007–1021 (2009). https://doi.org/10.1007/s00262-008-0616-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-008-0616-0