Abstract
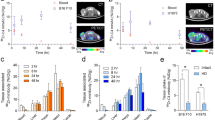
The Thomsen-Friedenreich disaccharide (TFα) is a promising antigen for tumor immunotargeting, since it is almost exclusively expressed on carcinoma tissues. So far, an obstacle preventing the exploitation of TF for immunotargeting has been the lack of suitable (non-IgM) antibodies with high affinity and specificity. Recently we reported on a novel strategy for generating antibodies toward small uncharged carbohydrates and the generation of recombinant antibodies toward TF. Among them, two multivalent scFv antibodies showed sub-micromolar functional affinities and appeared well suited for immunotargeting. In the present study, the trimeric scFv(1aa) and the tetrameric scFv(0aa) have been further developed for radioimmunotargeting. The scFvs were radiolabeled with 111In using DTPA as chelator without losing binding activity or molecular stoichiometry. Binding affinities as high as 1 × 10−7 M toward TF displayed on living cells were determined. Antibody biodistribution and tumor targeting efficacy were studied in TF-positive human breast cancer (ZR-75-1) bearing mice. TF was successfully targeted in vivo with tumor uptakes of ∼11 and 8% ID/g after 24 h for the trimeric and tetrameric scFv, respectively. These results validate TF as a potent antigen for tumor targeting. The biodistribution of the scFvs was comparable to that reported for IgGs. In contrast to the IgGs, the serum clearance of the scFvs was very fast, which could be an advantage in a therapeutic setting. Furthermore, kidney uptake, which is often critical for small recombinant antibodies labeled with radio-metals, was low with the tetramer (11% ID/g). We conclude that the multimeric anti-TF scFvs are promising candidates to be further developed toward therapeutic application.






Similar content being viewed by others
Abbreviations
- aa:
-
Amino acid
- DTPA:
-
Diethylenetriamine pentaacetic acid
- PAA:
-
Polyacrylamide
- RIA:
-
Radioimmunoassay
- scFv:
-
Single chain fragment variable
- TF:
-
Thomsen-Friedenreich antigen
- TLC:
-
Thin layer chromatography
- VH and VL :
-
Variable domain of heavy chain and light chain, respectively
- % ID/g:
-
Per cent injected dose per gram organ
- N :
-
Number of individual experiments
References
Adams GP, Schier R, McCall AM, Simmons HH, Horak EM, Alpaugh RK, Marks JD, Weiner LM (2001) High affinity restricts the localization and tumor penetration of single-chain fv antibody molecules. Cancer Res 61:4750–4755
Behr TM, Sharkey RM, Juweid ME, Blumenthal RD, Dunn RM, Griffiths GL, Bair HJ, Wolf FG, Becker WS, Goldenberg DM (1995) Reduction of the renal uptake of radiolabeled monoclonal antibody fragments by cationic amino acids and their derivatives. Cancer Res 55:3825–3834
Behr TM, Sharkey RM, Sgouros G, Blumenthal RD, Dunn RM, Kolbert K, Griffiths GL, Siegel JA, Becker WS, Goldenberg DM (1997) Overcoming the nephrotoxicity of radiometal-labeled immunoconjugates: improved cancer therapy administered to a nude mouse model in relation to the internal radiation dosimetry. Cancer 80:2591–2610
Cao Y, Karsten U, Liebrich W, Haensch W, Springer GF, Schlag PM (1995) Expression of Thomsen-Friedenreich-related antigens in primary and metastatic colorectal carcinomas: a reevaluation. Cancer 76:1700–1708
Cao Y, Stosiek P, Springer GF, Karsten U (1996) Thomsen-Friedenreich-related carbohydrate antigens in normal adult human tissues: a systematic and comparative study. Histochem Cell Biol 106:197–207
Clarke K, Lee FT, Brechbiel MW, Smyth FE, Old LJ, Scott AM (2000) In vivo biodistribution of a humanized anti-Lewis Y monoclonal antibody (hu3S193) in MCF-7 xenografted BALB/c nude mice. Cancer Res 60:4804–4811
Fukuda M, Lauffenburger M, Sasaki H, Rogers ME, Dell A (1987) Structures of novel sialylated O-linked oligosaccharides isolated from human erythrocyte glycophorins. J Biol Chem 262:11952–11957
Goldenberg DM (2003) Advancing role of radiolabeled antibodies in the therapy of cancer. Cancer Immunol Immunother 52:281–296
Goletz S, Cao Y, Danielczyk A, Ravn P, Schoeber U, Karsten U (2003) Thomsen-Friedenreich antigen: the “hidden” tumor antigen. Adv Exp Med Biol 535:147–162
Graff CP, Wittrup KD (2003) Theoretical analysis of antibody targeting of tumor spheroids: importance of dosage for penetration, and affinity for retention. Cancer Res 63:1288–1296
Holliger P, Prospero T, Winter G (1993) “Diabodies”: small bivalent and bispecific antibody fragments. Proc Natl Acad Sci USA 90:6444–6448
Hudson PJ, Souriau C (2003) Engineered antibodies. Nat Med 9:129–134
Hull SR, Carraway KL (1988) Mechanism of expression of Thomsen-Friedenreich (T) antigen at the cell surface of a mammary adenocarcinoma. FASEB J 2:2380–2384
Jain RK (1987) Transport of molecules in the tumor interstitium: a review. Cancer Res 47:3039–3051
Jain RK, Baxter LT (1988) Mechanisms of heterogeneous distribution of monoclonal antibodies and other macromolecules in tumors: significance of elevated interstitial pressure. Cancer Res 48:7022–7032
Juweid M, Neumann R, Paik C, Perez-Bacete MJ, Sato J, van Osdol W, Weinstein JN (1992) Micropharmacology of monoclonal antibodies in solid tumors: direct experimental evidence for a binding site barrier. Cancer Res 52:5144–5153
Lindmo T, Boven E, Cuttitta F, Fedorko J, Bunn PA (1984) Determination of the immunoreactive fraction of radiolabeled monoclonal antibodies by linear extrapolation to binding at infinite antigen excess. J Immunol Methods 72:77–89
Lloyd KO, Burchell J, Kudryashov V, Yin BW, Taylor-Papadimitriou J (1996) Comparison of O-linked carbohydrate chains in MUC-1 mucin from normal breast epithelial cell lines and breast carcinoma cell lines. Demonstration of simpler and fewer glycan chains in tumor cells. J Biol Chem 271:33325–33334
Lub-de Hooge MN, Kosterink JG, Perik PJ, Nijnuis H, Tran L, Bart J, Suurmeijer AJ, de Jong S, Jager PL, de Vries EG (2004) Preclinical characterisation of 111In-DTPA-trastuzumab. Br J Pharmacol 143:99–106
Mackenzie R, To R (1998) The role of valency in the selection of anti-carbohydrate single-chain Fvs from phage display libraries. J Immunol Methods 220:39–49
Potamianos S, Varvarigou AD, Archimandritis SC (2000) Radioimmunoscintigraphy and radioimmunotherapy in cancer: principles and application. Anticancer Res 20:925–948
Ravn P, Danielczyk A, Jensen KB, Kristensen P, Christensen PA, Larsen M, Karsten U, Goletz S (2004) Multivalent scFv display of phagemid repertoires for the selection of carbohydrate-specific antibodies and its application to the Thomsen-Friedenreich antigen. J Mol Biol 343:985–996
Rogers BE, Franano FN, Duncan JR, Edwards WB, Anderson CJ, Connett JM, Welch MJ (1995) Identification of metabolites of 111In-diethylenetriaminepentaacetic acid-monoclonal antibodies and antibody fragments in vivo. Cancer Res 55:5714s–5720s
Slovin SF, Ragupathi G, Musselli C, Fernandez C, Diani M, Verbel D, Danishefsky S, Livingston P, Scher HI (2005) Thomsen-Friedenreich (TF) antigen as a target for prostate cancer vaccine: clinical trial results with TF cluster (c)-KLH plus QS21 conjugate vaccine in patients with biochemically relapsed prostate cancer. Cancer Immunol Immunother 54:694–702
Springer GF (1984) T and Tn, general carcinoma autoantigens. Science 224:1198–1206
Springer GF (1997) Immunoreactive T and Tn epitopes in cancer diagnosis, prognosis, and immunotherapy. J Mol Med 75:594–602
Tahtis K, Lee FT, Smyth FE, Power BE, Renner C, Brechbiel MW, Old LJ, Hudson PJ, Scott AM (2001) Biodistribution properties of (111)indium-labeled C-functionalized trans-cyclohexyl diethylenetriaminepentaacetic acid humanized 3S193 diabody and F(ab’)(2) constructs in a breast carcinoma xenograft model. Clin Cancer Res 7:1061–1072
Todorovska A, Roovers RC, Dolezal O, Kortt AA, Hoogenboom HR, Hudson PJ (2001) Design and application of diabodies, triabodies and tetrabodies for cancer targeting. J Immunol Methods 248:47–66
Pisano A, Redmond JW, Williams KL, Gooley AA (1993) Glycosylation sites identified by solid-phase Edman degradation: O-linked glycosylation motifs on human glycophorin A. Glycobiology 3:429–435
Tsai SW, Li L, Williams LE, Anderson AL, Raubitschek AA, Shively JE (2001) Metabolism and renal clearance of 111In-labeled DOTA-conjugated antibody fragments. Bioconjug Chem 12:264–270
van Osdol W, Fujimori K, Weinstein JN (1991) An analysis of monoclonal antibody distribution in microscopic tumor nodules: consequences of a “binding site barrier”. Cancer Res 51:4776–4784
von Mehren M, Adams GP, Weiner LM (2003) Monoclonal antibody therapy for cancer. Annu Rev Med 54:343–369
Weinstein JN, van OsdolW (1992) Early intervention in cancer using monoclonal antibodies and other biological ligands: micropharmacology and the “binding site barrier”. Cancer Res 52:2747s–2751s
Willuda J, Honegger A, Waibel R, Schubiger PA, Stahel R, Zangemeister-Wittke U, Pluckthun A (1999) High thermal stability is essential for tumor targeting of antibody fragments: engineering of a humanized anti-epithelial glycoprotein-2 (epithelial cell adhesion molecule) single-chain Fv fragment. Cancer Res 59:5758–5767
Willuda J, Kubetzko S, Waibel R, Schubiger PA, Zangemeister-Wittke U, Pluckthun A (2001) Tumor targeting of mono-, di-, and tetravalent anti-p185(HER-2) miniantibodies multimerized by self-associating peptides. J Biol Chem 276:14385–14392
Yazaki PJ, Wu AM, Tsai SW, Williams LE, Ikler DN, Wong JY, Shively JE, Raubitschek AA (2001) Tumor targeting of radiometal labeled anti-CEA recombinant T84.66 diabody and t84.66 minibody: comparison to radioiodinated fragments. Bioconjug Chem 12:220–228
Acknowledgments
The work was supported by the Danish Research Training Council (Forskeruddannelsesrådet), NEMOD Biotherapeutics GmbH & Co.KG, and Glycotope GmbH.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Fig7
Supplementary Fig. 1 DTPA conjugation conditions and quality analysis after conjugation. a–c Binding activity of the scFv(1aa) after incubation in three different buffers used during the DTPA conjugation at different temperatures (N = 2). d Size exclusion chromatography of the chelated versus the nonchelated scFv(1aa). e Binding activity of the chelated versus the nonchelated scFv(1aa) to asialoglycophorin
Fig8
Supplementary Fig. 2 Quality analysis of 111In labeling. a Thin layer chromatography of labeled protein. Lanes: 1, free 111In; 2, 111In-labeled unbound DTPA (111In in excess); 3, 111In-labeled scFv(1aa). b Radiography of SDS-PAGE. 111In-labeled scFv(1aa) was loaded on SDS-PAGE and visualised via radiography. c Size exclusion chromatography. 111In-labeled scFv(1aa) and an 111In-labeled control mouse IgG were examined in size exclusion chromatography
Rights and permissions
About this article
Cite this article
Ravn, P., Stahn, R., Danielczyk, A. et al. The Thomsen-Friedenreich disaccharide as antigen for in vivo tumor targeting with multivalent scFvs. Cancer Immunol Immunother 56, 1345–1357 (2007). https://doi.org/10.1007/s00262-007-0292-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-007-0292-5