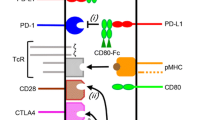
Abstract
Blockade of CTLA-4 by monoclonal antibodies (mAb) can mediate regression of tumors and increase the efficacy of tumor antigen specific vaccines. Blockade of CTLA-4 has also been shown to significantly increase the avidity of antigen-specific T cells after immunization with live recombinant viral vector based vaccine. Here, we demonstrate a biological synergy between CTLA-4 blockade and active vaccine therapy consisting of recombinant vaccinia and avipox viruses expressing carcinoembryonic antigen (CEA) and three T cell costimulatory molecules to enhance antitumor effects. However, this synergy was very much dependent on the temporal relationship of scheduling of the two agents. We evaluated the strategies in both a foreign antigen model using β-galactosidase as immunogen, and in a “self” antigen model using CEA as immunogen. For antitumor activity the model used consisted of mice transgenic for human CEA and a murine carcinoma cell line transfected with CEA. The enhanced antitumor activity after vaccine and CTLA-4 blockade did not result in any signs of autoimmunity. These studies form a rational basis for the use of vector-based vaccines with anti-CTLA-4 and demonstrate that both enhancement of positive costimulatory signals and inhibition of negative costimulatory signals can be simultaneously exploited. These studies also underscore the importance of “drug” scheduling in vaccine combination therapies.






Similar content being viewed by others
References
Allison JP, Krummel MF (1995) The Yin and Yang of T cell costimulation. Science 270(5238):932–933
Hilburger Ryan M, Abrams SI (2001) Characterization of CD8+ cytotoxic T lymphocyte/tumor cell interactions reflecting recognition of an endogenously expressed murine wild-type p53 determinant. Cancer Immunol Immunother 49(11):603–612
Leach DR, Krummel MF, Allison JP (1996) Enhancement of antitumor immunity by CTLA-4 blockade. Science 271(5256):1734–1736
Yang YF, Zou JP, Mu J, Wijesuriya R, Ono S, Walunas T et al (1997) Enhanced induction of antitumor T-cell responses by cytotoxic T lymphocyte-associated molecule-4 blockade: the effect is manifested only at the restricted tumor-bearing stages. Cancer Res 57(18):4036–4041
van Elsas A, Hurwitz AA, Allison JP (1999) Combination immunotherapy of B16 melanoma using anti-cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) and granulocyte/macrophage colony-stimulating factor (GM-CSF)-producing vaccines induces rejection of subcutaneous and metastatic tumors accompanied by autoimmune depigmentation. J Exp Med 190(3):355–366
Hurwitz AA, Foster BA, Kwon ED, Truong T, Choi EM, Greenberg NM et al (2000) Combination immunotherapy of primary prostate cancer in a transgenic mouse model using CTLA-4 blockade. Cancer Res 60(9):2444–2448
Pardoll D (2003) Does the immune system see tumors as foreign or self? Annu Rev Immunol 21:807–839
Engelhard VH, Bullock TN, Colella TA, Sheasley SL, Mullins DW (2002) Antigens derived from melanocyte differentiation proteins: self-tolerance, autoimmunity, and use for cancer immunotherapy. Immunol Rev 188:136–146
Guevara-Patino JA, Turk MJ, Wolchok JD, Houghton AN (2003) Immunity to cancer through immune recognition of altered self: studies with melanoma. Adv Cancer Res 90:157–177
Hodge JW, Chakraborty M, Kudo-Saito C, Garnett CT, Schlom J (2005) Multiple costimulatory modalities enhance CTL avidity. J Immunol 174(10):5994–6004
Schlom J, Tsang KY, Kantor JA, Abrams SI, Zaremba S, Greiner J et al (1999) Strategies in the development of recombinant vaccines for colon cancer. Semin Oncol 26(6):672–682
Eades-Perner AM, van der Putten H, Hirth A, Thompson J, Neumaier M, von Kleist S et al (1994) Mice transgenic for the human carcinoembryonic antigen gene maintain its spatiotemporal expression pattern. Cancer Res 54(15):4169–4176
Kass E, Schlom J, Thompson J, Guadagni F, Graziano P, Greiner JW (1999) Induction of protective host immunity to carcinoembryonic antigen (CEA), a self-antigen in CEA transgenic mice, by immunizing with a recombinant vaccinia-CEA virus. Cancer Res 59(3):676–683
Hodge JW, McLaughlin JP, Kantor JA, Schlom J (1997) Diversified prime and boost protocols using recombinant vaccinia virus and recombinant non-replicating avian pox virus to enhance T-cell immunity and antitumor responses. Vaccine 15(6–7):759–768
Hodge JW, Sabzevari H, Yafal AG, Gritz L, Lorenz MG, Schlom J (1999) A triad of costimulatory molecules synergize to amplify T-cell activation. Cancer Res 59(22):5800–5877
Kass E, Panicali DL, Mazzara G, Schlom J, Greiner JW (2001) Granulocyte/macrophage-colony stimulating factor produced by recombinant avian poxviruses enriches the regional lymph nodes with antigen-presenting cells and acts as an immunoadjuvant. Cancer Res 61(1):206–214
Robbins PF, Kantor JA, Salgaller M, Hand PH, Fernsten PD, Schlom J (1991) Transduction and expression of the human carcinoembryonic antigen gene in a murine colon carcinoma cell line. Cancer Res 51(14):3657–3662
Krummel MF, Allison JP (1995) CD28 and CTLA-4 have opposing effects on the response of T cells to stimulation. J Exp Med 182(2):459–465
Kantor J, Irvine K, Abrams S, Kaufman H, DiPietro J, Schlom J (1992) Antitumor activity and immune responses induced by a recombinant carcinoembryonic antigen-vaccinia virus vaccine. J Natl Cancer Inst 84(14):1084–1091
Bergmann-Leitner ES, Abrams SI (2001) Treatment of human colon carcinoma cell lines with anti-neoplastic agents enhances their lytic sensitivity to antigen-specific CD8+ cytotoxic T lymphocytes. Cancer Immunol Immunother 50(9):445–455
Chakraborty M, Abrams SI, Coleman CN, Camphausen K, Schlom J, Hodge JW (2004) External beam radiation of tumors alters phenotype of tumor cells to render them susceptible to vaccine-mediated T-cell killing. Cancer Res 64(12):4328–4337
Ercolini AM, Ladle BH, Manning EA, Pfannenstiel LW, Armstrong TD, Machiels JP et al (2005) Recruitment of latent pools of high-avidity CD8(+) T cells to the antitumor immune response. J Exp Med 201(10):1591–1602
Holmberg K, Mariathasan S, Ohteki T, Ohashi PS, Gascoigne NR (2003) TCR binding kinetics measured with MHC class I tetramers reveal a positive selecting peptide with relatively high affinity for TCR. J Immunol 171(5):2427–2434
Wang M, Bronte V, Chen PW, Gritz L, Panicali D, Rosenberg SA et al (1995) Active immunotherapy of cancer with a nonreplicating recombinant fowlpox virus encoding a model tumor-associated antigen. J Immunol 154(9):4685–4692
Aarts WM, Schlom J, Hodge JW (2002) Vector-based vaccine/cytokine combination therapy to enhance induction of immune responses to a self-antigen and antitumor activity. Cancer Res 62(20):5770–5777
Egen JG, Kuhns MS, Allison JP (2002) CTLA-4: new insights into its biological function and use in tumor immunotherapy. Nat Immunol 3(7):611–618
van Elsas A, Sutmuller RP, Hurwitz AA, Ziskin J, Villasenor J, Medema JP et al (2001) Elucidating the autoimmune and antitumor effector mechanisms of a treatment based on cytotoxic T lymphocyte antigen-4 blockade in combination with a B16 melanoma vaccine: comparison of prophylaxis and therapy. J Exp Med 194(4):481–489
Phan GQ, Yang JC, Sherry RM, Hwu P, Topalian SL, Schwartzentruber DJ et al (2003) Cancer regression and autoimmunity induced by cytotoxic T lymphocyte-associated antigen 4 blockade in patients with metastatic melanoma. Proc Natl Acad Sci USA 100(14):8372–8377
Phan GQ, Touloukian CE, Yang JC, Restifo NP, Sherry RM, Hwu P et al (2003) Immunization of patients with metastatic melanoma using both class I- and class II-restricted peptides from melanoma-associated antigens. J Immunother 26(4):349–356
Egen JG, Allison JP (2002) Cytotoxic T lymphocyte antigen-4 accumulation in the immunological synapse is regulated by TCR signal strength. Immunity 16(1):23–35
Espenschied J, Lamont J, Longmate J, Pendas S, Wang Z, Diamond DJ et al (2003) CTLA-4 blockade enhances the therapeutic effect of an attenuated poxvirus vaccine targeting p53 in an established murine tumor model. J Immunol 170(6):3401–3407
Gregor PD, Wolchok JD, Ferrone CR, Buchinshky H, Guevara-Patino JA, Perales MA et al (2004) CTLA-4 blockade in combination with xenogeneic DNA vaccines enhances T-cell responses, tumor immunity and autoimmunity to self antigens in animal and cellular model systems. Vaccine 22(13–14):1700–1708
Davila E, Kennedy R, Celis E (2003) Generation of antitumor immunity by cytotoxic T lymphocyte epitope peptide vaccination, CpG-oligodeoxynucleotide adjuvant, and CTLA-4 blockade. Cancer Res 63(12):3281–3288
Sutmuller RP, van Duivenvoorde LM, van Elsas A, Schumacher TN, Wildenberg ME, Allison JP et al (2001) Synergism of cytotoxic T lymphocyte-associated antigen 4 blockade and depletion of CD25(+) regulatory T cells in antitumor therapy reveals alternative pathways for suppression of autoreactive cytotoxic T lymphocyte responses. J Exp Med 194(6):823–832
Hernandez J, Ko A, Sherman LA (2001) CTLA-4 blockade enhances the CTL responses to the p53 self-tumor antigen. J Immunol 166(6):3908–3914
Hurwitz AA, Yu TF, Leach DR, Allison JP (1998) CTLA-4 blockade synergies with tumor-derived granulocyte-macrophage colony-stimulating factor for treatment of an experimental mammary carcinoma. Proc Natl Acad Sci USA 95(17):10067–10071
Allison JP, Chambers C, Hurwitz A, Sullivan T, Boitel B, Fournier S et al (1998) A role for CTLA-4-mediated inhibitory signals in peripheral T cell tolerance? Novartis Found Symp 215:92–8; discussion 98–102, 86–90
Read S, Malmstrom V, Powrie F (2000) Cytotoxic T lymphocyte-associated antigen 4 plays an essential role in the function of CD25(+)CD4(+) regulatory cells that control intestinal inflammation. J Exp Med 192(2):295–302
Kudo-Saito C, Schlom J, Hodge JW (2005) Induction of an antigen cascade by diversified subcutaneous/intratumoral vaccination is associated with antitumor responses. Clin Cancer Res 11(6):2416–2426
Pedersen AE, Buus S, Claesson MH (2005) Treatment of transplanted CT26 tumour with dendritic cell vaccine in combination with blockade of vascular endothelial growth factor receptor 2 and CTLA-4. Cancer Lett 235(2):229–238
Perrin PJ, Maldonado JH, Davis TA, June CH, Racke MK (1996) CTLA-4 blockade enhances clinical disease and cytokine production during experimental allergic encephalomyelitis. J Immunol 157(4):1333–1336
Zhu J, Zou L, Zhu S, Mix E, Shi F, Wang H et al (2001) Cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) blockade enhances incidence and severity of experimental autoimmune neuritis in resistant mice. J Neuroimmunol 115(1–2):111–117
Luhder F, Hoglund P, Allison JP, Benoist C, Mathis D (1998) Cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) regulates the unfolding of autoimmune diabetes. J Exp Med 187(3):427–432
Wang HB, Shi FD, Li H, Chambers BJ, Link H, Ljunggren HG (2001) Anti-CTLA-4 antibody treatment triggers determinant spreading and enhances murine myasthenia gravis. J Immunol 166(10):6430–6436
Hodi FS, Mihm MC, Soiffer RJ, Haluska FG, Butler M, Seiden MV et al (2003) Biologic activity of cytotoxic T lymphocyte-associated antigen 4 antibody blockade in previously vaccinated metastatic melanoma and ovarian carcinoma patients. Proc Natl Acad Sci U S A 100(8):4712–4717
Sanderson K, Scotland R, Lee P, Liu D, Groshen S, Snively J et al (2005) Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III and IV melanoma. J Clin Oncol 23(4):741–750
Ji Q, Gondek D, Hurwitz AA (2005) Provision of granulocyte-macrophage colony-stimulating factor converts an autoimmune response to a self-antigen into an antitumor response. J Immunol 175(3):1456–1463
Acknowledgments
This research was supported by the Intramural Research Program of the NIH, National Cancer Institute, Center of Cancer Research. The authors thank Dr. Scott Abrams and Dr. Connie Rogers for helpful review of this material and Debra Weingarten for her editorial assistance in the preparation of this paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chakraborty, M., Schlom, J. & Hodge, J.W. The combined activation of positive costimulatory signals with modulation of a negative costimulatory signal for the enhancement of vaccine-mediated T-cell responses. Cancer Immunol Immunother 56, 1471–1484 (2007). https://doi.org/10.1007/s00262-007-0291-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-007-0291-6