Abstract
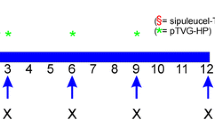
T cell immunotherapy of prostate cancer (CaP) offers the potential for less toxic, more effective outcomes. A clinical trial was conducted in 28 patients with locally advanced or metastatic CaP to determine whether an HLA-A2 binding epitope of prostate-specific antigen, PSA146–154 (PSA-peptide), can induce specific T cell immunity. Patients were vaccinated either by intradermal injection of PSA-peptide and GM-CSF or by intravenous administration of autologous dendritic cells pulsed with PSA-peptide at weeks 1, 4 and 10. Delayed-type hypersensitivity (DTH) skin testing was performed at weeks 4, 14, 26 and 52. Fifty percent of the patients developed positive DTH responses to PSA-peptide. The size of the DTH induration progressively increased over time in the majority of responding patients. Skin biopsies from seven DTH-positive patients were available and T cells that developed in situ were also characterized. The phenotype of recovered T cells demonstrated variable proportions of CD4+CD8−, CD4−CD8+ and CD4+CD8+ T cell populations. Cytokine analysis of PSA-peptide stimulated T cells per bead array assay exhibited specific IFN-γ and TNF-α response in six of seven patients. Specific IL-4 response was observed in five patients, while IL-10 response was detected in one patient. Purified CD4−CD8+ T cells isolated from four patients demonstrated specific cytolytic activity per chromium release assay. In conclusion, immunization with PSA-peptide induced specific T cell immunity in one-half of the patients with locally advanced and hormone-sensitive, metastatic CaP. DTH-derived T cells exhibited PSA-peptide-specific cytolytic activity and predominantly expressed a type-1 cytokine profile.




Similar content being viewed by others
Abbreviations
- CaP:
-
Prostate cancer
- CBA:
-
Cytokine bead array
- CRA:
-
Chromium release assay
- DC:
-
Dendritic cells
- DMSO:
-
Dimethyl sulfoxide
- DTH:
-
Delayed-type hypersensitivity
- PSA:
-
Prostate-specific antigen
- PSA:
-
Peptide-PSA146–154 peptide
References
Barrou B, Benoit G, Ouldkaci M, Cussenot O, Salcedo M, Agrawal S, Massicard S, Bercovici N, Ericson ML, Thiounn N (2004) Vaccination of prostatectomized prostate cancer patients in biochemical relapse, with autologous dendritic cells pulsed with recombinant human PSA. Cancer Immunol Immunother 53:453–460
Constant SL, Bottomly K (1997) Induction of Th1 and Th2 CD4+ T cell responses: the alternative approaches. Annu Rev Immunol 15:297–322
De Vries IJ, Krooshoop DJ, Scharenborg NM, Lesterhuis WJ, Diepstra JH, Van Muijen GN, Strijk SP, Ruers TJ, Boerman OC, Oyen WJ, Adema GJ, Punt CJ, Figdor CG (2003) Effective migration of antigen-pulsed dendritic cells to lymph nodes in melanoma patients is determined by their maturation state. Cancer Res 63:12–17
Dieckmann D, Schultz ES, Ring B, Chames P, Held G, Hoogenboom HR, Schuler G (2005) Optimizing the exogenous antigen loading of monocyte-derived dendritic cells. Int Immunol 17:621–635
Disis ML, Schiffman K, Gooley TA, McNeel DG, Rinn K, Knutson KL (2000) Delayed-type hypersensitivity response is a predictor of peripheral blood T-cell immunity after HER-2/neu peptide immunization. Clin Cancer Res 6:1347–1350
Eder JP, Kantoff PW, Roper K, Xu GX, Bubley GJ, Boyden J, Gritz L, Mazzara G, Oh WK, Arlen P, Tsang KY, Panicali D, Schlom J, Kufe DW (2000) A phase I trial of a recombinant vaccinia virus expressing prostate-specific antigen in advanced prostate cancer. Clin Cancer Res 6:1632–1638
Galea-Lauri J, Wells JW, Darling D, Harrison P, Farzaneh F (2004) Strategies for antigen choice and priming of dendritic cells influence the polarization and efficacy of antitumor T-cell responses in dendritic cell-based cancer vaccination. Cancer Immunol Immunother 53:963–977
Gulley JL, Arlen PM, Bastian A, Morin S, Marte J, Beetham P, Tsang KY, Yokokawa J, Hodge JW, Menard C, Camphausen K, Coleman CN, Sullivan F, Steinberg SM, Schlom J, Dahut W (2005) Combining a recombinant cancer vaccine with standard definitive radiotherapy in patients with localized prostate cancer. Clin Cancer Res 11:3353–3362
Heiser A, Coleman D, Dannull J, Yancey D, Maurice MA, Lallas CD, Dahm P, Niedzwiecki D, Gilboa E, Vieweg J (2002) Autologous dendritic cells transfected with prostate-specific antigen RNA stimulate CTL responses against metastatic prostate tumors. J Clin Invest 109:409–417
Lau R, Wang F, Jeffery G, Marty V, Kuniyoshi J, Bade E, Ryback ME, Weber J (2001) Phase I trial of intravenous peptide-pulsed dendritic cells in patients with metastatic melanoma. J Immunother 24:66–78
Li TS, Beling CG (1973) Isolation and characterization of two specific antigens of human seminal plasma. Fertil Steril 24:134–144
Meidenbauer N, Harris DT, Spitler LE, Whiteside TL (2000) Generation of PSA-reactive effector cells after vaccination with a PSA-based vaccine in patients with prostate cancer. Prostate 43:88–100
Morse MA, Coleman RE, Akabani G, Niehaus N, Coleman D, Lyerly HK (1999) Migration of human dendritic cells after injection in patients with metastatic malignancies. Cancer Res 59:56–58
Nair SK, Hull S, Coleman D, Gilboa E, Lyerly HK, Morse MA (1999) Induction of carcinoembryonic antigen (CEA)-specific cytotoxic T-lymphocyte responses in vitro using autologous dendritic cells loaded with CEA peptide or CEA RNA in patients with metastatic malignancies expressing CEA. Int J Cancer 82:121–124
Nestle FO, Alijagic S, Gilliet M, Sun Y, Grabbe S, Dummer R, Burg G, Schadendorf D (1998) Vaccination of melanoma patients with peptide- or tumor lysate-pulsed dendritic cells. Nat Med 4:328–332
Nestle FO, Farkas A, Conrad C (2005) Dendritic-cell-based therapeutic vaccination against cancer. Curr Opin Immunol 17:163–169
Perambakam S., Xue B-H, Sosman JA, Peace DJ (2002) Induction of Tc2 cells with specificity for prostate-specific antigen from patients with hormone-refractory prostate cancer. Cancer Immunol Immunother 51:263–270
Perambakam SM, Srivastava R, Peace DJ (2005) Distinct cytokine patterns exist in peripheral blood mononuclear cell cultures of patients with prostate cancer. Clin Immunol 117: 94–99
Reichardt VL, Brossart P, Kanz L (2004) Dendritic cells in vaccination therapies of human malignant disease. Blood Rev 18:235–243
Romagnani S (1997) The Th1/Th2 paradigm. Immunol Today 18:263–266
Sanda MG, Smith DC, Charles LG, Hwang C, Pienta KJ, Schlom J, Milenic D, Panicali D, Montie JE (1999) Recombinant vaccinia-PSA (PROSTVAC) can induce a prostate-specific immune response in androgen-modulated human prostate cancer. Urology 53:260–266
Stewart SL, King JB, Thompson TD, Friedman C, Wingo PA (2004) Cancer mortality surveillance—United States, 1990–2000. MMWR Surveill Summ 53:1–108
Waanders GA, Rimoldi D, Lienard D, Carrel S, Lejeune F, Dietrich PY, Cerottini JC, Romero P (1997) Melanoma-reactive human cytotoxic T lymphocytes derived from skin biopsies of delayed-type hypersensitivity reactions induced by injection of an autologous melanoma cell line. Clin Cancer Res 3:685–696
Watt KW, Lee PJ, M’Timkulu T, Chan WP, Loor R (1986) Human prostate-specific antigen: structural and functional similarity with serine proteases. Proc Natl Acad Sci USA 83:3166–3170
Walker EB, Haley D, Miller W, Floyd K, Wisner KP, Sanjuan N, Maecker H, Romero P, Hu HM, Alvord WG, Smith JW II, Fox BA, Urba WJ (2004) Gp100(209-2 M) peptide immunization of human lymphocyte antigen-A2+ stage I-III melanoma patients induces significant increase in antigen-specific effector and long-term memory CD8+ T cells. Clin Cancer Res 10:668–680
Xue B-H, Zhang Y, Sosman JA, Peace DJ (1997) Induction of human cytotoxic T lymphocytes specific for prostate-specific antigen. Prostate 30:73–78
Acknowledgements
This work was supported by the National Cancer Institute (CA88062), the Department of Army (DAMD17-98-1-8489), the American Cancer Society (IRG 99-224-01), the Illinois Department of Public Health (prostate cancer research fund 4328301), the Cancer Research Institute, New York City, and the General Clinical Research Center of University of Illinois, Chicago, funded by NIH grant M01-RR-13987. The contents in the manuscript are solely the responsibility of the authors and do not necessarily reflect the official views of the respective funding agencies. The authors would like to thank Susan Nishimura and Susan Cascio for their excellent clinical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Perambakam, S., Hallmeyer, S., Reddy, S. et al. Induction of specific T cell immunity in patients with prostate cancer by vaccination with PSA146–154 peptide. Cancer Immunol Immunother 55, 1033–1042 (2006). https://doi.org/10.1007/s00262-005-0090-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-005-0090-x