Abstract
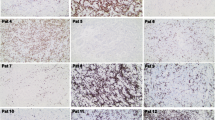
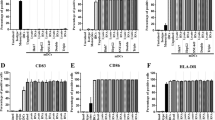
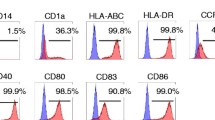
Adoptive immunotherapy with antitumor effector cells is an attractive therapeutic approach in metastatic renal cell carcinoma (RCC). The aim of the work was to enhance in vitro activation of lymphocytes with optimal cytotoxic activity against tumor cells. We evaluated a procedure based on the use of dendritic cells (DCs) loaded with irradiated tumor cells (DC-Tu) to stimulate lymphocytes. Experimental conditions were established with cells from healthy donors and melanoma cell lines. Procedures were then applied to cells from RCC patients. A total of 30 tumor biopsies, 14 proximal lymph nodes, and 17 peripheral blood samples from 30 patients were used. When lymphocytes were stimulated in vitro with DC-Tu, they responded to tumor cells with an increased cytolytic activity for all the assays with donor cells (n=18). For RCC patients, DC-Tu stimulation improved the final cytotoxic activity in only half of the assays (16/31). When significantly enhanced (>10%, n=8), responder cells resulted in a final 43% cytotoxicity against autologous RCC cells. Mechanism of lysis was at least in part class I mediated. Effector cells have no lytic activity against normal renal cells. Percentage of cells with regulatory T-cell phenotype was not found to be enhanced in the DC-Tu stimulated lymphocytes. Individual differences were observed in the characteristics of DCs generated from RCC patients in contrast to that observed in donors and could explain why lymphocyte stimulation was not improved by DC-Tu in half of the RCC assays. T-cell spreading was suitable for a therapeutic use (>109 cells) irrespective of the procedure (with or without DC-Tu stimulation) or the tissular origin of lymphocytes from patients. Data show that precursors of selective antitumor effector cells are present in patients with RCC and can be amplified in vitro either with or without DC-Tu stimulation. One of these populations could be chosen for an adoptive transfer immunotherapy.


Similar content being viewed by others
References
Almand B, Clark JI et al (2001) Increased production of immature myeloid cells in cancer patients: a mechanism of immunosuppression in cancer. J Immunol 166:678–689
Belldegrun A., W. Pierce et al (1993) Interferon-α primed tumor-infiltrating lymphocytes combined with interleukin-2 and interferon-α as therapy for metastatic renal cell carcinoma. J Urol 150:1384–1390
Bernhard H, Karbach J et al (1994) Cellular immune response to human rena-cell carcinomas: definition of a common antigen recognized by HLA-A2-restricted cytotoxic T-lymphocyte (CTL) clones. Int J Cancer 59:837–842
Boisteau O, Gautier F et al (1997) Apoptosis induced by sodium butyrate treatment increases immunogenicity of a rat colon tumor cell line. Apoptosis 1997:403–412
Bouet-Toussaint F, Genetet N et al (2000) Interleukin 2 expanded lymphocytes from lymph node and tumor biopsies of human renal cell carcinoma, breast and ovarian cancer. Eur Cytokin Network 11(2):217–224
Caignard A, Guillard M et al (1996) In situ demonstration of renal-cell-carcinoma-specific T-cell clones. Int J Cancer 66:564–570
Delneste Y, Magistrelli G et al (2002) Involvement of LOX-1 in dendritic cell mediated antigen cross-presentation. Immunity 17(3):353
Dudley M, Wunderlich J et al (2002) A phase I study of nonmyeloablative chemotherapy and adoptive transfer of autologous tumor-antigen specific T lymphocytes in patients with metastatic melanoma. J Immunother 25(3):243–51
Figlin R, Pierce W et al (1997) Treatment of metastatic renal cell carcinoma with nephrectomy, interleukin-2 and cytokine-primed or CD8+ selected tumor infiltrating lymphocytes from primary tumor. J Urol 158:740–745
Figlin RA, Thompson JA et al (1999) Multicenter, randomized, phase III trial of CD8+ tumor-infiltrating lymphocytes in combination with recombinant interleukin-2 in metastatic renal cell carcinoma. J Clin Oncol 17:2521–2529
Finke JH, Rayman P et al (1992) Characterisation of a human renal cell carcinoma specific cytotoxic CD8+ T cell line. J Immunother 11:1–11
Fuhrman SA, Lasky LC et al (1982) Prognostic significance of morphologic parameters in renal cell carcinoma. Am J Surg Pathol 6(7):655–663
Goey SH, Verweij J et al (1996) Immunotherapy of metastatic renal cell cancer. Annals of Oncology 7:887–900
Groh V, Rhinehart R et al (2001) Costimulation of CD8αβTcells by NKG2D via engagement by MIC induced on virus-infected cells. Nature Immunology 2:255–260
Guinan P, Sobin LH et al (1997) TNM staging of renal cell carcinoma: workgroup No. 3. Union International Contre le Cancer (UICC) and the American Joint Committee on Cancer (AJCC). Cancer 80(5): 992–993
Hänninen EL, Kirchner H et al (1996) Interleukin-2 based home therapy of metastatic renal cell carcinoma: risks and benefits in 215 consecutive single institution patients. J Urol 155:19–25
Hayakawa M, Hatano T et al (1994) Treatment of advanced renal cell carcinoma using regional arterial administration of lymphokine-activated killer cells in combination with low doses of rIL-2. Urol Int 53:117–124
Hoffmann T, Meidenbauer N et al (2000) Generation of tumor-specific T-lymphocytes by cross-priming human dendritic cells ingesting apoptotic tumor cells. Cancer Res 60(13):3542–3549
Jonuleit H, Schmitt E et al (2001) Dendritic cells as a tool to induce anergic and regulatory T cells. Trends in immunology 22(7):394–400
Jotereau F, Pandolfino MC et al (1991) High fold expansion of human cytotoxic t-lymphocytes specific for autologous melanoma cells for use in immunotherapy. J Immunother 10:405–411
Kugler A, Stuhler G et al (2000) Regression of human metastatic renal cell carcinoma after vaccination with tumor cell-dendritic cell hybrids. Nat Med 6(3):332–336
Liau LM, Black KL et al (1999) Treatment of intracranial gliomas with bone marrow-derived dendritic cells pulsed with tumor antigens. J Neurosurg 90:1115–1124
Mathiot C, Thiounn N et al (1995) Non-cytotoxic CD4 Tumour-infiltrating lymphocytes induce responses in patients with metastatic renal cell carcinoma previously treated with interleukin-2. Eur J Cancer 31A(9):1552–1553
Menetrier-Caux C, Thomachot M et al (2001) IL-4 prevents the blockade of dendritic cell differentiation induced by tumor cells. Cancer Res 61(7):3096–3104
Motzer RJ, Mazumdar M et al (1999) Survival and prognostic stratification of 670 patients with advanced renal cell carcinoma. J Clin Oncol 17(8):2530–2540
Mulders P, Tso C-L et al (1999) Presentation of renal tumor antigens by human dendritic cells activates tumor-infiltrating lymphocytes against autologous tumor: implications for live kidney cancer vaccines. Clin Cancer Res 5:445–454
Negrier S, Escudier B et al (1998) Recombinant human interleukin-2, recombinant human interferon alpha-2a, or both in metastatic renal-cell carcinoma. N Engl J Med 338:1272–1278
Nestlé FO, Alijagic S et al (1998) Vaccination of melanoma patients with peptide- or tumor lysate-pulsed dendritic cells. Nat Med 4(3):328–332
Sallusto F, Lanzavecchia A (1994) Efficient presentation of soluble antigen by cultured human dendritic cells is maintained by granulocyte/macrophage colony-stimulating factor plus interleukin 4 and downregulated by tumor necrosis factor alpha. J Exp Med 179(4):1109–1118
Thurnher M, Radamyr C et al (1996) Human renal-cell carcinoma tissue contains dendritic cells. Int J Cancer 68:1–7
Weng NP, Levine BL et al (1995) Human naive and memory T lymphocytes differ in telomeric lenght and replicative potential. Proc Natl Acad Sci USA 92:11091–11094
Acknowledgements
We are grateful to Marc Grégoire (INSERM U419, Nantes) for helpful discussion of the data.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by grants from the Comité Grand Ouest de La Ligue Contre le Cancer and from the Faculté de Médecine de Rennes.
Rights and permissions
About this article
Cite this article
Bouet-Toussaint, F., Patard, JJ., Gervais, A. et al. Cytotoxic effector cells with antitumor activity can be amplified ex vivo from biopsies or blood of patients with renal cell carcinoma for cell therapy use. Cancer Immunol Immunother 52, 699–707 (2003). https://doi.org/10.1007/s00262-003-0412-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-003-0412-9